Q: 1. The solubility of Ag₂SO₄ in water at 25 °C is 0.0155 M. What is Ksp for Ag₂SO
A: Since you have posted multiple question. We will solve only the first question for you. To get the…
Q: The following solutions were prepared: Solution A: 0.01 mole of perchloric acid (HClO4) dissolved…
A: Given- Moles of HClO4 = 0.01 Volume of water = 1L Moles of ethylamine hydrochloride = 0.01 Volume…
Q: 1. Write a balanced chemical reaction for the precipitation of Fe (after the addition of ammonium…
A: 1. 6NH4NO3 + Fe2O3 ----> 3O(NH4)2 + 6NO3…
Q: Distinguish between physical and chemical properties and changes (§1.1) (SPs 1.1, 1.2) (EPs 1.1,…
A: Matter is anything that occupies space and volume. Matter is made up of atoms. Matter has many…
Q: What is the pH of a 0.15 M solution of sodium nitrite? The pKa of nitrous acid is 3.15.
A: Consider the given information is as follows; Concentration of sodium nitrite = 0.15 M pKa of…
Q: What is the concentration of F− ions in a solution prepared by saturating a 0.0045 M Ba(NO3)2…
A: The concentration of Ba2+ is 0.0045 M. The dissociation of BaF2 is given below. An equilibrium…
Q: Phosphoric (H,PO4) is a triprotic acid with pKa values of 2.12, 7.21, and 12.32. It is solution for…
A:
Q: (i) Me3Sn. OEt Pd(PPH3)4/CO A (ii) Br Ph Pd(PPH3)4, KOH -B(OH)2 В Br +
A:
Q: Calculate the pH of an aqueous solution prepared by dissolving 5.61 g of KOH pellets in water and…
A: Mass of KOH = 5.61 g Volume = 0.500 L Molar mass of KOH = 39.10 + 16.00 + 1.008 = 56.108 g/mol pH of…
Q: The pH of a 0.02M solution of a weak acid was measure at 4.6 a. What is the [H+] in this solution?…
A: Given : pH = 4.6 Molar concentration of weak acid = 0.02 M
Q: When 55.5 g of calcium chloride CaC12 was dissolved in 0.5 L of water, so the concentration of the…
A: Given Mass of CaCl2 = 55.5 gram Volume = 0.5 Liter Molarity = ?
Q: How many milligrams of NaOH must be added to 100.0 mL of a 1.25 x 10-9 M CeCl3 solution in order to…
A: Recall the dissociation of CeCl3 CeCl3→Ce3+ + 3Cl-Find concentrations of ions obtained from…
Q: Write the equation for the reaction of Pb2 with CrO42-.
A: The reaction between Pb+2 and CrO4-2 is a precipitation reaction.
Q: What is the role of ammonium oxalate? Make a balanced chemical equation for the reaction of ammonium…
A: Ammonium oxalate is used as an analytical reagent and general reducing agent. It is used as…
Q: How did you know to make Fe 54 x and Fe 57 .0825 -x
A:
Q: Which is more acidic? Is the most dangerous acid the strongest acid? Include a comparison of pH and…
A: Acidic character of a compound can be explained from the values of equilibrium constant. If the…
Q: The first stage in corrosion of iron upon exposure to air is oxidation to Fe2+. (a) Write a balanced…
A: Corrosion is a galvanic process by which metals deteriorate through oxidation, usually but not…
Q: Calculate the PH and the POH FOR a solution that is ISM IN Hellz and .04M IN HALO₂, Heloz K; FOR…
A: HClO3(aq) is a strong acid, whereas HClO2(aq) is a weak acid with Ka = 1.1*10-2 Given [HClO3(aq)] =…
Q: What is the solubility of La(IO₃)₃ in water? (Ksp of La(IO₃)₃ is 7.5 × 10⁻¹²)
A: Solubility :- The maximum amount of a solute that can be dissolved in specified amount of solvent at…
Q: At 25C, 0.00188 g of AgCl dissolves in one liter of water Write a balanced chemical equation What…
A:
Q: Salts of hypoiodite ion behave as a weak base, undergoing hydrolysis in water according to the…
A: Given Reaction:- IO- + H2O <----------> HIO + OH- Kh = 0.00044 Kh = [HIO ] [ OH- ] / […
Q: 24) What is the pH of a 0.570 M solution of aniline? Kb = 7.4 x 10-10 A) 2.6 В) 4.7 C) 11.4 D) 9.3…
A: The given aniline is a weak base and has Kb = 7.4 x10-10 . The equilibrium reaction for the aqueous…
Q: b) A solution of chlorine in water is used as a disinfectant. The equation below indicates the…
A: Rxn of chlorine with water forms hypochlorous acid. This reaction is termed as hydrolysis rxn. Cl2 +…
Q: What is the pH of a saturated solution of magnesium hydroxide, Ca(OH)2 (slaked lime)? Kep for…
A: Ksp for Ca(OH)2 = 5.5 × 10-5 (given) pH of saturation soln of Mg(OH)2 = ? The expression for the…
Q: Why does dry HCl gas not change the colour of the dry litmus paper?
