1. Some cars can use butane (C4H10) as fuel: 2 C4H10 + 13 O2 → 8 CO2 + 10 H20 a. How many grams of CO2 are produced from the combustion of 150 grams of butane? b. How many grams of O2 are needed to react with 150. grams of butane? c. How many grams of H2O are produced 5.73 g of Oz is reacted?
1. Some cars can use butane (C4H10) as fuel: 2 C4H10 + 13 O2 → 8 CO2 + 10 H20 a. How many grams of CO2 are produced from the combustion of 150 grams of butane? b. How many grams of O2 are needed to react with 150. grams of butane? c. How many grams of H2O are produced 5.73 g of Oz is reacted?
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter3: Mass Relations In Chemistry; Stoichiometry
Section: Chapter Questions
Problem 58QAP: Sand is mainly silicon dioxide. When sand is heated with an excess of coke (carbon), pure silicon...
Related questions
Question
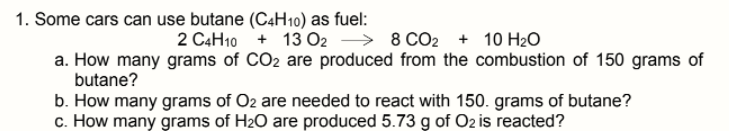
Transcribed Image Text:1. Some cars can use butane (C4H10) as fuel:
2 C4H10 + 13 O2 → 8 CO2 + 10 H20
a. How many grams of CO2 are produced from the combustion of 150 grams of
butane?
b. How many grams of O2 are needed to react with 150. grams of butane?
c. How many grams of H2O are produced 5.73 g of Oz is reacted?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning