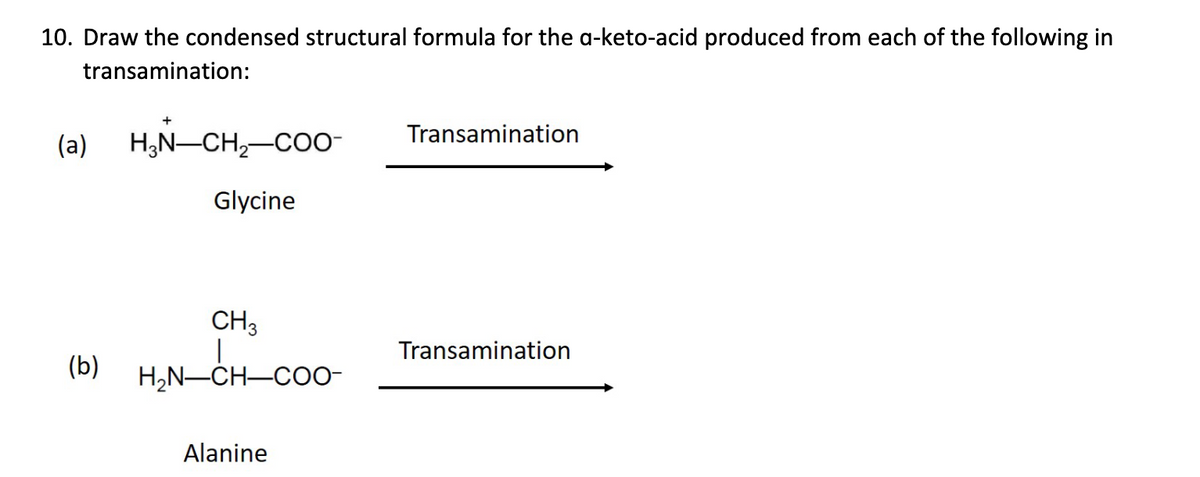
10. Draw the condensed structural formula for the a-keto-acid produced from each of the following in transamination: (a) H,N-CH,-Co- Transamination Glycine CH3 Transamination (b) H,N-CH-CO0- Alanine
Q: Assuming the molar extinction coefficient for para-nitrophenyl phosphate (PNP) is 55,290 mol "dm…
A: Given Values: ε=55290 c = ?A = 0.77 l = 1.5 cm
Q: HO H. H. H. Но ÓH OH H H. H Но OH 1,1 О 1,2 1,3 1,4 1,5 1,6
A: Photosynthesis produces carbohydrates in plants. Animals rely on natural carbohydrate supplies such…
Q: An endorgonic reaction has a ____ G value, which means that it ____ Choices: a. Positive;…
A: In chemical thermodynamics an endergonic reaction is a nonspontaneous and unfavorable reaction.
Q: Absorbance spectrum of Ninhydrin curve 1,6 1,4 1,2 0,8 0,6 0,4 0,2 100 200 300 400 500 600 700 800…
A: Proteins are composed of amino acids, which are bound together by peptide linkage. Amino acids…
Q: Enzyme involved in the second step of ketogenesis
A: Ketogenesis is the formation of ketone bodies (β-Hydroxybutyrate and acetone) from acetyl-CoA.
Q: Compare and contrast the following items related to lipid metabolism. Cite their main…
A: 1)Dehydrogenase enzyme vs. dehydratase enzyme.- Dehydrogenase Enzyme- 1)Pyruvate dehydrogenase is a…
Q: If Dr. Aikenhed wanted to see if there was mutation within the protein-coding sequence of the gene…
A: The technique which can be used to compare protein-coding sequence i.e the nucleotide sequence of…
Q: Why is potential Hydrogen (pH) important to Biology?
A: The pH of aqueous or other liquid solutions indicates how acidic or basic they are. The phrase,…
Q: Polypeptide synthesis proceeds in either direction once initiated. proceeds from the…
A: Peptides are short chains of amino acids that are linked together by means of a peptide bond.…
Q: Which of the following best characterizes ANABOLISM? 1.It is largely exergonic reaction because it…
A: The process of metabolism encompasses both anabolism and catabolism.
Q: Answer the following questions briefly and concisely with not more than 5 sentences. QUESTION : What…
A: Introduction: Body-building is an exercise designed to enhance the human body's muscular…
Q: Polypeptide synthesis (CHOOSE ONE)
A: Proteins are one of the most important macromolecule in living organisms. Proteins are consist of…
Q: In human beings, what is the major control of de novo pyrimidine nucleotide synthesis? A. substrate…
A: De novo synthesis of pyrimidine nucleotides takes place in cytosol, first the pyrimidin is…
Q: For every 6 molecules of 3-phosphoglycerate produced by rubisco, _____ are converted to hexoses…
A: Calvin cycle is the second stage of photosynthesis, where the products of light reactions ATP and…
Q: Hormone Inactive adenylyl cyclase GTP Target cell GDP Blood vessel a Subunit of G, protein 3…
A: Metabolic regulation: Energy production or the synthesis of various end products to meet the energy…
Q: 3. Which metabolic reaction requires an input of energy?
A: Metabolic reactions are those that occur in living organisms that proceed through either synthesis…
Q: Which of the following indicates buffering in the titration given in the image below? 12 1000000 11…
A: A buffer is a aqueous solution which is used to resist the pH change upon addition of acid or base.…
Q: Kindly refer to image and answer the question. 1. Why is there a need to regulate the metabolic…
A: Endocrinology is the branch of science that includes the study of cells and the group of cells…
Q: he following nutrient molecule is digested and transported to a cell where it undergoes further…
A: Beta oxidation is a catabolic process which involves the breakdown of fatty acids into acetyl CoA,…
Q: e Galapagos islands over four generations. How can you explain this ta using your knowledge of…
A: In Galapagos island, these finches (Darwin's finches) adapted various beak sizes depending upon…
Q: Enzymes can be regulated in a many different ways. Covalent modification is one way. Here, the…
A: Phosphorylation- It is effective way of regulating proteins and is reversible post translational…
Q: BIOMOLECULES - MULTIPLE CHOICE - Please answer properly QUESTION : Which of the following best…
A: Introduction: Enzymes are proteinaceous in nature and they are of two types: Protoenzyme which…
Q: where, in relation to the protein-coding region of a gene, are the binding sites that become…
A: Receptors are proteins present in the plasma membrane or cytoplasm or nucleus of a responding cell.…
Q: 7. Complete the table below. Function Catabolic hormone Disorder/Disease due to abnormal activity
A: Cells communicate with each other via signal-receptor signaling. A signal molecule binds to…
Q: hich of the following is correct about the structure of proteins? a- the number of peptide bonds is…
A: The peptide bond is the bond that connects individual amino acids in a polypeptide. It is an amide…
Q: P*O*Z*Y# Example lac+ I*p*o+z*Y* FP*0*Z*Y* IP*o*z*Y* -P#O*Z¥Y* I*p*oCz*Y C I*p*o+z*Y
A: The lac operon is a catabolic operon and is expressed in the presence of the inducer lactose.
Q: how many atp can a palmitic acid yield from nadh?
A: A fatty acid is made up of a straight chain of an even number of carbon atoms with hydrogen atoms…
Q: 11. How can you relate waterfalls to a mole of glucose? 12 What are the stens in catabolism?
A: Potential energy- Energy in stored form Kinetic energy- Energy when it gets released
Q: Choose the INCORRECT statement: Group of answer choices All statements are incorrect. Each ribosome…
A: The ribosome is a translation machine that with the help of translation factors and amino acyl tRNAs…
Q: The dose to the body was 40 mSv.
A: Environmental Radiation Protection Standards for Nuclear Power Operation According to this rule the…
Q: 1.Rubisco, which catalyzes the fixation of carbon dioxide during photosynthesis in plants and many…
A: Ribulose-1,5-bisphosphate carboxylase/oxygenase (Rubisco) is the predominant protein in…
Q: Draw the Fischer projection of galactcose and show its catabolism until it releases carbon dioxide…
A: Galactose is an aldohexose. It is a C-4 epimer of glucose. The source for the galactose is a…
Q: If the pH of a voledronic acid solution is 5.8, and the voledronate concentration is 9 mM, what is…
A: Henderson Hasselbalch equation can be used to determine the concentration of a weak acid in an…
Q: What four major classes of eicosanoids are derived from arachidonate, and what are representative…
A: Introduction: Eicosanoids are the metabolites of arachidonic acid derived from the action of the…
Q: A sample was placed on a chromatography column. Dichloromethane was used as the eluting solvent. All…
A: Column chromatography is a technique that allows chemists to separate substances based on their…
Q: What are two different ways that cytochrome oxidase contributes to the proton gradient?
A: Glucose is broken down into pyruvate and pyruvate is used to produce acetyl CoA that enters TCA…
Q: Show the complete reaction mechanism for the deamination of alanine until the amino group is…
A: The deamination of the alanine amino acid takes place with the help of the transaminase enzyme to…
Q: REVIEW QUESTIONS: What are the basic concepts and precautions to be observed in specimen collection…
A: “Since you have asked multiple question, we will solve the first question for you. If youwant any…
Q: 3. For an endergonic, what is the value of AG?
A: Reactions are classified as exergonic reaction and endergonic reactions based on the amount of…
Q: What are the two major products of phospholipid digestion?
A: Phospholipids are a kind of lipid that includes a hydrophilic "head" that contains a phosphate group…
Q: Compound X is an inhibitor in respiratory electron transfer. It binds to the Fe3+ of Complex IV…
A: Complex IV is the terminal electron acceptor in the electron transport chain (ETC). ETC is an…
Q: Discuss the importance of stereochemistry in biomolecules.
A: Stereochemistry is the study of spatial arrangement of of molecules and the relation between its…
Q: Which of the following is correct regarding the relationships of intermediates in Lipogenesis and…
A: The citric acid cycle (TCA cycle ) is the central metabolic cyclic pathway in aerobic cellular…
Q: Question: Glutamic acid is the parent compound of monosodium glutamate (known as MSG), which is used…
A: Glutamic acid is a polar acidic amino acid. In addition to the amino and carboxyl groups attached to…
Q: hoose a specific drug name (NOT A DRUG CLASS) under CELL WALL SYNTHESIS INHIBITORS, and DRAW the…
A: Answer for the following question are :
Q: questions properly. - Multiple choice 1. Find out the reason why antiparallel beta sheets are more…
A: Primary amino acid sequences forms secondary structures like alpha-helix wnd Beta-sheets. t-RNA…
Q: Read each pair of sentences and then choose the letter of the correct answer. Your answer must be in…
A: Glycolysis is a cytoplasmic pathway. In this pathway glucose is broken down into two three-carbon…
Q: Use the Michaelis-Menten equation to complete the enzyme kinetic data set, when Km is known to have…
A: The Michaelis-Menten equation can be written as: V0=Vmax × [S]Km + [S] From the question it is given…
Q: Why doesn’t the brain utilize fatty acids for energy
A: The oxidation of fatty acids provides around 95% of the energy derived from fat. Free fatty acids…
Q: Sequence of Carminic acid
A: Carminic acid is a type of red dye that is extracted from various insect taxa like- Kermes vermilio,…
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 4 images

- 8. The chemical structure of Coenzyme A contains the following except— a phosphoanhydride moiety. a β-mercaptoethylamine residue. a lipoic acid residue. a pantothenic acid residue. an adenosine-3’- phosphate.1. provide three reasons why most of the research on carotenoids concentrates on b-carotene. 2. draw the retinol ester that has stearic acid as the carboxylic acid portion of its structure.draw all the structures of the tribasic amino acid lysine involved in the equilibrium reactions that would take place during titration against NaOH, starting with the fully protonated form below (draw the R-group in full). H;N+- CH - COOH (CH2)4 NH3+
- What term best describes the isomeric relationship between alpha-D- glucopyranose and alpha-L-glucopyranose? A) structural isomers B) enantiomers C) epimers D) diastereoisomers, but not epimers E) not isomersName and draw the structure of the a-keto acid resulting when each of the following amino acids undergoes transamination with a-ketoglutarate: (a) aspartate, (b) glutamate, (c) alanine, (d) phenylalanine.6 (a) A decapeptide has the following amino acid composition: Ala2 , Arg, Cys, Glu, Gly, Leu, Lys, Phe, Val Partial hydrolysis yields the following tripeptides: Cys-Glu-Leu + Gly-Arg-Cys + Leu-Ala-Ala+ Lys-Val-Phe + Val-Phe-Gly. Reaction of the decapeptide with 2,4-dinitrofluorobenzene yields 2,4-dinitrophenylysine. From the experimental data, deduce the primary structure of the decapeptide. (b) Suggest a scheme you will follow to synthesize the dipeptide Ala-Gly
- 1) Draw the structures and provide the systematic names of the following fatty acids (all double bonds are in cis- configuration).a) 24:0b) 18:3(Δ6,9,12)c) 20:3(Δ8,11,14)2) Define and briefly explain the process of saponification.3) Commercial vegetable oils are subjected to partial hydrogenation processing before they are made available on the market;a) Explain the importance of this partial hydrogenation on cooking oil.b) Give two reasons why un-hydrogenated vegetable oil is not suitable for cooking.c) What undesirable effect does partial hydrogenation cause?4) Platelet-activating factor and Prostacyclin are lipids that play roles affecting coagulation.a) Differentiate the roles of each aforementioned lipid with reference to coagulation. b) What specific type of lipids does each molecule belong to?5) Draw the structures of lecithin and cephalin, outlining the general features. 6) Explain the chemical importance of having 2-deoxy-D-ribose in DNA over D-ribose.7) Draw a schematic…9 draw an isomer of the triglyceride in sunflower seed oil. Show the reaction and write out the products when this triglyceride is hydrogenated.4. Draw the condensed structural formula for the triacylglycerol made from 3 saturated fatty acids that are 24 carbons long Using diagram from 1 as the reactant, draw the full equation (using condensed structural formulas) for this triacylglycerol undergoing saponification, using NaOH
- 1. a. Explain why the melting point of palmitic acid (16 carbons, no double bonds) is slightly lower thanthat of stearic acid (18 carbons, no double bonds). Explain why the melting point of oleic acid (18carbons, one double bond) is lower than that of stearic acid b. A mixture of lipids containing phosphatidic acid, cholesterol, testosterone, phosphatidylserine, andphosphatidylethanolamine was applied to a hydrophobic interaction chromatography column. Thecolumn was washed with a high salt buffer, and the lipids were then eluted with decreasing saltconcentrations. In what order would the lipids be eluted from the column? Explain your answer.IV. Draw the Fischer projection formula for the enantiomer of each of the following monosaccharides.What is the product of intramolecular dehydration of pentanol-2 CH3CH(OH)CH2CH2CH3? Select one:a. Pentene-2b. Pentene-3c. Pentene-1d. Pentane