A: Colour of the litmus paper is changed by the hydrogen ions. Dry HCl gas does not contain H+ ions.
Q: alculate the alkalinity of a lake if you know that the concentration of h2cos is 3000 um, hcos…
A:
Q: E°cell = 1.9791 ΔG° = -381.91 What is K?
A: The relationship between the E0cell and equilibrium constant is given by RTln(K) = nFE0cell where…
Q: Explain The Relationship between ?G°rxn and K?
A: ΔG° is defined as the change in standard Gibbs free energy. K is the equilibrium constant and is…
Q: Find the pH of a 0.381 M aqueous solution of hypobromous acid (HOBr), for which Ka = 2.1 * 10-⁹.…
A:
Q: Fe3+ + OH- FEOH²+ + OH- Fe(OH)2* + OH- Fe(OH)3(ferihydrite) + OH- Fe(OH)4
A: pH is the negative logarithm of the hydrogen ion concentration. It is a measure of acidity or…
Q: A0.300 M solution of nitrous acid (HNO2) had a pH of 2.07. What is the Ka value for nitrous acid. 1.…
A: The molarity of a nitrous acid solution is = 0.300 M The pH of the solution is =2.07 The Ka value of…
Q: A 0.200 M solution of nitrous acid (HNO,) had a pH of 2.07. What is the Ka value for nitrous acid 1.…
A:
Q: Write the equation for thermal decomposition of CaCO3 and Mg(NO3)2
A: When CaCO3 is heated at high temperature, it is decomposed to calcium oxide and carbon dioxide as…
Q: 1. Answer the following questions: a) Why aqueous solutions of KMN04 are not entirely stable? b)…
A: KMnO4 is an intense purple color salt. The color of KMnO4 is so intense due to the charge transfer…
Q: 2. What test could you perform to detect the presence of NH4* ion? Write a reaction and explain what…
A: As per our guidelines we can only solve first question. Please resubmit the other questions as next…
Q: Determine the pH of a 10-3 mol/L solution of (a) hydrogen sulfate ion (pKa = 1.99), (b) formic acid…
A:
Q: 18. How many grams of ammonium nitrate must be weighed out to make 415 g of a 58.0% by mass…
A: A solution consists of solute and solvent. A solute is defined as the component that is present in a…
Q: Fluoride treatment strengthens tooth enamel by converting hydroxyapatite, Ca5(PO4)OH, into…
A: Ca5(PO4)3F(s) ----> 5Ca2+ + 3PO43- + F- The solubility product constant,Ksp is the equilibrium…
Q: What is the pH of a solution that is 0.063 M in HA and also 0.023 M in NaA? (Ka = 4.1 x 10-6) %3D O…
A: given, concentration of HA = 0.063M concentration of NaA= 0.023M Ka = 4.1×10-6
Q: Phosphoric acid (H₃PO₄) is a triprotic acid with pKa values of 2.12, 7.21, and 12.32. It is a useful…
A: "Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: What is An for the following equation in relating Kc to Kp? 2 SO2(g) + O2(g) = 2 SO3(g) O3 O-1 O-2…
A: Relation between Kc, Kp and ∆n:
Q: Will acidic food coked in a cast iron skillet become FE2+, enriched because of a reaction between…
A:
Q: 38. Determine the pH of 0.42 M for formic acid, HCOOH solution. The value of pka for HCOOH is 3.75.…
A: We have to calculate the pH of Solution.
Q: List the dominant (strongest) type of IMF for the pure substances, then rank the strength of each…
A: IMF (inter molecular force) are mainly of 4 types i.e. dipole -dipole, hydrogen bonding, dispersion…
Q: What solid would form when phosphoric acid (H3PO4) is added to milk of magnesia (Mg(OH)2)?
A: When an acid reacts with a base it produces salt and water. Such reactions are known as acid-base…
Q: H Diethyl tartarate Titanium isopropoxide, 8.
A:
Q: Silica (SiO2) is an impurity that must be removed from ametal oxide or sulfide ore when the ore is…
A: Balanced chemical reaction is the chemical equation that contains an equal number of atoms of each…
Q: B- Describe the preparation of 5 L of 0.1 M NaHCO3 (Na= 23 , C= 12 , 0= 16 , H=1 ) from the primary…
A: We have given that Volume of solution (V) = 5 L Molarity (M) = 0.1 M Molar mass of NaHCO3 = 84…
Q: Question 15.22
A: Given,Temperature = 303K.Kp = 34.5.
Instructions: Answer the following questions regarding the test for phenol.
