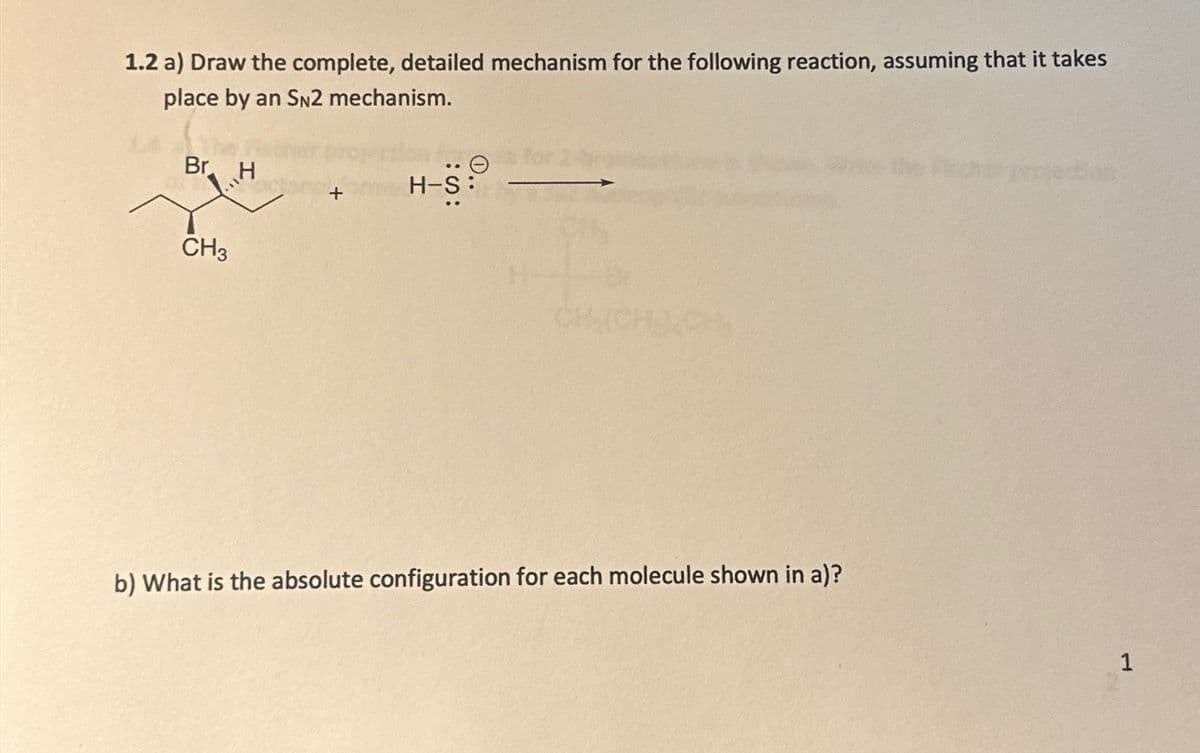
1.2 a) Draw the complete, detailed mechanism for the following reaction, assuming that it takes place by an SN2 mechanism. Br H Θ H-S :S: CH3 b) What is the absolute configuration for each molecule shown in a)? 1
Q: How many moles of potassium fluoride should be dissolved in 1.98 L of solvent to produce a 10.2 M…
A: Given: V=1.98L;[KF]=10.2MStep 1: What is the number significant figures for the final answer.The…
Q: Identify the most acidic solution. options: 0.001 M solution of HNO3 10 M solution of CH3COOH 0.001…
A: Solution: To identify the most acidic solution, we can compare the strength of the acids. The…
Q: What is the mass in grams of 5.2 X1024 formula units of sodium nitrate?
