11) q of combustion of the food sample equals a) q absorbed by the calorimeter -q absorbed by the water b) q absorbed by the water -q absorbed by the calorimeter c) negative (q absorbed by the water +q absorbed by the calorimeter) d) (q absorbed by the water + q absorbed by the calorimeter) 12) Heat produced by combustion of a 3.000 g food sample was -9643J. How much heat, in Joules, was pipduced per gram? a) 3 J/g b) 4.18 J/g c) 9643 J/g d) 3214 J/g 13) Heat produced by combustion of a different food sample is 10,000 J/g. How many Calories per gram is that? (1 capital C Calorie = 4184 J). %3D a) 2 Cal/g b) 4 Cal/g c) 9 Cal/g d) 4184 Cal/g
11) q of combustion of the food sample equals a) q absorbed by the calorimeter -q absorbed by the water b) q absorbed by the water -q absorbed by the calorimeter c) negative (q absorbed by the water +q absorbed by the calorimeter) d) (q absorbed by the water + q absorbed by the calorimeter) 12) Heat produced by combustion of a 3.000 g food sample was -9643J. How much heat, in Joules, was pipduced per gram? a) 3 J/g b) 4.18 J/g c) 9643 J/g d) 3214 J/g 13) Heat produced by combustion of a different food sample is 10,000 J/g. How many Calories per gram is that? (1 capital C Calorie = 4184 J). %3D a) 2 Cal/g b) 4 Cal/g c) 9 Cal/g d) 4184 Cal/g
Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter10: Energy
Section: Chapter Questions
Problem 81AP
Related questions
Question
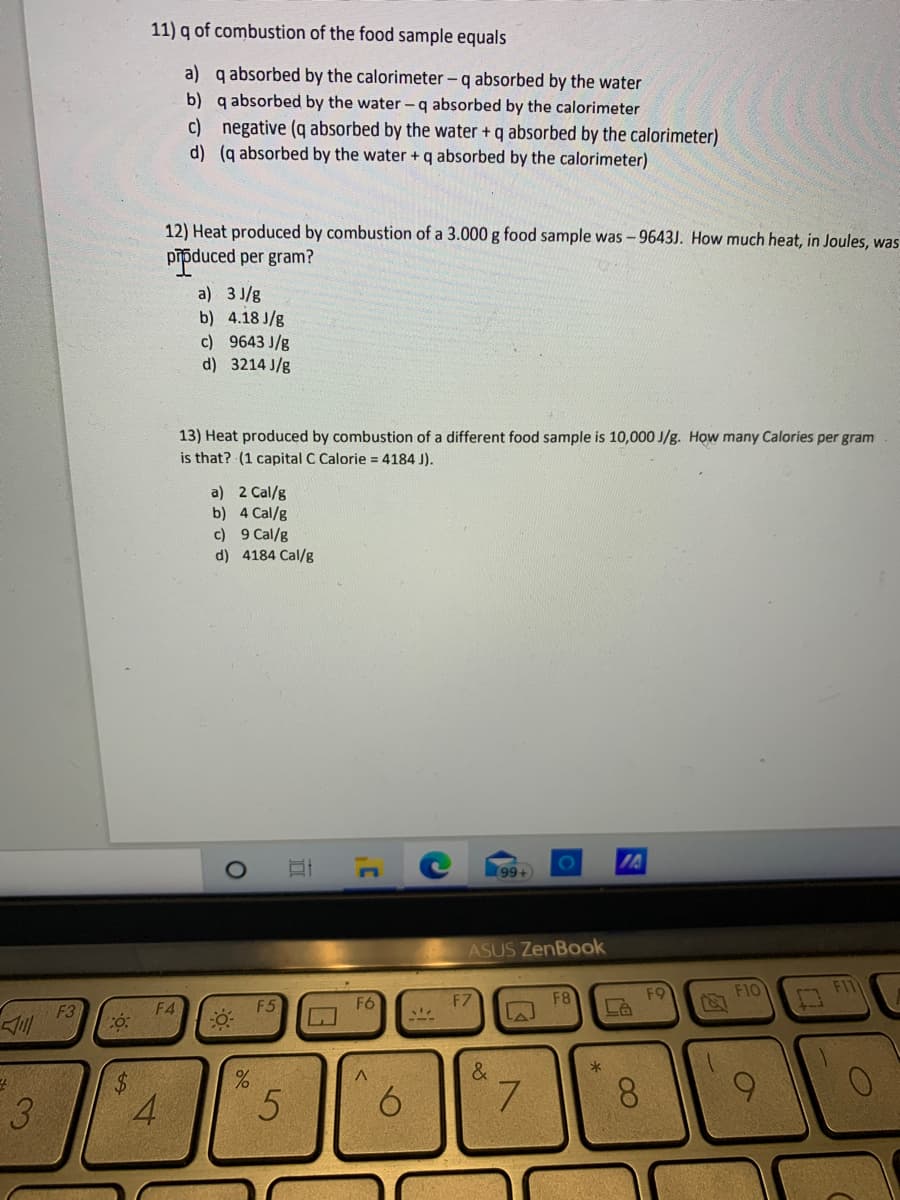
Transcribed Image Text:11) q of combustion of the food sample equals
a) q absorbed by the calorimeter - q absorbed by the water
b) q absorbed by the water - q absorbed by the calorimeter
c) negative (q absorbed by the water + q absorbed by the calorimeter)
d) (q absorbed by the water + q absorbed by the calorimeter)
12) Heat produced by combustion of a 3.000 g food sample was -9643J. How much heat, in Joules, was
pipduced per gram?
a) 3 J/g
b) 4.18 J/g
c) 9643 J/g
d) 3214 J/g
13) Heat produced by combustion of a different food sample is 10,000 J/g. How many Calories per gram
is that? (1 capital C Calorie = 4184 J).
a) 2 Cal/g
b) 4 Cal/g
c) 9 Cal/g
d) 4184 Cal/g
99+
ASUS ZenBook
F10
F1
F7
F8
F9
F3
F4
F6
$4
5
7.
8.
3.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

World of Chemistry
Chemistry
ISBN:
9780618562763
Author:
Steven S. Zumdahl
Publisher:
Houghton Mifflin College Div

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

World of Chemistry
Chemistry
ISBN:
9780618562763
Author:
Steven S. Zumdahl
Publisher:
Houghton Mifflin College Div

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning