Q: Indicate the stereochemical outcomes of the following SN1 reactions. Assume that all chiral centers…
A: We know that SN1 reaction is a two step reaction, in which 1st step includes removal of leaving…
Q: Compound A has a molecular formula C6H10 when A reacts with (BH3) in the presence of H2C2 it gives…
A: The question is based on the concept of organic reactions. We need to identify A,B &C based on…
Q: Determine the number of atoms in 27.0 grams of bromine, Br. (The mass of one mole of bromine is…
A: This problem is based on mole concept.
Q: Use the solubility rules to predict whether the following compounds are soluble AgCl PbSO4 CaCl2…
A: Solubility rules are a set of guidelines that help predict whether a compound is likely to be…
Q: A reaction mixture (mixture 1) at 298 K has a N2O4(g) partial pressure of 0.1 bar and a NO2(g)…
A:
Q: What is the concentration of a solution formed by diluting 25.0 mL of a 3.2 M NaCl solution to 135.0…
A:
Q: A mixture of argon and neon has a measured gas density of 1.40 g/L at STP. Assuming ideal-gas…
A: Density of mixture = 1.40 g/L
Q: The dipole moment (μ�) of HBrHBr (a polar covalent molecule) is 0.831D0.831D (debye), and its…
A: This question is based upon chemical Bonding.
Q: Methane and hydrogen sulfide form when hydrogen reacts with carbon disulfide. C5, + 4H2 -> CHa + 2…
A: Methane (CH4) and hydrogen sulfide (H2S) form when hydrogen (H2) reacts with carbon disulfide (CS2).…
Q: When 18.0 mL of a 6.86 x 10^-4 M lead acetate solution is combined with 12.0 mL of a 2.28 X10^-4 M…
A:
Q: Answer the questions about the balanced reaction The molar masses are as follows: A = 10. g How many…
A: Answer: This question is based on stoichiometric calculation where we have to convert the moles of C…
Q: What change, if any, would you expect if the aromatic substituents were in the meta position, rather…
A: Benzyl alcohol with para-substituted aromatic groups(para chloro,para methoxy,para methyl), the…
Q: Draw both the SN1 and E1 products of each reaction. CH3 a. -Br + H₂O CH3 b. CH3-C-CH₂CH₂CH3 CI +…
A:
Q: 10 9 3 (ppm) - 0 8 2 7 6 5 ð (ppm) 1.00 4 0.67 3 2 0.67 1 1.00 0
A: The question is based on the concept of organic spectroscopy. We need to identify the compound.
Q: ← Draw the major product of this reaction. Ignore inorganic byproducts. x H₂O* Drawing O Problem 310…
A:
Q: Balance the following oxidation/reduction equation carried out in acid. H_2S (aq) + HCrO_4 =…
A: Balance the following oxidation/reduction equation carried out in acid. H_2S (aq) + HCrO_4 =…
Q: A mixture of liquids A and B exhibits ideal behavior. At 84 °C, the total vapor pressure of a…
A: This question is based on Raoults law.
Q: A 0.10 M weak acid is titrated with a 0.10 M strong base. Plot the titration curves on the same…
A:
Q: If 5.0 mL of 0.20 M NaOH is added to 35.0 mL of 0.20 M HCl, what will be the pH of the resulting…
A: Here, Molarity of NaOH = 0.20 M Volume of NaOH = 5.0 mL Molarity of HCl = 0.20 M Volume of HCl =…
Q: a) b) c) HNO3 H₂SO4 O CI AICI 3 Na₂Cr₂O7 H* Cl2, AICI 3 Zn-Hg HCI, heat
A: Given reactions are shown below We have to draw the major organic product of the above reactions.
Q: Question 19 options: The planet Vulcan is known for its thin atmosphere. The pressure on the…
A: Check the correct law which includes the pressure of all gases in it and use it to calculate the…
Q: If a system absorbs 155.0 kJ of heat from its surroundings and the system does 45.0 kJ of work on…
A:
Q: Calculate the pOH of the post-equivalence solution for the titration of 25.00 mL of a 0.025 M H2SO4…
A: The balanced chemical equation for the reaction between sulfuric acid (H2SO4) and sodium hydroxide…
Q: How would you prepare 150 mL of 0.85% saline solution in water from a 15% saline solution? A. 8.5 mL…
A: We have given saline solution concentration is 15% . We need to prepare 150ml of 0.85% saline…
Q: Choose the best reagent to accomplish the following reaction. О н30+ O NaOH 0 H20 O HI НI. OH ОН
A: Organic reactions are those in which organic reactant react to form organic products. In the given…
Q: PLEASE HELP ME ANSWER QUESTION 5) A student starts a titration without phenolphthalein in the…
A: This question belongs to titration.
Q: Question 18 of 20 If a system does 84.0 kJ of work on its surroundings and releases 105 kJ of heat,…
A:
Q: From the abbreviated name of the compound Gal (β1->4) Glc, we know that— C4 of galactose is…
A: From the abbreviated name of the compound Gal (β1->4) Glc, we know that— C4 of galactose…
Q: draw IR for the following methyl butanoate benzaldehyde 1-chlorobutane
A: Since you have asked multiple sub-questions, we will solve the first three subparts for you. If you…
Q: From the given situations, explain whether its solution requires a qualitative analysis or…
A: Qualitative analysis deals with the determination if some component is present in a sample whereas…
Q: The circus clown is a chemistry geek and likes to fill his balloons to exactly 30 moles of helium to…
A:
Q: 2. NaOH is commonly used to produce paper, dyes and soap. Calculate the mole fraction of NaOH in a…
A: Since each question is different, we request you to upload two questions differently as we solve…
Q: What is the molarity of a 25.0% (v/v) aqueous isopropyl alcohol solution? The density of isopropyl…
A:
Q: X₂ x H H H₂O H₂O x :CI:O H HH Select to Add Arrows H 0: H₂O Select to Add Arrows
A: We have to draw the curved arrows for the every steps.
Q: At 2.86 atm and 494 K, what volume of NO₂ gas forms from 59.66 g of O₂? 4 NH3(g) +70₂(g) → - 4NO₂(g)…
A:
Q: What alkenes are formed from each alkyl halide by an E2 reaction? Use the Zaitsev rule to predict…
A: In E2 elimination, the attack of base and elimination of leaving group takes place in a single step.…
Q: Which of the following could enter into H-bonding interactions with water? (Select all that apply.)…
A: We know that, hydrogen bonding or H-bond is an interaction formed between the hydrogen atom and a…
Q: 6.44 L cylinder contains 2.27 mol of gas A and 3.70 mol of gas B, at a temperature of 29.2 °C.…
A:
Q: Calculate the solubility of CuCO3 in water at 25 °C. You'll find Kp data in the ALEKS Data tab. sp…
A:
Q: Answer the questions about the balanced reaction 3A + 2B The molar masses are as follows: A = 10. g…
A: Answer: This question is based on stoichiometric calculation where we have to convert the moles of C…
Q: 2-phenyl-propanoic acid 1) 2) PBr3 Br₂, HCl (aq.)
A: The given reaction scheme is shown below We have to draw the mechanism of the above reaction and we…
Q: Draw an E1 mechanism for the following reaction. Draw the structure of the transition state for each…
A: The given reaction scheme is shown below
Q: Provide the correct IUPAC name for the skeletal (line-bond) structure shown here.
A: The question is based on the concept of IUPAC naming. we need to write IUPAC name of given compound…
Q: A group of students is doing an analysis of copper in a 1-peso coin. They were tasked to prepare…
A: Since you have asked a question with multiple sub-parts, as per our company guidelines we are…
Q: = the relationship and the is typically written RT 8.314 J/mol. K), quency factor, and or the…
A:
Q: 6.1 Calculate the enthalpy of the reaction 2NO (g) + O2(g) -2NO2 (g), given the following reactions…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Provide the IUPAC name for the compound shown here. SH SH OH
A: Since,Rule of IUPAC-1) Longest chain as parent chain.2) Numbering start from those side where more…
Q: НО
A:
Q: Write the basic equilibrium equation for HS. Be sure to include the proper phases for all species…
A: Bronsted and Lowry concept of Acids & bases is used to write basic equilibrium equation as…
Q: What is the maximum number of electrons in the entire shell?
A: The number of electrons in a shell = 2×n2 Here, n = the Principal quantum number
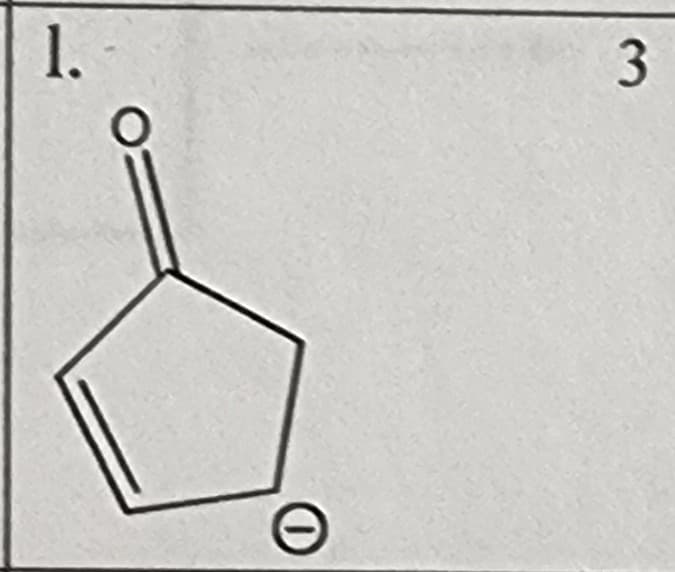
What would the resonance for this structure be?
Step by step
Solved in 4 steps with 1 images


