Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter20: Environmental Chemistry-earth's Environment, Energy, And Sustainability
Section: Chapter Questions
Problem 49IL
Related questions
Question

Transcribed Image Text:1. What is the length of the rod shown below?
3
4
7
10
2. If Student A measures the volume of a liquid and reports its volume as 23.50 mL
and Student B measures a different amount of the same liquid and reports its
volume as 15.0 mL, is it likely that the students used the same measuring device?
Yes or no? Explain your answer.
3. If you were asked to measure 25.0 mL of a liquid and to transfer it to another
container, which of the following glassware could you use? Choose all that apply.
Give reason(s).
a) A 50-mL buret with smallest divisions of 0.1 mL
b) A 50 mL beaker with smallest divisions of 10 mL
c) A 50-mL volumetric flask
d) A 50-mL graduated cylinder with smallest divisions of 1 mL
e) A 25-mL pipet
4. A group of five students attempted to estimate 100 g of a substance by balancing
the amount of the substance with a 100-g standard mass and obtained the following
masses after each estimated amount was weighed: 95.8634 g, 80.8125 g, 106.5078
98.2865
and 86.4453
Create a data table as shown below and use the data
g,
to determine the answers to the following questions:
Mass (g)
g,
g.
Deviation (g)
% Deviation
Error (g)
% Error
a) What is the mean? And what is the true value?
b) What are the deviation and % deviation from the mean for each value?
c) What are the error and % error for each value?
Expert Solution
Step 1
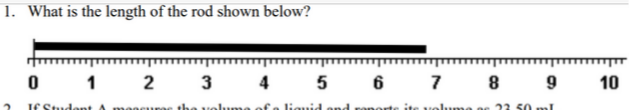
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning
