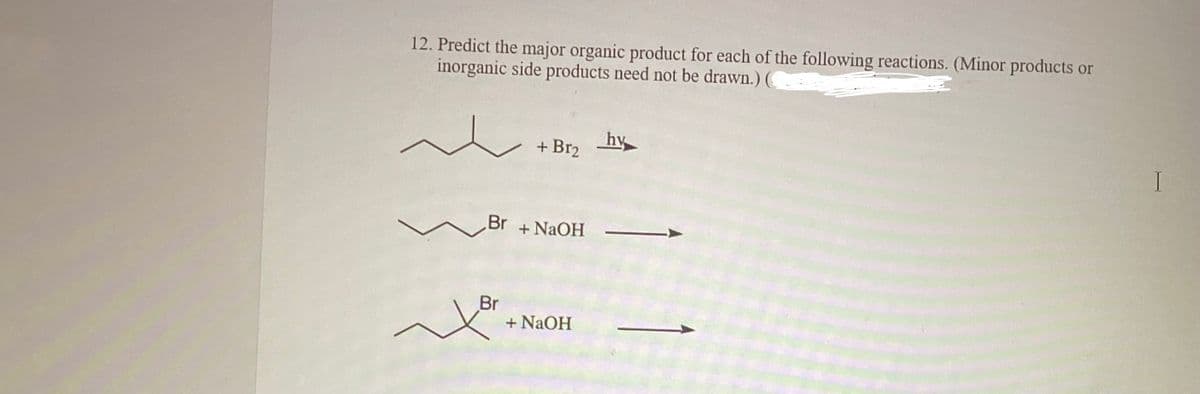
12. Predict the major organic product for each of the following reactions. (Minor products or inorganic side products need not be drawn.) ( ~ + Br₂ Br + NaOH Br you + NaOH hy
Q: n-a transition can be found in these compounds: Alcohols and Aromatic Hydrocarbons. Alcohols and…
A:
Q: Check that the spectrophotometer is on and set at 440 nm. If it is not set up correct D. ill the…
A: We have to plot the graph of absorbance vs time and determine the rate of the reaction.
Q: sing a table of standard reduction potentials, determine which of these reactions (if any) is/are…
A: If the value of standard reduction potential is positive it means the reduction process is…
Q: When the following skeletal equation is balanced under basic conditions, what are the coefficients…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: tch each line drawings on the left to a general functional group representation on the righ CH3- OH…
A:
Q: The graph is derived from the data in the table. Label the axes of the graph correctly such that the…
A: Slope of this plot must be equal = Rate constant = K And the slope must be positive as can be seen…
Q: 5. List the following alkyl halides in order of decreasing reactivity toward SN1/E1 reactions (from…
A: SN1 reaction proceeds through carbocation formation, stability of carbocation increases, reaction…
Q: Determine the [OH-], pH, and pOH of a solution with a [H+] of 4.7 x 10-¹² M at 25 °C. [OH-] = = pH =…
A:
Q: Chemical For How many grams of solid sodium hypochlorite should be added to 1.50 L of 0.185 M…
A: Answer: Buffer solution is a type of solution that resists the change in its pH on adding small…
Q: Hydrogen gas was cooled from 373 K to 283 K. Its new volume is 750.0 mL. What was the original…
A:
Q: I OI and II OI and III Br xxxxx O I, II, III O II, III, and IV O I, II, and IV O IV, V H₂O || shown…
A: Alkyl halide can proceed through SN1 mechanism. In this mechanism there is Formation of Carbocation…
Q: What is the expected major product of the following reaction? Include appropriate stereochemistry,…
A: The given reaction is an example of the base catalysed hydrolysis reaction of ester to form…
Q: A student is making a titration curve. To do so, they added 15.00 mL of 0.2170 M HCl to a flask.…
A: we have to calculate the pH of the solution
Q: Calculate the volume in liters of a 46.2 g/dL iron(11) bromide solution that contains 50.0 g of iron…
A: Concentration of iron(II) bromide solution = 46.2 g/dL Mass of iron(II) bromide = 50.0 g Volume of…
Q: Determine the molarity of a solution with a volume of 490. mL and 0.480 mol of solute dissolved.
A: Since, Molarity is the number of moles of solute dissolve in one litre of the solution. Thus,
Q: If 31.9 g of NaI (MM = 149.89 g/mol) are added to a 500.0 mL volumetric flask, and water is added to…
A:
Q: 1. MeLi 2. 3. H3O+
A: Here new C-C bond is formed Between terminal carbon of alkyne with carbonyl carbon of cyclohexanone.…
Q: 8- Draw the 1H NMR spectrum of Ethyl bromide or 1-Bromoethane. Show the splitting indicating which…
A: The given molecule is an example of alkyl hallide and two different types protons are present.
Q: In the reaction shown below, methane is being H H-C H 0₂ 01C 0 H reduced and is therefore the…
A: ddd Since, In oxidation reaction, Oxidation number increases or number of oxygen increases or number…
Q: draw the Lewis dot structure for both the neutral atom and the stable ion of oxygen.
A: To solve this problem we have to draw the Lewis Structure of Oxygen Atom and Ion .
Q: How many grams (of mass m�) of glucose are in 195 mLmL of a 5.50%% (m/v) glucose solution?
A:
Q: An ideal gas initially at 710 torr and 30.0 °C occupies volume of 2.600 L. Calculate the final…
A:
Q: Provide the name of each compound in the reaction b) Provide the mechanism of the transformation
A:
Q: SECTION 2: Thinking 3. How many grams of air are required for an automobile to travel from Thunder…
A: A question based on mole concept. A set data for combustion of Octane is given using that the mass…
Q: If the Kb of a weak base is 4.7×10-6 ,what is the pH of a 0.12 M solution of this base?
A:
Q: Given that Kw for water is 2.4 × 10-¹4 at 37 °C, calculate the pH of a neutral aqueous solution at…
A:
Q: In the reaction shown below, the benzene ring is NH3 Na ROH HH HH
A: An oxidizing agent is a substance that causes another substance to lose electrons (i.e., oxidizes…
Q: (A) A sample of carbon dioxide gas at a pressure of 1.14 atm and a temperature of 27.1 °C, occupies…
A: Ideal gas equation: PV = nRT At constant T and n PV = constant P1V1 = P2V2 P1 = initial pressure…
Q: 9. Of the compounds below, which is most likely to be one of the products observed for the following…
A: Given reactant is carrying two alcohol and two aldehyde functional groups. Reagent in the first…
Q: Which of the following phase changes is not spontaneous at room temperature? Assume only the first…
A: Which phase change is not spontaneous at room temperature?
Q: H E - N - с || I c=0 I снороза- н-с-он CH₂OPO H он H₂O + Е — N = C но-с - н H CHJOPO3- H - - он он с…
A: In the above reaction hydroxy amine phosphate react with beta hydroxy acetone phosphate to form…
Q: 2. 100.0 mL of a 0.0875 M weak base solution is titrated with 0.250 M HCI. The pH after the ad 13.66…
A: Given, Concentration of weak base (say B) = 0.0875 M Volume of B in mL = 100.0 mL Concentration…
Q: Complete and balance the following redox equation using the smallest whole-number coefficients.…
A: Oxidation half Sn ----> SnO2 Reduction half: HNO3 --->…
Q: A buffer was made using 0.10 moles of A- and 0.01 moles of HA. If the pKa is 5.71, what is the pH of…
A:
Q: Use pH, POH, [H₂O*], and [OH-] relationships. (a) The hydronium ion concentration in an aqueous…
A: pH is the negative logarithm of hydronium ion concentration. pH = -log[H3O+] pH + pOH = 14…
Q: a. How many shared and unshared electrons are there in the structure? b. how many shared and…
A:
Q: What is the pH of a 0.558 M HCIO solution
A: Concentration (c)= 0.558 M pH = ?
Q: If the ?a of a monoprotic weak acid is 8.1×10-6 ,what is the pH of a 0.10 M solution of this acid?
A:
Q: Question 21 Indicate all possible products in the following reaction. (Hint: Under these conditions…
A: To solve this problem we have to complete the given chemical reaction.
Q: A reaction has the stoichiometry: 3A + B → C+ D. The following data were obtained for the initial…
A: this question is based on rate laws and order of reaction
Q: [2] Another group of Aggie students prepared their juice by using an initial concentration of Sodium…
A: The question is based on the concept of chemical equilibrium. we need to calculate concentration of…
Q: 17. What is the AGran at 300K for: 2 NO2(g) → N₂O4(g)? At 300K: Known S (J/(mole-K)): Known AHF…
A: 17. Recall the given reaction, 2 NO2 g → N2O4 g Known S…
Q: Jer 37 Prezo A physician assistant must calculate how many milliliters of a liquid medicine must be…
A: To solve this problem we have to do unit conversion.
Q: When CH, NH, is treated with HCI, one of the products is Cl. Draw the other product. Click and drag…
A: The given question discusses about the reaction of base with acid to form ammonium salt.
Q: When titrating a 25 mL solution of 0.20M CH3COOH with 0.10M KOH, you realize you forgot to add an…
A: The question is based on the titrations. we are titrating a weak acid acetic acid with a strong…
Q: 15 ng 16
A: This reaction will go through mainly the mechanism of 4+2 pi electron system which is a concerted…
Q: 10. Iron has an atomic number of 26 and has an electronic configuration 18² 2s² 2p 3s² 3p6 4s² 3dº.…
A: Number of d electrons in Fe2+ is??
Q: Write the products formed in the following acid-base reaction. NaOH + HCl →> Then, describe the…
A: The hydrogen ion of acid reacts with the hydroxide ion of a base to form salt and water, the…
Q: 6. Which of the following is a conjugate acid-base pair? H3PO4 and HPO4²- HCO3 and CO3²- NaOH and OH…
A:
Q: CAN YOU TYPE IT
A: Let V1 mL of 4% solution and V2 mL of 20% solution are mixed to prepare 400 mL of 8% solution
Step by step
Solved in 3 steps with 2 images

- The two Grignard reactions and the reduction reaction below are all believed to have similar mechanisms. Use the curved arrow formalism to draw a single mechanism for these reactions using :Nuc-1 to indicate the nucleophile. The reacations are: cyclohexylmagnesium chloride + acetone in ether --> 2-cyclohexyl-2-propanol phenylmagnesium bromide + acetone in ether --> 2-phenyl-2-propanol Acetone + NaBH4 --> 2-propanol3-Bromocyclohexene is a secondary halide, and benzyl bromide is a primary halide. Both halides undergo SN1 substitution about as fast as most tertiary halides. Use resonance structures to explain this enhanced reactivity.Rank the primary, secondary, tertiary, and aryl halides in terms of reactivity with the nucleophile. Which type of halide react faster by an SN2 reaction? Explain

