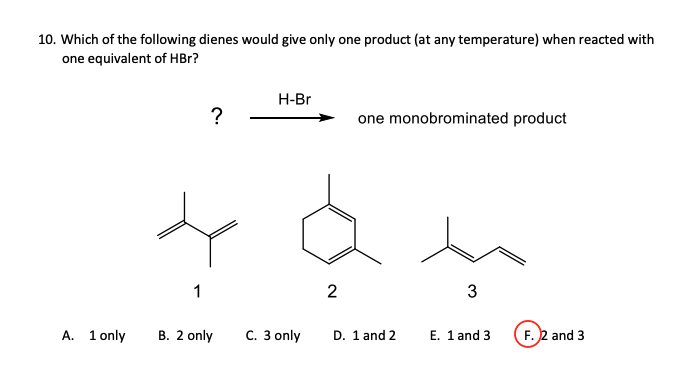
10. Which of the following dienes would give only one product (at any temperature) when reacted with one equivalent of HBr? ? H-Br one monobrominated product
Q: Which mechanism below depicts the expected flow of electrons in homolysis of a covalent bond? Br A)…
A: Free radicals are highly reactive intermediate species that are formed by the homolytic cleavage of…
Q: x100 eyepiece graticule 0 10 20 30 40 50 60 70 80 90 100 40 հավիտենականի 70 60 stage micrometer…
A: First do the addition of all the numbers and then divide it by the total number of observations to…
Q: 7. Determine the pH for the titration of 25.0 mL of 0.500 M ammonia, NH3, with 1.00 M hydrochloric…
A:
Q: In the 1H-NMR spectrum of 1,2,3- tribromopropane, the signal of the circled proton will appear as a
A: NMR spectroscopy is a very important tool for the determination of the structure of the organic…
Q: For the reaction 2NH3(g) + 3N₂O(g) → 4N₂ (9) + 3H₂O(g) = -879.5 kJ and AS = 288.1 J/K The standard…
A: According to the second law of thermodynamics, the spontaneous nature of the reaction is determined…
Q: For which of the following alkenes will cis- and trans- isomers not exist? H3C CH3 H3C H₂C- -CH3 H…
A:
Q: Consider the foo ng data on some weak acids and weak Dases. formula hydrocyanic acid HCN…
A: The question is based on the concept of salt hydrolysis.We need to arrange given salts in increasing…
Q: What will be the product in the given reaction ОН CH3C AIC13
A: In the Fridel-Craft reaction, a hydrogen atom of benzene or benzene derivatives is substituted by an…
Q: When potassium chloride reacts with oxygen under the right conditions, potassium chlorate is formed:…
A: Answer:Here: ai and bi are the stoichiometric coefficients of respectiev species.
Q: Provide the correct IUPAC name for the compound shown here. H3C. .. CH3
A: -Select the parent chain with the longest possible continuous carbon chain with a maximum number of…
Q: Write a balanced chemical reaction for the oxidation of succinate to carbon dioxide coupled to the…
A: Answer:Oxidation: Lose of electronsReduction: Gaining of electrons
Q: Hydrolysis of decapeptide P with the enzyme trypsin affords the following fragments: Gly-Asp,…
A: This is fragmentation of peptide chain. We know trypsin cleaves C-terminal of Arg, Lys. while…
Q: Draw the Lewis structure for ammonium (NH). Does this molecule exhibit resonance?
A: We are to draw Lewis structure of ammonium ion and find out if it exhibit resonance.When a H atom is…
Q: 8. For the compound (E)-3-methyl-2-hepten-6-yne [a.k.a. (E)-3-methylhept-2-en-6-yne] whose structure…
A: We have to predict the products of the ozonolysis of the given reactant.
Q: 5. Predict the correct product for the following Aldol reaction and draw the detailed mechanism…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: Predict the GEOMETRY of the NITROGEN atom(s) in the following compound:
A: The structure of a molecule can be determined by using the VSEPR theory. According to this theory…
Q: Classify each of the molecules below. 1° alcohol 2° alcohol 3° alcohol 4° alcohol OH 1° alcohol O2°…
A: Any carbon atom attached to one other carbon atom or no other carbon atom is called as primary…
Q: ▼ Part A At what temperature would a 1.95 m NaCl solution freeze, given that the van't Hoff factor…
A: Given,A) molality of NaCl = 1.95 m Van't Hoff factor = 1.9 Kf = 1.86 oC/mB) Osmatic…
Q: Convert the following carbohydrate from a Chair Conformation to a Fischer Projection.…
A:
Q: In the laboratory, a general chemistry student measured the pH of a 0.513 M aqueous solution of…
A:
Q: Consider the following compound: Part 1 of 2
A: Identical compounds: Same compoundConstitutional isomers: Compounds with same molecular formula but…
Q: Assuming complete dissociation of the solute, how many grams of KNO3 must be added to 275 mL of…
A:
Q: Bi Tutored Practice Problem 7.3.1 COUNTS TOWARDS GRADE Apply the Pauli exclusion principle. (a)…
A: According to Pauli exclusion principle no two electrons in same atom can have identical set of…
Q: Which of the following is the most likely structure of the following cation after rearrangement? Xo
A: Given is carbocation. Carbocation is species in which carbon carries positive charge.
Q: Explain the relationship between an atom’s number of electrons and its relative stability.
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: Which is an AB3 molecule that has μ ‡0? O PC13F2 O SeCl₂ O NBr3 O SeO₂ O Bl3
A:
Q: How many grams of sulfur are there in 197 g of Al2(SO4)3 (342.2 g/mol)?
A: If we divide the mass of a compound by its molar mass, we will get the number of moles of the…
Q: Calculate a value for the equilibrium constant for the reaction O2 (g) + O(g) 03 (9) given hv NO₂…
A: Equilibrium constant K for a reaction is defined as the product of the molar concentrations of the…
Q: 12. Which two of the following molecules lack a plane of symmetry? Me Me ار و Hil OH MC H Br' ... OH…
A: Given are organic compounds.Plane of symmetry is an imaginary line or plane that bisects molecules…
Q: Write the systematic name of each organic molecule: CH3 CH3 CH3-CH-CH-CH-CH₂-CH3 CH3 structure…
A: Given Organic molecules :We need to write the systematic name of the above organic molecules.
Q: Need help with this pjhysics theoretical question: The most surprising evidence from Rutherford’s…
A: Ernest Rutherford's gold foil experiment, conducted in 1909, marked a pivotal moment in the history…
Q: P Please don't provide the handwriting solution
A: Whether equilibrium lies on the right side or left side depends upon the acidity of the reactant and…
Q: The concentration of Ag+ in a saturated solution of Ag2CrO4 at a certain temperature is 1.30 x10-4…
A:
Q: Five- and six-membered ring structures are common in carbohydrates and are often in equilibrium with…
A: The compounds with the same molecular formula but different bond connectivity are constitutional or…
Q: Select to Draw Q 1. NaOCH2CH3 2. (CH3)2CHCI (1 equiv) H3O+ heat Select to Draw 1. NaOCH2CH3 2. CH3Br…
A: Given is organic reaction.These reactions collectively known as Acetoacetic Ester synthesis…
Q: In much the same way that they react with H₂, alkenes also react with D2 (deuterium is an isotope of…
A: Alkenes are hydrogenated by the reaction with hydrogen in presence of finely divided nickel,…
Q: Evaluate the following statement: The following compound violates Bredt's rule. True False
A: It states that double bond cannot be placed at the bridgehead of a bridged ring system, unless the…
Q: 1. Identify the Lewis acid and the Lewis base in the following reaction: Ag (aq) + 2NH₂ (aq) à…
A: Ag+(aq)+2NH3(aq)-------->Ag(NH3)2(aq) We have to identified Lewis acid and Lewis base.Identify…
Q: 3. Identify the stronger Brønsted-Lowry base of the following pairs of bases. Justify your answer…
A:
Q: C21-6: Estimate AH for the following reaction: 2Br2 (9) C₂H₂Br4 (9) C₂H₂(g) +
A: The given balanced reaction is The bond energies for each bond are-
Q: Predict the major products of both organic reactions. Be sure to use wedge and dash bonds to show…
A: Hydration of alkene proceeds by an electrophilic addition reaction mechanism to form an alcohol.…
Q: Draw the structures of two different alkyl bromides that could yield the alkene product sho t shown…
A: Given that, the structure of the alkene product is:
Q: If the Ka of HCIO is 3.0x 10-7, what will the pH be if you add 3.8 g HCIO to a 200 mL solution of…
A: Given,mass of HClO = 3.8 gMolarity of NaOH = 0.1 Mvolume of NaOH = 200 mLKa of HClO = 3.0 x 10-7
Q: Propose structures for compounds that fit the following ¹H NMR data: (b) C³H5Br (a) C5H10O 0.95 8 (6…
A: Givena): Molecular formula C5H10Ob): Molecular formula C3H5Br
Q: For the following reactions, predict whether the mole fraction of the reactants or products…
A: Answer:Le-chatalier's principle states that on changing any parameter of the system that is at…
Q: What is the rate at which Br⁻(aq) disappears in the reaction below if the rate of disappearance of…
A: Given reaction is:BrO3- + 5Br- + 6H+ => 3Br2 + 3H2O
Q: Reaction 1: H I Br: + How н I :O:
A: In the two incomplete reactions, the reactants are haloalkane and water. The two haloalkane…
Q: Draw the product of the substitution reaction shown below. Ignore the inorganic byproducts. CH3SNa…
A: Alkyl halide undergo substitution reaction with CH3SNa.
Q: Select all of the curved arrows that are correct for this reaction. The products of this reaction…
A:
Q: An arctic weather balloon is filled with 25.3 L of helium gas inside a prep shed. The temperature…
A: The initial volume of the balloon = 25.3 LThe initial temperature of the balloon =The final…
Step by step
Solved in 3 steps

- (1) Which is an isolated diene? (2) Which is an alkene with possible rearrangement product (1,2-H shift) with reaction to HI? (3) Whixh isa diene capable of having 1,2 and 1,4-addition products? (4) Which is a conjugated diene?17.18 Rank the following dienes in terms of reactivity in DielsAlder reactions (from least reactive to most reactive).Consider the following dienes in a Diels–Alder reaction with ethene. Which will react the fastest? Which will react the slowest? Explain
- The Birch reduction is a dissolving metal reaction that converts substituted benzenes to cyclohexa-1,4-dienes using Li and liquid ammonia in the presence of an alcohol. Draw a stepwise mechanism for the following Birch reductionRank the following dienophiles in order of increasing reactivity.Explain the Carbocation Rearrangements ?
- When Br2 is added to buta-1,3-diene at -15 °C, the product mixture contains 60% ofproduct A and 40% of product B. When the same reaction takes place at 60 °C, theproduct ratio is 10% A and 90% B.Show why A predominates at -15 °C and B predominates at 60 °C.Why a conjugated diene is more stable than an isolated diene ?1. Which among these would be the most stable carbocation?a. Allylicb. Vinylicc. Primaryd. Secondary2. What is the primary reason for the stability of tertiary carbocations?a. Free rotationb. Resonancec. Hyperconjugation
- What diene and dienophile are needed to prepare each compound by a Diels–Alder reaction?Rank the following dienes in their rate of reaction with the same dienophile in a Diels-Alder reaction. For example, 1 = fastest or most reactive diene, 4 = slowest or least reactive diene.What diene and dienophile are needed to prepare attached compound by aDiels–Alder reaction?

