Results, and calculations: Complete the table below, using Fe as the metal Wt. of Calorimeter Wt. of Calorimeter + water Weight of Water Initial temperature of water, (t)w (e) Initial temperature of aluminum calorimeter, (t)cal (f) Initial temperature of metal (t)metal (g) Weight of metal sample, mmetal (h) Final temperature of water, (te)w (i) Final temperature of calorimeter, (t)eal. (1) Final temperature of metal (k) Heat absorbed by the metal, gcal (1) Heat absorbed by the water, gw (m) Experimental specific heat of metal sample (n) Theoretical specific heat of metal sample (0) Percent Error 20g 105g 85g 28 °C 100 C 26 g 30.5 °C Joule Joule J/g°C 0.460 J/gC
Results, and calculations: Complete the table below, using Fe as the metal Wt. of Calorimeter Wt. of Calorimeter + water Weight of Water Initial temperature of water, (t)w (e) Initial temperature of aluminum calorimeter, (t)cal (f) Initial temperature of metal (t)metal (g) Weight of metal sample, mmetal (h) Final temperature of water, (te)w (i) Final temperature of calorimeter, (t)eal. (1) Final temperature of metal (k) Heat absorbed by the metal, gcal (1) Heat absorbed by the water, gw (m) Experimental specific heat of metal sample (n) Theoretical specific heat of metal sample (0) Percent Error 20g 105g 85g 28 °C 100 C 26 g 30.5 °C Joule Joule J/g°C 0.460 J/gC
Chemistry: Principles and Practice
3rd Edition
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Chapter5: Thermochemistry
Section: Chapter Questions
Problem 5.59QE
Related questions
Question

Transcribed Image Text:Results, and calculations: Complete the table below, using Fe as the metal
20g
105g
Wt. of Calorimeter
Wt. of Calorimeter + water
Weight of Water
Initial temperature of water, (t)w
85g
28 °C
(e) Initial temperature of aluminum calorimeter, (t)cal
(f) Initial temperature of metal (ti)metal
(g) Weight of metal sample, mmetal
(h) Final temperature of water, (tg)w
) Final temperature of calorimeter, (to)cal.
G) Final temperature of metal
(k) Heat absorbed by the metal, gcal
(1) Heat absorbed by the water, gw
100 C
26 g
30.5 °C
Joule
Joule
(m) Experimental specific heat of metal sample
(n) Theoretical specific heat of metal sample
(0) Percent Error
J/gC
0.460 J/g C
%
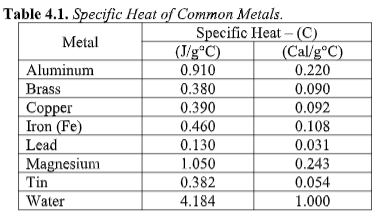
Transcribed Image Text:Table 4.1. Specific Heat of Common Metals.
Specific Heat – (C)
(Cal/g°C)
0.220
Metal
Aluminum
(J/g*C)
0.910
Brass
0.380
0.090
0.390
0.092
Сорper
Iron (Fe)
0.460
0.108
0.031
0.243
Lead
0.130
Magnesium
Tin
1.050
0.382
0.054
Water
4.184
1.000
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning