13. In reference to Table 1 on page 19, what functional groups are always found in amino acids? AMine group TAILRO Droteins called polypeptides? Functional Structure Group Description Example found in alcohols R-OH Hydroxyl e.g., ethanol (beer) нн H-C-C-OH double-bonded oxygen on exterior car- bon Carbonyl Aldehyde e.g., formaldehyde (preservative) 11 R-C-H H. double-bonded oxygen on interior car- bon R-C-R H. Carbonyl (Molecules must contain at least three carbons to contain a ketone) H-C-C-C-H Ketone НО О ОН e.g., acetone; found in fingernail polish remover found in many organic acids; contains a hydroxyl group e.g., formic acid, smallest organic acid; makes fire ant bites hurt 3D0 R-C Carboxyl Он HO. found in amino acids, which are used to make protein e.g., the amino acid glycine Н O=c-C-N H- R-N H. Amine Но molecules that contain -SH e.g., ethanethiol H-C-C-SH Sulfhydryl R-SH нн common to nucleic acids; contains a hydroxyl group e.g., glycerol phosphate OH OH H H-C-C-C-0-P-O Phosphate R-0-P-O ОН molecules with side groups of -CH,, the only nonpolar and hydrophobic functional H. C-H Methyl R-C-H group HICIH O=P
13. In reference to Table 1 on page 19, what functional groups are always found in amino acids? AMine group TAILRO Droteins called polypeptides? Functional Structure Group Description Example found in alcohols R-OH Hydroxyl e.g., ethanol (beer) нн H-C-C-OH double-bonded oxygen on exterior car- bon Carbonyl Aldehyde e.g., formaldehyde (preservative) 11 R-C-H H. double-bonded oxygen on interior car- bon R-C-R H. Carbonyl (Molecules must contain at least three carbons to contain a ketone) H-C-C-C-H Ketone НО О ОН e.g., acetone; found in fingernail polish remover found in many organic acids; contains a hydroxyl group e.g., formic acid, smallest organic acid; makes fire ant bites hurt 3D0 R-C Carboxyl Он HO. found in amino acids, which are used to make protein e.g., the amino acid glycine Н O=c-C-N H- R-N H. Amine Но molecules that contain -SH e.g., ethanethiol H-C-C-SH Sulfhydryl R-SH нн common to nucleic acids; contains a hydroxyl group e.g., glycerol phosphate OH OH H H-C-C-C-0-P-O Phosphate R-0-P-O ОН molecules with side groups of -CH,, the only nonpolar and hydrophobic functional H. C-H Methyl R-C-H group HICIH O=P
Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter20: Organic Chemistry
Section: Chapter Questions
Problem 80QAP
Related questions
Question
100%
Can you help me answer this? (This is not for a grade but, rather, an exercise to facilitate acquisition).

Transcribed Image Text:13.
In reference to Table 1 on page 19, what functional groups are always found in amino acids?
AMine group
TAILRO Droteins called polypeptides?
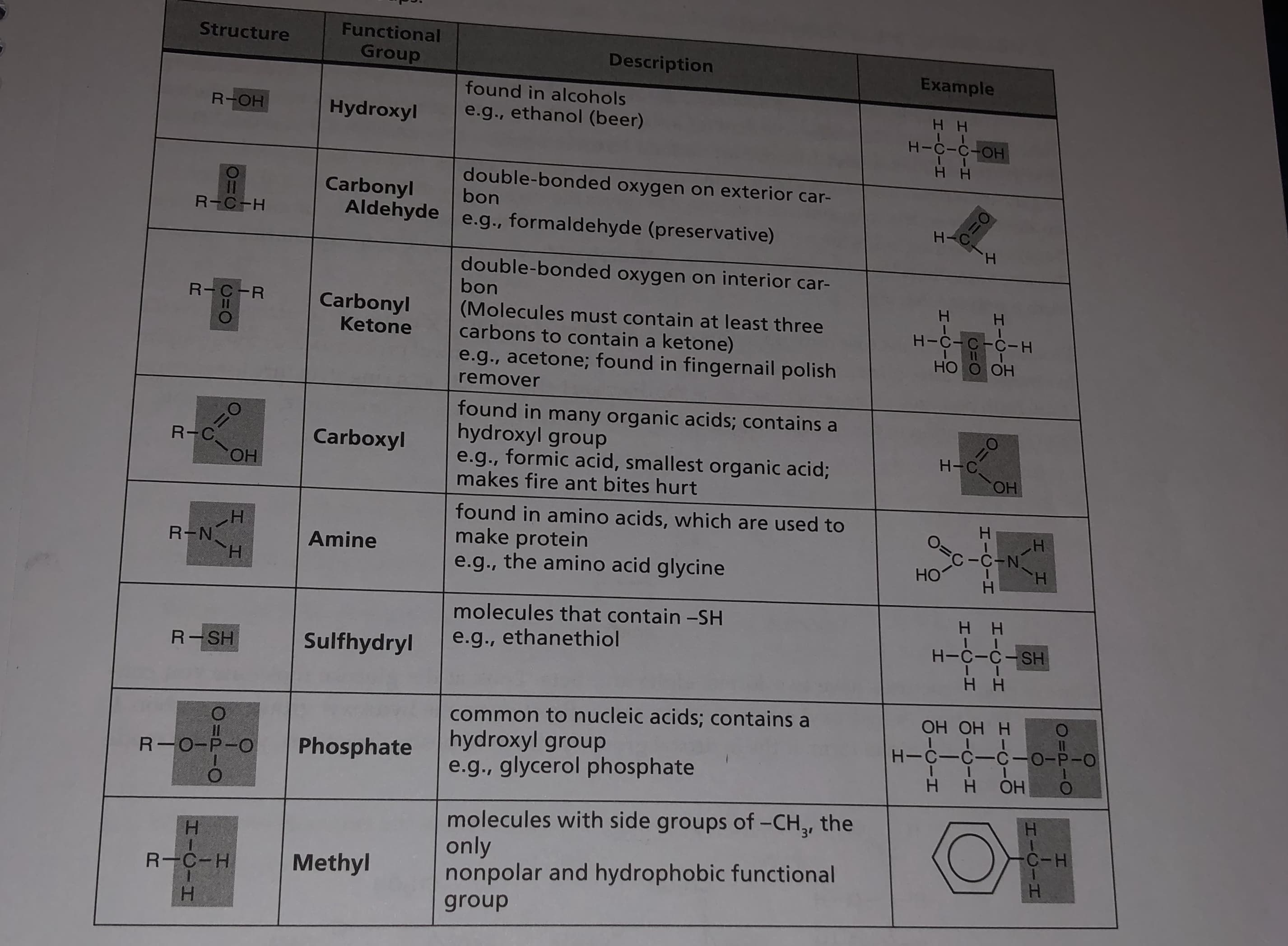
Transcribed Image Text:Functional
Structure
Group
Description
Example
found in alcohols
R-OH
Hydroxyl
e.g., ethanol (beer)
нн
H-C-C-OH
double-bonded oxygen on exterior car-
bon
Carbonyl
Aldehyde e.g., formaldehyde (preservative)
11
R-C-H
H.
double-bonded oxygen on interior car-
bon
R-C-R
H.
Carbonyl
(Molecules must contain at least three
carbons to contain a ketone)
H-C-C-C-H
Ketone
НО О ОН
e.g., acetone; found in fingernail polish
remover
found in many organic acids; contains a
hydroxyl group
e.g., formic acid, smallest organic acid;
makes fire ant bites hurt
3D0
R-C
Carboxyl
Он
HO.
found in amino acids, which are used to
make protein
e.g., the amino acid glycine
Н
O=c-C-N
H-
R-N
H.
Amine
Но
molecules that contain -SH
e.g., ethanethiol
H-C-C-SH
Sulfhydryl
R-SH
нн
common to nucleic acids; contains a
hydroxyl group
e.g., glycerol phosphate
OH OH H
H-C-C-C-0-P-O
Phosphate
R-0-P-O
ОН
molecules with side groups of -CH,, the
only
nonpolar and hydrophobic functional
H.
C-H
Methyl
R-C-H
group
HICIH
O=P
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

World of Chemistry
Chemistry
ISBN:
9780618562763
Author:
Steven S. Zumdahl
Publisher:
Houghton Mifflin College Div