A:
Q: Don't use hand raiting please
A:
Q: An aqueous potassium carbonate solution is made by dissolving 5.84 moles of K2CO3 in sufficient…
A: Step 1: Given: An aqueous potassium carbonate solution s made by dissolving 5.84 moles of K2CO3 in…
Q: Please correct answer and don't use hend raiting
A: Step 1: Determine if structures ( A ) and ( B ) have the same molecular formula. Yes, structures ( A…
Q: Select the TRUE statement regarding the diffusion of gas particles. Gas particles diffuse from areas…
A: The objective of the question is to identify the correct statement about the diffusion of gas…
Q: Please don't use hend raiting step by step solutions
A: First, the base will abstract alpha H and alkylation will occur twice to form the 6-membered cyclic…
Q: please draw out a complete synthesis for the following molecule starting with benzene:…
A: Step 1: Step 2: Step 3: Step 4:
Q: Calculate the missing molarity in the line notation below that would cause this electrochemical cell…
A: Approach to solving the question: First of all see, which side molarity is missing. It is clearly…
Q: None
A:
Q: The Kp for the reaction A(g) ⇌ 2B(g) = 0.0450. So, what is Kp for the reaction 2 A (g) ⇌ 4 B (g)
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: None
A: Step 1: When 1-chloropropane reacts with Li and Cu, it forms Gilman reagent as shown below. This is…
Q: 2Fe2O3(s) 4Fe(s) + 302(g). Use the following data: So Fe(s) = 27.31 J/K, S° O2(g) = 205.15 J/K, S°…
A: The objective of the question is to calculate the change in entropy (ΔS°) for the given chemical…
Q: Consider the reaction. :0: 1. NaBH4 2. D₂O H Complete the electron-pushing mechanism for the…
A: Step 1: Reaction mechanism: Step 2: Step1 of mechanism:
Q: Sally obtains a standard calibration curve for their assigned food dye by plotting absorbance versus…
A: The objective of this question is to determine the concentration of the food dye in the original…
Q: A student uses 10.543 g of an NaHCO, (84.0 g/mol) solution to perform a gas stoichiometry…
A: Mass of NaHCO3 = 10.543 gMolar mass of NaHCO3 = 84.0 g/molVolume of displaced saturated NaCl = 103.2…
Q: None
A: While directly synthesizing 2-methylbutanoic acid (b) from benzene is a complex process with several…
Q: None
A:
Q: 1. Calculate the mass percent, % (m/m), for the solute in the following: 24.0 g of KCI and 200. g of…
A: 1.) Given:mass of solute (KCl) = 24.0 gmass of solvent (H2O) = 200 gMass of solution = 24.0 + 200 =…
Q: The following graph shows the pH curve for the titration of 25 mL of a 0.1 M monoprotic acid…
A: Step :
Q: help 5
A: Step 1 : Reaction mechanism : Step 2: The answer is:
Q: A researcher wants to determine the concentration of beta-carotene in carrots. She obtains a 0.661 M…
A: The objective of this question is to determine the concentration of beta-carotene in Dilution C,…
Q: When a 22.7 mL sample of a 0.472 M aqueous hydrofluoric acid solution is titrated with a 0.493 M…
A: The objective of this question is to calculate the pH at the midpoint of a titration between…
Q: 3. The heat of reaction for Equation 1 is equal to the heat of formation of solid magnesium oxide.…
A: The objective of the given question is to determine the percent error in the experimental…
Q: None
A:
Q: Starting Material 1-methyl-cyclopent-1 - ene Target 3 methyl cyclopent-1 - ene Synetheis reaction…
A: The objective of the question is to provide a synthesis reaction that converts…
Q: If 2.20 kg of solution contains 0.59 kg of solute, what is the percent by mass concentration? 0.27%…
A: The objective of this question is to calculate the percent by mass concentration of a solution. The…
Q: What is the concentration of a dextrose solution prepared by diluting 11 mL of a 4.0 M dextrose…
A: The objective of this question is to find the concentration of a dextrose solution after it has been…
Q: 4. Consider the reaction "He(p. d) (a) Complete the reactions, calculate their Q values using mass…
A: The objective of the question is to complete the given reaction Calculate the Q value and predict…
Q: The enthalpy of vaporization of Substance x is 7.00 kJ/mol and its normal boiling point is -10c .…
A:
Q: Balanced equation of ethambutol hydrochloride and acetous perchloric acid
A: Step 1: Here's how to work out the balanced equation for the reaction between ethambutol…
Q: The synthesis of ethanol from ethylene and water proceeds according to the reaction C2H4(g) + H2O(1)…
A: The objective of the question is to calculate the Gibbs free energy of the reaction under…
Q: 106.54 mg of an impure sample of the drug was diluted in 100 ml and injected in a HPLC, the AUC of…
A: The objective of this question is to determine the weight by weight percentage of purity for a drug…
Q: Draw the mechanism of the transformation from 3 to 4. F EtO HO 3 OEt NaOH, MeOH HO OH
A: The objective of the question is to understand the mechanism of the transformation from compound 3…
Q: Experimental Volumes
A: MixtureVolume (mL) Fe(NO3)3Volume (mL) KSCNVolume (mL)…
Q: Consider the OH radical having a vibrational constant of 3735.2cm¹ and a rotational constant of…
A: ### 1. Energy Levels of the OH Radical- **Vibrational Levels**: These are determined by the…
Q: Show reaekson and don't use hend raiting and step by step solutions please
A: Step 1:Step 2:The reaction simply follows the SN2 mechanism which is a most favoured solvent that is…
Q: The solubility of AgCl in water at 25 °C is measured to be 0.0019 Round your answer to 2 significant…
A: Given solubility of AgCl is 0.0019 g/LMolar mass of AgCl is 143.5 g/molSolubility in mol/L can be…
Q: None
A:
Q: Show reaekson and don't use hend raiting and step by step solutions
A: I can't access the specific image you sent, but I can still explain why alkenes undergo alkene…
Q: None
A: The synthesis steps would be:1. **Preparation of the β-Keto Esters:** React ethyl acetoacetate with…
Q: please answer provide
A: The objective of this question is to calculate the solubility of magnesium hydroxide (Mg(OH)2) in a…
Q: 15 g of polyethylene glycol is added to 5.0 g of glucose in a bag and then water is added to…
A: Mass of polyethylene glycol = 15 gMass of glucose = 5.0 gInitial volume - 50 mLGlucose Polyethylene…
Q: Instructions:1. Read each question carefully.2. Answer each question, showing all your work for any…
A: Approach to solving the question:ANSWER # 1 : Detailed explanation: ANSWER # 2: Surely, Metal A…
Q: Identify the steps (reagents) necessary for the transformations below.
A: Step 1: Explanation: Esters can be converted into various different compounds using various…
Q: I need answer expert solutions I need answer step by step
A: Please Rate if the solution helps you.Thank You
Q: Fill in the boxes with the appropriate structures. I know this is hard but this is great practice…
A: Detailed explanation: a.) Ethylidenecyclohexane undergoes hydroboration in the presence of borane…
Q: Between a 2.0 M aq. NaNO3 and 1.5 M aq. AlCl3 solution: Which would have a lower freezing point? ○…
A: The objective of the question is to determine which solution, 2.0 M NaNO3 or 1.5 M AlCl3, would have…
Q: Payalben
A: The objective of the question is to estimate the concentration of E. coli bacteria at the end of the…
2 a) Draw the complete, detailed mechanism for the following reaction, assuming that it takes place by an SN2 mechanism. b) What is the absolute configuration for each molecule shown in a)?
Step by step
Solved in 2 steps with 3 images

- a. Which of the following would be more reactive in an electrophilic substitution and why? Use 1image b. Arrange the compounds in each set-in order of decreasing reactivity (fastest and slowest) toward electrophilic aromatic substitution and explain. Use 2 imagea. What are the products of following reaction?b. Write the reaction mechanism for each using the right arrows and define it as Sn2, Sn1, E2 or E1.c. Explain why do you choose that product and mechanism or in case something else happens explainwhy.Provide the complete mechanism using Curved Arrow Formalism for the reaction of 1-chloro-1-methylcyclopentane treated with NaOMe in MeOH. If more than one product is formed, which is the major, minor, and very minor product? Is the reaction SN1, SN2, E1, or E2. I appreciate the help on this question.
- Draw a detailed step-wise mechanism for the following reactions. Be sure to show all steps, formal charges, and show the movement of electrons with curved arrows.ochem help please Predict the major product(s) for the following reaction sequences and provide the stepwise mechanism for both steps 1 and 2 .... (see attached image)Mechanism 1 Draw a plausible arrow pushing mechanism for the reaction below. You must use the exact structures shown (for example, don’t simplify or substitute the structure with an R)
- Using Curved Arrow Formalism, draw the reaction of 1-methylcyclopentanol treated with aqueous sulfuric acid and identify the rate determining step. If more than one product is formed, identify which is major, minor, very minor, etc. Thank you for the help with this question. I appreciate it.An SN2 mechanism's top requirement from an aprotic solvent is to ensure that it can stabilize the leaving group.Draw a complete mechanism, using arrow convention to indicate electron movement, for the following transformation:
- Could you show the mechanism used to transform example c into the most stable carbonation by using a methyl shiftPlease help with the following: Draw a detailed step-wise mechanism for the following reaction. Be sure to show all steps, intermediates, formal charges, and show the movement of electrons with curved arrows.a. What are the products of following reactions?b. Write the reaction mechanism for each using the right arrows and define it as Sn2, Sn1, E2 or E1.c. Explain why do you choose that product and mechanism or in case something else happens explain why.