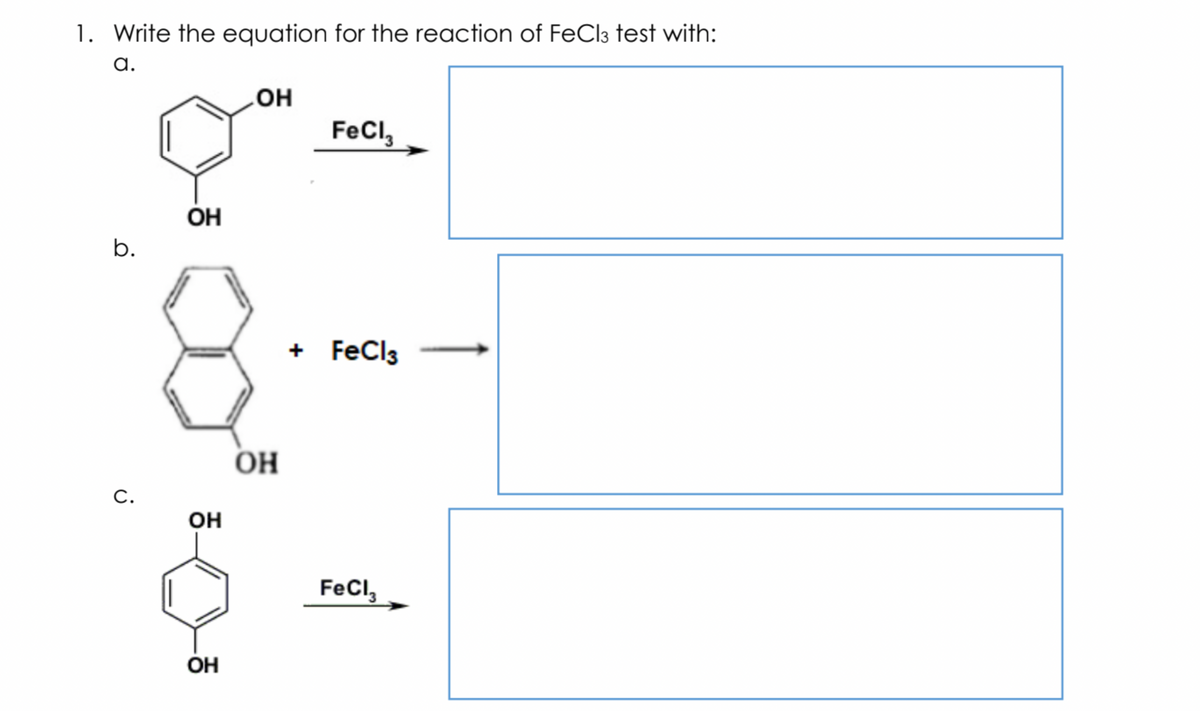
Step by step
Solved in 3 steps with 2 images

- 31. A sample of meat scrap weighing 2.000 g is digested with concentrated H2SO4 and a catalyat. The resulting solution is made alkaline with NaOH and the liberated ammonia distilled into a 50.0 mL of 0.6700 N H2SO4. The excess then requires 30.10 mL of 0.06520 N NaOH for neutralization. What is the percentage of nitrogen in the meat?Sodium lauryl sulfate has the same use in embalming fluids as: 1. sodium phosphate 2. citrates 3. sulfonates 4. sodium salt of EDTA 1 & 4 only 3 only 4 only 1,2,3,and 424. (a) How many picomoles of Ca are in 0.0100 g of wollastonite (Ca2Si2O6)? (b) How many picograms of Ca are in 0.0100 mol of wollastonite?
- A 0.9056 g sample of KBrO3 (MM=167.0) was dissolved in dilute HCl and treated with an unmeasured excess of KI. The liberated iodine required 37.21 mL of Na2S2O3. Calculate the molar concentration of sodium thiosulfate. 1 mol BrO3- = 3 mol I2 = 6 mol S2O32- 0.8744 M 2.623 M 0.8791 M 0.2915 MWhy alkali is added to the nitrification tank? A. to provide electron donors B. to provide alkalinity for the nitrification reaction C. to acidify the reaction solution D. to provide nutrients for the nitrifier bacteriaA 0.1017 g sample of KBrO3 (MM=166.1) was dissolved in dilute HCI and treated with an unmeasured excess of Kl. The liberated iodine required 39.75 mL of Na₂S₂O3. Calculate the molar concentration of sodium thiosulfate. 0.2561 M 0.08512 M 0.09242 M 0.1041 M
- Use your calculated values of Kspand Kf to explain why CdC2O4 is dissolved by ammonia. Ksp = 7.12 x 10-8 Kf = 45.5250.00 mL of a water sample is diluted with 150.00mL deionized water. The diluted sample has a lead content of 17.8 ppb. What us the led content (in ppb) of the original sample?Calmagite is often used as a substitute for Eriochrome Black T as indicatorin the determination of Ca. List down two main advantages of calmagite overEriochrome Black T.
- A 0.951-g sample containing (NH4)2C2O4 and compounds was dissolved in water and made alkaline with KOH. The liberated ammonia was distilled into 50.00 mL of 0.1007 N H2SO4. The excess H2SO4 was back titrated with 11.13 mL of 0.1214 N NaOH. Calculate the percentage of nitrogen in the sample.What is the reducing and oxidizing agent in the formation of potassium trioxalatoferrate(III) trihydrate? What is the reducing and oxidizing agents when this is titrated with KMnO4?How do you find the simpliest oxidation step by step for Fe3+?

