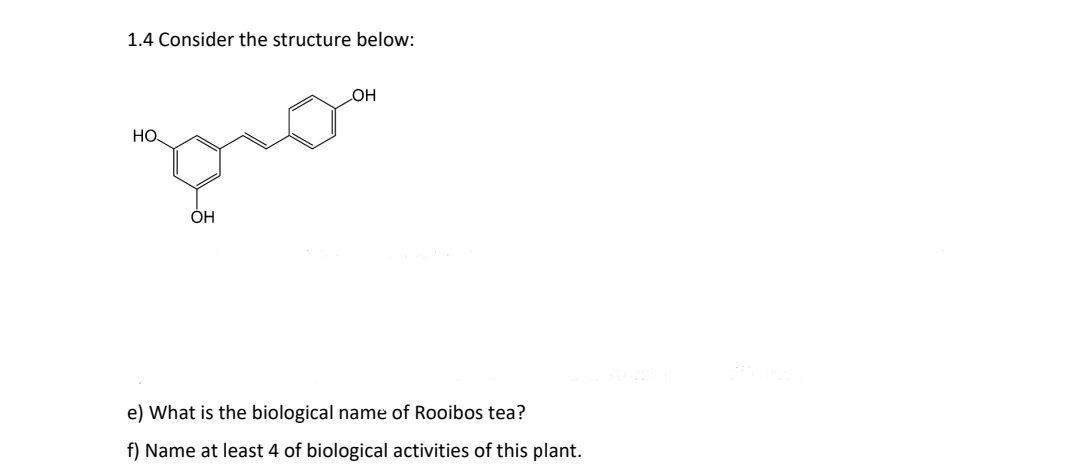
1.4 Consider the structure below: OH НО. po OH e) What is the biological name of Rooibos tea? f) Name at least 4 of biological activities of this plant.
Q: A formula that shows the arrangement of all bonds in a molecule is called a(n) A) molecular formula.…
A: A chemical formula indicates the atoms in a molecule. It shoes the arrangement, number and type of…
Q: A. Н.С. Br B. CH3 с. ЊC. OH OH CH
A:
Q: For this method to work the solid must (a) be more dense than the liquid, and (b) not react with nor…
A: we know density = mass volume for metal piece, if we know the mass and volume of piece of metal…
Q: What is the connection between electrons and protons
A: In a neutral atom, the number of electrons is equal to the number of protons. Both of them are found…
Q: COO + 1 H₂N-C-H 1 €ƒ_ƒ_ƒ_⁄__£ CH₂ 1 CH₂ 1 CH₂ 1 NH C=NH2 NH₂ Arginine, Arg, R
A:
Q: Question 2. Draw the line angle formula for the following alkenes. 4-methylcylcohexene • 2-hexene…
A:
Q: How do I calculate the volume I recorded with a 10 mL pipet?
A: To calculate the volume of a pipet ,first we have to know the rules of pipetting to minimize the…
Q: The vapor pressure of liquid cobalt is 400 mm Hg at 3.03x10³ K. Assuming that its molar heat of…
A: Given, P1 = 400 mm T1 = 3030 K T2 = 3050 K □H = 450 kJ/mol Using Von't Hoff reaction ln(P2/P1) =…
Q: ZSM-5, a catalyst material, has a surface area of 425 m³/g. What is the surface area of this…
A:
Q: E STATES OF MATTER Using heat of fusion or vaporization to find the heat needed to... OD 0/5…
A: 100 g of ice H2O is heated and bring it to a temperature of 50.5 °C .We have to calculate the…
Q: 2. What is the structure of the compound which gives rise to this data? Explain your reasoning. NMR…
A: Spectroscopic identification of organic compounds.
Q: Shown below is the equilibrium between methylammonia and the methylammonium ion. H H3C-N-H H…
A: Given : pKa = 10.62 Ionization of methyl ammonia = 1/5 So, ionization of methyl ammonium = 4/5
Q: #6 pH and pOH calculations 1. Determine the pH, pOH, [H], and [OH-] for each of the following…
A: According to the bartleby guidelines I can solve only first three subparts of the given questions.…
Q: Draw an “Energy level diagram” for a nitrogen atom. Can you help me? I don't understand how to solve…
A: An energy level diagram for nitrogen atom means we have to draw the atomic orbitals of nitrogen atom…
Q: A sample of nitrogen gas occupies a volume of 7.76 L at 63.0°C and 379 torr. If the volume of the…
A:
Q: 0179 7. The volume of blood plasma in adults is 5.5 L The density of blood plasma is 1.03 body?…
A:
Q: How many grams of oxygen (O) are present in a 6.41 g sample of potassium nitrate (KNO3)? Enter your…
A: Potassium nitrate (KNO3) is ionic compound and it contains one potassium, one nitrogen and three…
Q: Which of these two gases has the larger number of molecules? Flask A Flask B
A: Given- Flask A contains H2 gas and flask B contains CO2 gas. Volume of flask A = volume of…
Q: Use the molecular orbital theory to explain the fundamentals of acid-base interactions (frontier…
A: Use the molecular orbital theory to explain the fundamentals of acid-base interactions (frontier…
Q: Give a clear handwritten answer with explanation needed..given below some options in which compound…
A:
Q: use data to discuss whichc calculated density is more accurate.
A: Density is obtained by dividing mass by volume Density(d) = mass(m)Volume(v) => m = d*v (or) v =…
Q: 4. N-Phenylsydnone, so-named because it was first studied at the University of Sydney, Australia,…
A:
Q: Substance X is known to exist at 1 atm in the solid, liquid, or vapor phase, depending on the…
A:
Q: apor pressure data are given here for acetic acid, CH3 emperature (°C) Vapor Pressure (mm Hg) 25…
A:
Q: When 3.59 g of a nonelectrolyte solute is dissolved in water to make 605 mL of solution at 29 °C,…
A:
Q: Liquid nitrogen has a density of 0.808 g/ml, and boils at 77 K. Researchers often purchase liquid…
A: To calculate the ratio, we would first calculate the volume of nitrogen gas in L after vaporization.…
Q: Which of the following compounds would have the highest boiling point? OCH3CH2OH СН3СН3 СН3ОН…
A:
Q: Matching type: What type of organic compound? 6) CH3CH₂CH=CH₂ 7) O || CH3-C- CH3 8) CH3OH 9) O…
A:
Q: If 28.8 g of O₂ is required to inflate a balloon to a certain size at 32.0 °C, what mass of O₂ is…
A: Given, Mass of O2, w1 = 28.8 g Initial temperature, T1 = 32 °C Final temperature, T2 = 9 °C
Q: An atomic absorption method for the determination of the amount of iron present in used jet engine…
A: Solutions- a standard deviation s = 3.1 µg Fe/mL. calculate the 95 and 99% confidence intervals…
Q: antimal hen 3.18 e freezin alculate molar ma
A:
Q: What distance in miles can you drive in your new Prius hybrid, if your car gets 48.0 miles per…
A: We would use the given car average and volume of gas to calculate the distance.
Q: can u please re do this, i require all 8 steps The following fictitious unbalanced REDOX reaction…
A: Redox is a type of chemical reaction in which the oxidation state of substance change... Oxidation…
Q: сов - с OH CN он OH OH CN
A:
Q: What is the correct assignment of the names of the following heterocycles?
A: Heterocyclic Compounds are ring compounds having atoms of two or more elements in a ring beside…
Q: TRANSPETTANCE 1962 2945 3000 capillary file between salt plates 2500 AVENUMBERS 2000 1371 1192 1004…
A:
Q: A The substance is likely lead. B C D LU E Substance Density (g/cm3) Substance Density (g/cm3) 1.00…
A:
Q: 2. A piece of Cu metal is heated in a test until the temperature reaches 96 °C and dropped into a…
A: 2. A piece of hot Cu metal at 96 °C is dropped into a calorimeter, containing 50 mL of water at 27…
Q: Which of the following compounds is most acidic? Vor for OH OH 11 Al O..N EV Br Ill OH G IV OH ELOH…
A: Acidity of carboxylic increases in the presence of electron withdrawing group and decreases in the…
Q: B. Name the following organic compounds: 16. 17. 18.
A: According to IUPAC nomenclature is an organic compound is named by identifying molecule's longest…
Q: Classify/ Name the cycloalkanes below:
A: The naming of organic compounds can be done with the help of rules of international union of pure…
Q: A 1.47 g sample of CO2 is contained in a 708 mL flask at 24.9 °C. What is the pressure of the gas?…
A: Given -> Weight of CO2 = 1.47 gm Volume (V) = 708 ml = 0.708 L (1L = 1000ml) T = 24.9°C = 24.9 +…
Q: Which compound will be more likely to exist as a hydrated solid? O KCl O CuSO4
A: Some compound when comes in contact with water vapors present in the atmosphere absorbs them into…
Q: 3. Give the name of this compound, including stereochemistry if relevant: H CI HCECCH₂-C-CH₂CH₂Br
A: The IUPAC name of the compound can be written on the basis of the number of carbon atoms in the main…
Q: The Ksp of zinc carbonate, ZnCO3, is 1.46 x 10-10. Calculate the solubility of this compound in g/L.…
A:
Q: Density is an intensive physical property. Look-up the meaning of an intensive property of matter…
A: Intensive properties are those properties whose value do not change with the amount of substance.…
Q: For a particular reaction at 187.3 °C, AG = -989.95 kJ, and AS = 662.97 J/K. Calculate AG for this…
A:
Q: 2) How many non-hydrogen atoms have sp² hybridization in the following molecule: NH₂ A) 0 atoms B) 1…
A: We have to give the number of sp2 hybridized non-hydrogen atom. Concept: Those non-hydrogen atoms…
Q: 2. Calcium Flourite, CaF2, has a structure based on the fec lattice and a basis with Ca at 000 and…
A: Given CaF2 has FCC structure. Ca(000),F(1/4 1/4 1/4),F(3/4 3/4 3/4)
Q: Give a clear handwritten answer with explanation...given below some compounds and options....give…
A: Introduction Any material that has the ability to turn blue litmus paper red has an acidity…
Step by step
Solved in 2 steps with 2 images

- the context is "Cosmone is a molecule used by fragrance manufacturers to provide a rich and elegant musky essence to many perfumes. Cosmone has the molecular formula C15H26O." I need help on part (h).the context is "Cosmone is a molecule used by fragrance manufacturers to provide a rich and elegant musky essence to many perfumes. Cosmone has the molecular formula C15H26O." I need help on part (e) and (f).2 help include sig figs
- 2. What is (A) and (B)?Case #3: Jeepers! Someone broke into the Funnel Cake Factory and stole all the batter. Without it, they won't be able to feed all those hungry fair goers! A liquid found at the crime scene was found to be 13.14% H, 34.75% O, and the remaining portion was C. Who committed the crime? Whodunnit? Suspect 1: George Norman Suspect 2: Becky Baker George Norman recently had an accident where he cut himself with a knife. He cleaned the wound with rubbing alcohol. The formula for rubbing alcohol (isopropanol) is C3H8O. Becky Baker is a world famous pastry chef. She had just finished baking a cake at the time of the crime. The recipe called for vanilla. The formula for vanilla is C8H8O3. Suspect 3: Liz Lemon Suspect 4: Ben Linus Liz Lemon just had her nails done and she still has traces of nail polish remover on her hands. Nail polish remover contains acetone. The formula for acetone is C3H6O. Ben Linus had been walking around in the rain for…USE THE CONDENSED STRUCTURAL FORMULA IN WRITING THE ORGANIC COMPOUND AND GIVE THE NECESSARY COEFFICIENT BESIDE EACH COMPOUND IF NEEDED ANSWER ONLY 7 AND 8
- The compound in Figure 1 can be obtained from the oyster mushroom. It is used to slow down the production of cholesterol in the body. Which of the following functional groups is not present in this connectionHello, I was wondering if anyone had some suggestions for the following conversions? Feel free to take as many steps as needed for each question mark. Thank you in advance!Can you please help me with parts a,b, and c?
- The following essential oil is one of the most used fragrances and fixatives in perfumes, colognes, cosmetics, and soaps, and is often used as a substitute for expensive jasmine essential oil due to its similar properties and perfume: Choose one answer. a. petitgrain b. ylang ylang c. vetiver d. patchouliNo. Question 1 What is the condensed structural formula heptane? 2 Is it solid, liquid or gas at room temperature? 3 Is it soluble in water? 4 Will it float or sink in water? Answer the following questions correctly using the statement below: Heptane, C7H16, has a density of 0.68 g/ml and boils at 98°C.Box 1-3 box 3 answer choices: benzene or cyclohexane



