1. Potassium exists in living systems as the potassium ion, K. Fill in the energy-level diagram below for the K+ ion, being sure to follow the aufbau principle (orbital filling) and Hund's rule (electron pairing rule) for energy-level diagrams (8.4). 4p 3d 4s 3p 3s 2p 2s 1s Energy
1. Potassium exists in living systems as the potassium ion, K. Fill in the energy-level diagram below for the K+ ion, being sure to follow the aufbau principle (orbital filling) and Hund's rule (electron pairing rule) for energy-level diagrams (8.4). 4p 3d 4s 3p 3s 2p 2s 1s Energy
Chemistry by OpenStax (2015-05-04)
1st Edition
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Chapter6: Electronic Structure And Periodic Properties Of Elements
Section: Chapter Questions
Problem 58E: Which of the following atoms contains only three valence electrons: Li, B, N, F, Ne?
Related questions
Concept explainers
Atomic Structure
The basic structure of an atom is defined as the component-level of atomic structure of an atom. Precisely speaking an atom consists of three major subatomic particles which are protons, neutrons, and electrons. Many theories have been stated for explaining the structure of an atom.
Shape of the D Orbital
Shapes of orbitals are an approximate representation of boundaries in space for finding electrons occupied in that respective orbital. D orbitals are known to have a clover leaf shape or dumbbell inside where electrons can be found.
Question
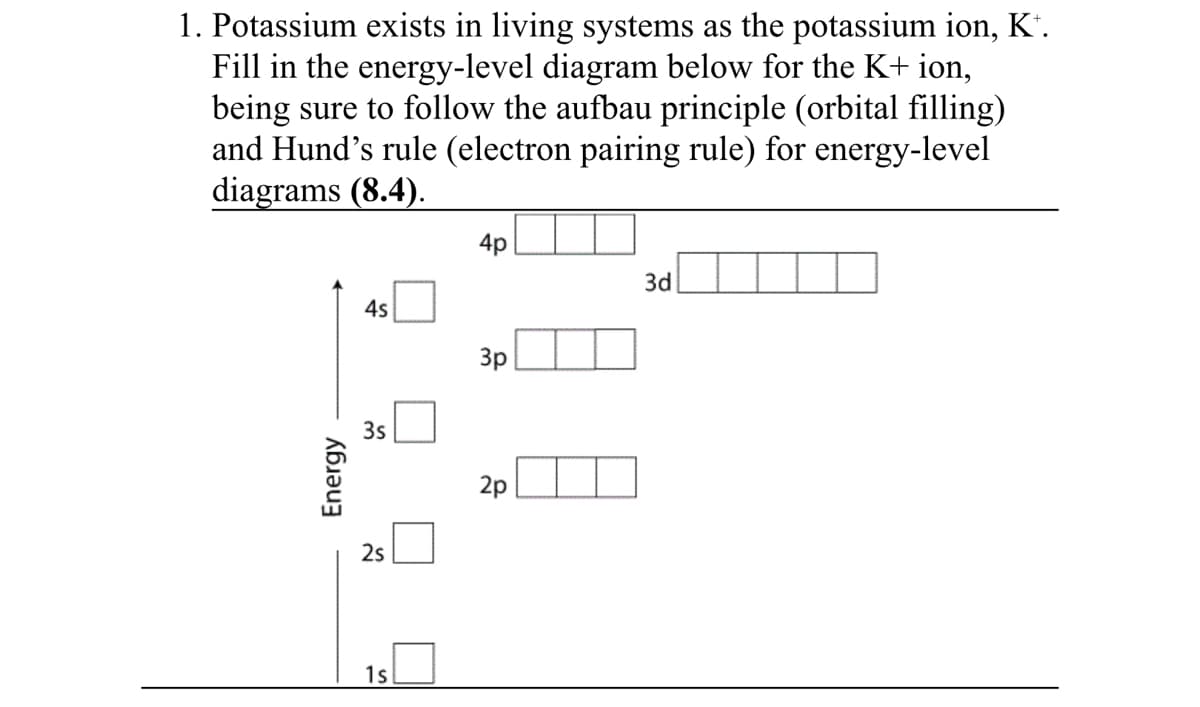
Transcribed Image Text:1. Potassium exists in living systems as the potassium ion, K".
Fill in the energy-level diagram below for the K+ ion,
being sure to follow the aufbau principle (orbital filling)
and Hund's rule (electron pairing rule) for energy-level
diagrams (8.4).
4p
3d
4s
3p
3s
2p
2s
1s
Energy
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax