13. Under base – catalysed conditions, two molecules of acetone can condense to - form diacetone alcohol. At room temperature (25°C), about 5% of the acetone is converted to diacetone alcohol. Determine the value of AG° for this reaction. OH HO. acetone diacetone alcohol
13. Under base – catalysed conditions, two molecules of acetone can condense to - form diacetone alcohol. At room temperature (25°C), about 5% of the acetone is converted to diacetone alcohol. Determine the value of AG° for this reaction. OH HO. acetone diacetone alcohol
Living By Chemistry: First Edition Textbook
1st Edition
ISBN:9781559539418
Author:Angelica Stacy
Publisher:Angelica Stacy
ChapterU6: Showtime: Reversible Reactions And Chemical Equilibrium
SectionU6.2: How Backward: Reversible Reactions
Problem 5E
Related questions
Question
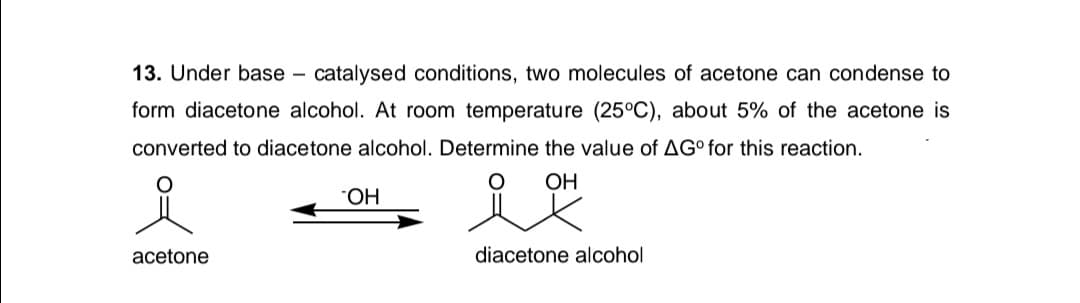
Transcribed Image Text:13. Under base – catalysed conditions, two molecules of acetone can condense to
form diacetone alcohol. At room temperature (25°C), about 5% of the acetone is
converted to diacetone alcohol. Determine the value of AG° for this reaction.
OH
НО.
acetone
diacetone alcohol
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:
9781559539418
Author:
Angelica Stacy
Publisher:
MAC HIGHER

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:
9781559539418
Author:
Angelica Stacy
Publisher:
MAC HIGHER