Q: Which one of the following sets of quantum numbers is not possible? m, 1 n 4 +/2 3. -2 1 -1/2 C 3 +/...
A:
Q: ОН ОН H H H HO HO HO Estradiol Cholesterol Estraiol Estrone
A:
Q: 8) What is the pH of a 0.035 M HNO3 solution? HNO3 (aq) + H,O (1)
A:
Q: Select which species below can undergo B-elimination. [RhPh(C3H7)2(PME3)4] O [RhH(C=CH)2(PME3)4] [Sc...
A: The possibility of the beta elimination reaction in the above mentioned metal complexes is heavily d...
Q: For the molecule whose condensed structural formula is given below, decide if cis-trans isomers are ...
A:
Q: 1. How does a mixture of a weak acid and its conjugate base help buffer a solution against pH change...
A: Buffer solution is the solution which resist the change in pH on dilution or with addition of small...
Q: 2) Identify the conjugate base and conjugate acid of each of the following. Conjugate Base Atom/ion ...
A: Compounds can be classified as acid, base or neutral compounds. Various theories have been given to ...
Q: Based on question 2 and 3 what combination of temperature and vinegar concentration would best achie...
A: An acid is a chemical substance that can enhance the hydrogen ion [H+] ion concentration in an aqueo...
Q: What happens when the area In whlch you llve tilts away from the sun? It's summer In your area. It's...
A: Given What happens when the area which you live tilts away from the sun
Q: An acid, HEl, is formed from a hypothetical element, El, and a subsequent aqueous solution is create...
A: Given, Mass percentage of acid, HEI = 20.09% Atomic Mass: H: 1.00784 g/mol O: 15.999 g/mol El: 142....
Q: 10. Which of the following subs CBr CBr F CBr,F2 CBRF CF 11. Which of the following liq CH;OCH; CH,C...
A: CF4 has lowest boiling point Explanation: The higher the intermolecular forces, the more the boiling...
Q: Which of the following nuclear reactions is INCORRECTLY categorized? Select one: O a. H + H → He + ¿...
A: Option ( C) is incorrectly categorized.
Q: Suppose that a solution made from an unknown solute dissolved in solvent X. Its molality is 0.0125 a...
A:
Q: 4. Consider a 150.0 g sample of nitrogen trifluoride (NF3). Show your work for each of the calculati...
A: Given: The mass of NF3 = 150 g. The molar mass of NF3 = 71 g/mol.
Q: Which names are correct for 102 and HIO2, respectively? A lodate and hypo iodous acid B lodite and i...
A: The correct name of given formula is given below.
Q: If I have 21 liters held at a pressure of 78 atm and a temperature of 900 k, what will be the volume...
A:
Q: -ది CH3 %3D C. H H3C C A 1. an enolizable carbonyl compound Answer: 2. an oxidation product of metha...
A: Enolization is a process in which an aldehyde or ketone toutomerizes to enol ( alkene + alcohol ) or...
Q: OH НО СНО Myrcene Citral Citral Lavandulol (±) - Linalool HO Menthol Limonene camphor pinane carvone...
A: When four different groups/atoms are attached to a particular centre , then it is called chiral cent...
Q: A solution is formed by volatile liquids A and B. The attractive forces between A & A, between B & B...
A: delta H( change in enthalpy): delta H is positive for bond breaking and negative for bond making. de...
Q: 7) What is the [OH] in a solution with a pH of 6.84?
A:
Q: Draw the Lewis structure of ozone, O3. Describe why ozone has a bent shape instead of a linear shape...
A: Lewis structure represents the chemical bonds and valency shell electrons of each and every atom of ...
Q: Determine the AH of the reaction 1 2NO29) +502cg) N205(s) 3 N205(s) → 2NO(9) + +502(g) O2(9) AH = 22...
A: Given: Reaction (1): N2O5(s) ---> 2NO(g) + (3/2)O2(g) ∆H1 = 223.7 kJ Reaction (2): NO(g)...
Q: Assuming the forward reactions are favored in the chemical equations below, arrange the acids accord...
A:
Q: S-1 Which is not the unit for the gas constant R? A. cal K1 mol 1 B. J K-1 mol1 C. atm L K mol 1 D. ...
A:
Q: A patient is prescribed Nexium as an I.V. infusion in 5.00% dextrose solution. The dose to be admini...
A: Answer~
Q: A 0.396 molal glucose solution has density of 1.16 g/mL. What is the molarity of glucose in this sol...
