Q: If (1.7479x10^-6) mol of CaCO3 (calcite) and (5.91x10^-8) mol CaSO4•2H2O (gypsum) were to completely…
A: Ionic activity product is defined as the product of activity of all components present in the…
Q: 1.1 For the following reaction carried out at constant temperature and pressure, what is true about…
A: Given : gaseous reaction
Q: The activated complex is an unstable molecule has the maximum potential energy possible may continue…
A: The activated complex, also known as the transition state, is a short-lived intermediate species in…
Q: Using the table of standard formation enthalpies that you'll find under the ALEKS Data tab,…
A: The given reaction is as follows: C2H2(g) + 2H2(g) →C2H6(g) The enthalpy of reaction = ?
Q: 2. (a) MeO Least basic List the following compounds in order of increasing basicity. NH₂ NH₂ NH (N-J…
A:
Q: The solubility of O2 in water is 5.85 x 10-4 M at 25 oC and 0.45 atm of O2 pressure . What will the…
A:
Q: Complete each of the following statements for a molecule of SbCl3: Drag the terms on the left to the…
A: we have to fill the missing boxes for the SbCl3 molecule
Q: 7. Predict the product(s) AND draw the mechanism for the following reaction. TO CH3CH2OH/H2SO4 وی کے…
A: In ring opening of epoxide in presence of acid first protonation of epoxide takes place followed by…
Q: The units for the rate constant for a zero order reaction are A) S-¹ B) M-¹s¹ C) M. s-¹ D) M-²S-¹ T
A:
Q: 385000 Jam STARTING AMOUNT 385000 km x 1000 m 1 km X X 11080 s 3470 min X 1000 1 1 min 60 s = min…
A:
Q: reacting with 1mol HBr, for each compound which double bond is more reactive and why
A: Addition reaction of HBr on alkene is known as hydrobromination reaction. Addition bromine is takes…
Q: following equilibria would be shifted to the left? ОН A) :0: B) mbe 9qvi C) OH, то CH,Ö: T + CH OH_…
A: Whenever an acid loses hydrogen ion it forms anion. The lion is called conjugate base of that acid.…
Q: If the K₂ of a weak acid is 1.6 x 10-8, the K, of its conjugate base partner must be which of the…
A:
Q: he enthalpy change for the reaction of hydrogen gas with iodine to produce hydrogen iodide is 53.1…
A: Given reaction is, H2(g) + I2(s) -----> 2 HI(g) ΔH = 53.1 kJ
Q: 7) How many isomers can you construct for the molecular formula C,H,O? Use a red ball with two holes…
A: Isomers are those compounds which have the same molecular formula but different structures either…
Q: Ⓒ Diaw structural formulas for the major organic products for XOH SOCI₂ CH b) 2) HO Hcl -OH HBr…
A:
Q: OH CH₂OH H OH Н OH OH In the box below, draw the open-chain structure (as a Fischer projection)…
A: We have to convert this compound from Haworth to fisher projection.
Q: The plot below shows the concentration of reactant A during a chemical reaction (A (ag) + B(ag) C…
A: We have find out the instantaneous rate at 80 seconds.
Q: Nitrogen monoxide, NO, is formed in automobile exhaust by the reaction of N₂ and O2 (from air).…
A: Given that, a equilibrium reaction is N2g + O2g ⇌ 2NOg. The equilibrium constant (Kc) is 0.0025 at…
Q: < 2:19 A) S-¹ B) M S-¹ What are the units for the rate constant of a first-order reaction? C) M-¹ s¹…
A: Introduction The rate constant of a reaction is an important parameter that describes how quickly a…
Q: onsider the table. Metal Tm (K) ΔHfus (kJ/mol) Tb (K) ΔHvap (kJ/mol) Li 454 2.99 1615 134.7 Na 371…
A: Given, Tm (K)=302 ΔHfus(kJ/mol)=2.10 Tb (K)=942 ΔHvap (kJ/mol)=66 To calculate, ΔSfus and ΔSvap
Q: OH + Na OE a. draw the products and label the acid, base, conjugate acid and conjugate base with pk,…
A: A base is a compound which abstracts a proton from an acid and form a conjugate acid. And the acid…
Q: See Figure 3-5. Which is a bond line drawing of (CH3)2CHCH₂OC(CH3)3? A B с
A: Answer : Given that, The molecule is : (CH3)2CHCH2OC(CH3)3
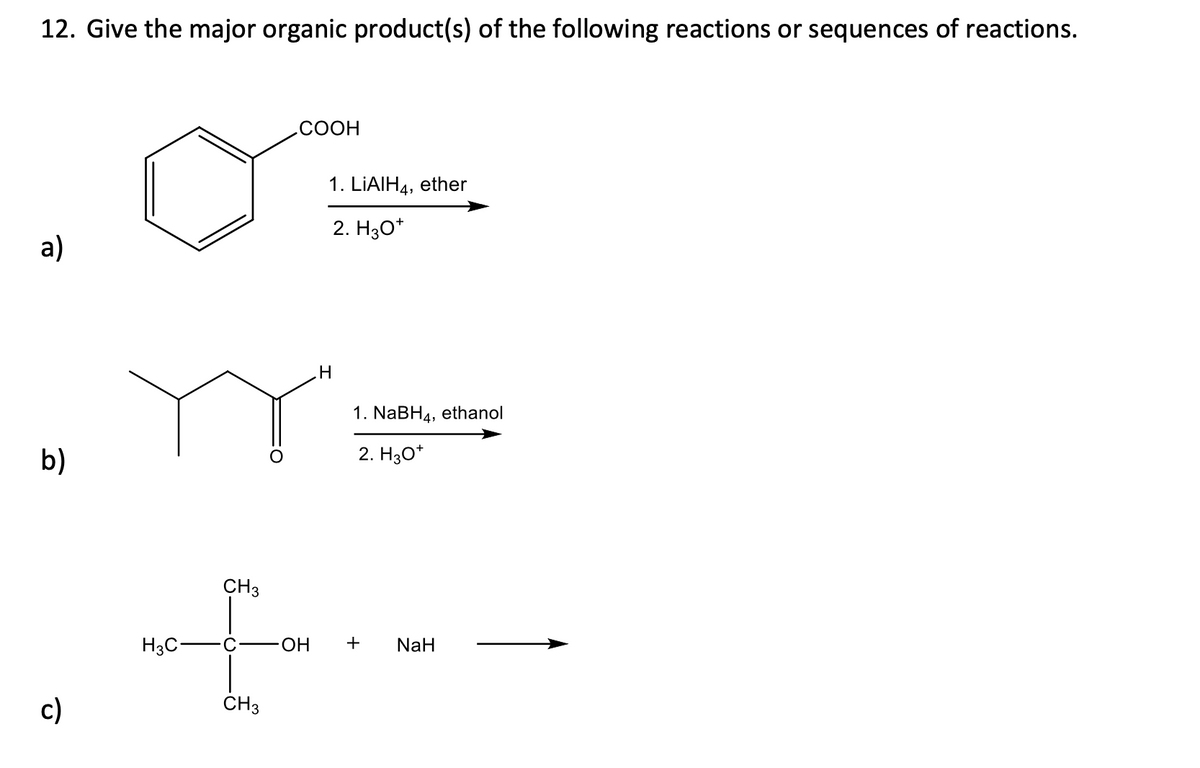
Q: 4. Predict the product(s) for the followi O O 1. excess LiAlH4 2. H₂O
A: LiAlH4 is a reducing agent. The carbonyl C=O gets reduced to C-OH. The lactone gets reduced to two…
Q: A 300 mOsm glucose solution is separated from a 250 mOsm NaCl solution by a membrane that is…
A: Given , Glucose = 300 mOsm NaCI = 250 mOsm
Q: If a reaction is first order with a rate constant of 0.0450 s¹, how much time is required for 65% of…
A: The given data is as follows: The rate constant for first order reaction = 0.0450 s-1 Let the…
Q: Practice problems for Markovnikov addition of water and hydroboration/oxidation. Give the complete…
A: According to Markovnikov's addition, the hydrogen adds onto the carbon with more number of hydrogens…
Q: Stereoisomers There is another kind of isomeric relationship possible for organic structures. This…
A: This question belongs to stereoisomerism.
Q: A compound has the molecular formula: C4H8O2 and gives the following C-13 NMR spectrum. Provide the…
A: Given 13 CNMR spectrum of molecule of C4 H8O2
Q: 2 A(g) + B(g) → C(g) + D(g) is [A]. (M) 0.150 0.450 0.450 [B]. (M) 0.300 0.300 0.150 Question 10 of…
A: Introduction The rate law in chemistry is an equation used to describe the rate of a chemical…
Q: An aromatic ring has how many units of unsaturation? 1 2 3 4 5
A: Introduction
Q: Question 1 The reaction below is third order in A and second order in B, what is the rate law? The…
A: Given: Third order in A Second order in B To determine: The rate law The units of rate constant must…
Q: Consider the representation below of an ideal gas in a tiny container at a given temperature. Which…
A: Answer : Ideal gas are those gases which have no interaction among the gases molecules. There exists…
Q: What are intermolecular forces and how are they related to the solubility and properties of liquids…
A: Intermolecular forces (IMFs) are the forces of attraction or repulsion between neighboring molecules…
Q: Suppose that the freezing point of a 1.0 m electrolyte solution is normally 10.521 C with a Kb of…
A: Colligative properties are properties of a solution that depend on the number of solute particles…
Q: one is incorrect
A: The given structure:
Q: Draw the 2D formula for the following condensed formulas
A:
Q: K Problem 36 of 40 Draw the thiol needed to produce the sulfide under the conditions shown. Draw…
A: In the first question, we will draw the structure of the starting material ( which is thiol ) by…
Q: If 7.90 kJ of heat is needed to raise the temperature of a sample of metal from 15 °C to 20. °C, how…
A: The heat transfer is given by q = m x c x ΔT where: q = heat gained or lost (in Joules) m = mass of…
Q: Draw the two products of the reaction shown below. Ignore inorganic byproducts. HBr
A:
Q: 11. How much fluorouracil powder is in 5% Efudex® cream 50 g? 12. How much oxiconazole nitrate…
A:
Q: When solid lead(II) phosphate is in equilibrium with its ions, the ratio of lead(II) ions to…
A: Given : lead(ii) phosphate is at equilibrium with its ions
Q: 30.0 mL of 0.50 M ethylamine (Kb = 5.6 x 10-4) is titrated with 0.75 M HCI. 4.99 7.00 4.56 5.91 6.23…
A: Answer:- This question is answered by using the simple concept of titration of weak base against the…
Q: We mixed some ethylene glycol with 346 g water to reduce the equilibrium vapor pressure of water…
A: Answer: When a non-volatile solute is added in a solvent it causes a depression in freezing point…
Q: The density of an (NaCl) ionic solid is 3.34 g/cm3 and the length of an edge is 482 pm. a. How…
A: Given: density of an (NaCl) ionic solid = 3.34 g/cm3 length of an edge (a) = 482 pm…
Q: Provide a name or a Structure
A: Given: name and structure of molecule Tip : identify parent group
Q: In Part C of the experiment, you added a 6.0 NaOH solution to the aqueous solution of NaCl. Did the…
A: When NaCl is dissolved in water , it undergoes dissociation to form sodium and chloride ions.…
Q: calculate the temperature change when 32 grams of tungsten absorbs 21 joules of heat
A:
Q: What mass of NH4Cl must be dissolved in 100 mL of H₂O to make a 0.250 M solution?
A:
Q: only need help with PART B. what is the biggest groups? The NH2? The CO2H? the CH3 groups.?
A: We have find out the Newman projection of the highest and lowest energy confirmation.
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 3 images

- please quickly thanks ! 3.Please write out the major reaction and side-reaction in the preparation of ter-butyl.chloride, and write out the key points to use separation funnel in this process.What is reaction 1 and 2 called? Choices: A. Williamson ether synthesis, B. Reduction with Grignard reagent, C. Acidic ether cleavage, D. FC Alkylation What are the reagents for reaction 1 and 2? Choices: A. methanol in acidic medium, B. sodium hydride and bromomethane, C. methyl bromide and aluminum bromide, D. methanoyl bromideI need specific description of the compounds involved in the synthesis (functional groups, specific features). and also describe the steps involved in the synthesis and the role of each step. And most important!!devote one to each step of the synthesis. Each step should clearly indicate the reagent used, the stereochemistry involved in the reaction and its importance or relevance to the multistep synthesis
- (a) Illustrate the following name reactions giving suitable example in each case :(i) Clemmensen reduction (ii) Hell-Volhard-Zelinsky reaction(b) How are the following conversions carried out?(i) Ethylcyanide to ethanoic acid (ii) Butan-l-ol to butanoic acid(iii) Benzoic acid to m-bromobenzoic acidcan i get help drawing out actual structures including the nucleophilic addition of Cy2NH to parafomaldehyde and its hemiaminal intermidiate and the condensation step when it is displaced by terminal alkyne forming allene, also what is dioxane getting rid of as the solvent, thanks(a) Illustrate the following name reactions giving a chemical equation in each case :(i) Clemmensen reaction (ii) Cannizzaro’s reaction(b) Describe how the following conversions can be brought about :(i) Cyclohexanol to cyclohexan-1-one (ii) Ethylbenzene to benzoic acid(iii) Bromobenzene to benzoic acid
- Choose the best reagents from the list provided below for carrying out the following conversion. Match the reagent with the step number. HCl (aq), Zn(Hg) KMnO4, H3O+ CH3Cl, AlCl3 HNO3, H2SO4 Cl2, FeCl3 fuming sulfuric acidPlease complete reactions in clear handwritten of all subpartsCH₂ + Using the reagents below, list in order (by letter, no period) those necessary to prepare 2-butyne from acetylene. Note: Not all spaces provided may be needed. Type "na" in any space where you have no reagent. a. Br₂, heat, light b. Na, (-H₂) c. Nal(SN2) d. NH4NO3 e. 1) NaNH2, liq. NH3, 2) CH3Br f. (CH3)3COK, (CH3)3COH, heat g. NaOEt, EtOH, heat Step #1 Step #2 Step #3
- Propose a short synthesis f or ONE of the following molecules. You can start with acetylene, and alkyl halides from 1-4 carbons in length. You can also use an reagents you wish. a) Hexan-3-one (CH3CH2COCH2CH2CH3)Give mekanisms for reaction 4a and 4 b. Question 5- show general mechanisms of carbonyl group(ketone, aldehyde) reacting in acidic conditions and basic conditions, explain the difference.Provide the reagent(s) for the reactions.