A: Molality of glucose, m = 0.396 molal Density of glucose, d = 1.16 g/mL Molarity of glucose, M = ? ...
Q: 7) What is the [OH] in a solution with a pH of 6.84?
A:
Q: 1. Based on the results of the solubility tests and chemical tests, what is a possible structure of ...
A: Hydrocarbon is a chemical species that contains only carbon and hydrogen atoms as their key constitu...
Q: Choose the reactions below where K and K, are equal. 302 (g) = 203 (g) CO (g) + H2O (g) = CO2 (9) + ...
A: Kp and Kc are the same for those equilibrium reactions where the difference between the gaseous mole...
Q: 1. Ptotal = 6 MPAX1 = 0.6 exerts greater partial pressure than Ptotal = 5.3 MPAX2 = 0.5 2. P total =...
A: Given: 1. When Ptotal = 6 MPa then X1 = 0.6 When Ptotal = 5.3 MPa then X2= 0.5 2. When Ptotal =...
Q: (i) Calculate the mass of NaHCO3 (84.007 g mol ') must be added to 4.00 g of K2CO3 (138.206 g mol1) ...
A:
Q: ION EXTRACELLULAR FLUID (mM) IN TRACELLULAR FLUID (mM) Eion AT 37 C K* 5 mM (normal range: 3.5-5) 10...
A: Given, Ion Extra cellular Fluid (mM) Intra cellular Fluid (mM) ...
Q: An ideal gas originally at 0.85 atm and 62°C was allowed to expand to become its inal volume, pressu...
A:
Q: Atom or lon of Element Species A Species B Species C Number of Electrons 36 18 Number of Protons 35 ...
A:
Q: ) Given the following reaction what is the expression for K? 2SO2 (g) + O2 (9) = 2S03 (g)
A: Given: The reaction is as follows: 2SO2(g) + O2(g) ⇌ 2SO3(g).
Q: A student planned to utilize HPLC to separate her chemically synthesized product (1) from th reactan...
A: HPLC also known as high-pressure liquid chromatography, is a separation technique used to separate a...
Q: Identify the correct net ionic equation for the reaction that occurs when solutions of HI and Be(OH)...
A:
Q: Consider the following reaction:
A:
Q: A rhodium metal centre undergoes cyclometallation with two units of C=C, what is the expected produc...
A: Correct choice is option ( A) 5 membered rhodacycle ring is formed.
Q: Consider the compounds and ions with curved arrows drawn below. When the curved arrows give a secon ...
A: Answer - Resonance structure - Resonance structures are sets of Lewis structures that describe the d...
Q: 2. Avogadro does not "waste" his time drawing a Lewis structure before determining the shape of PF3....
A:
Q: Benzene (i), toluene (ii), acetonitrile(iii), and methane(iv) are among the chemical wastes discover...
A:
Q: Figure 1 shows the crystal structure of sodium chloride salt (NaCI). Let the effective radius of Na ...
A:
Q: A student was given an unknown solution monoprotic acid to analyze. The student decided to use poten...
A: This is a titration method to determine the concentration of an unknown acid. The primary concept be...
Q: Glycophospholipids- its classification and role in the body. Give examples of it and draw a structur...
A:
Q: HNO3(aq) + Na2CO3(aq) > NaNO3(aq) + CO2(g) + H2O(l) does this reaction demonstrate oxidation - reac...
A: We need to find the oxidation state of atoms of both reactant and product side. Then we can say whet...
Q: In the synthesis of Sudan-1, the following precursors were used: Aniline – 0.2 mL β-naphthol – 0.3...
A: Sudan-1 dye is prepared by the conversion of 1 mole of aniline to 1 mol benzene diazonium chloride i...
Q: Kindly xplain the solubility of alkanes, alkenes, and alkynes to H2SO4.
A: Solubility of alkane alkene alkyne with H2SO4 depend upon the reaction of these molecule with H2...
Q: Identify each of the following reactions as endothermic or exothermic Exothermic Or Sign Reaction En...
A: It is given that whether the reaction is endothermic or exothermic .
Q: Given the following equation: 2 C3H8 + 9 O2 à 4 CO2 + 2 CO + H2O + Heat If we have 14.8g of propane,...
A:
For each write the correct IUPAC name
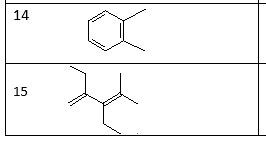
Step by step
Solved in 2 steps


