Q: Consider the following equilibria: 28(g) + C(g) →→→ 2A(g) 2D(g) 2E(x) + C(K) Calculate the…
A: Since,Equilibrium constant or Kc is the ratio of the equilibrium concentrations of product over…
Q: the reaction of (see image) is found to have an equilibrium constant of 1.1 x 10^8 at a particular…
A:
Q: Concentration (grams/kg H₂O) 1000 23. 900 800 700 600 500 400 300 200 100 0 Solubility Curve for…
A: We have to answer the following questions from the given temperature dependent solubility curve.
Q: Compound X, C42H72BrF, reacts with excess H₂/Pd to give a C42H78BrF compound. How many rings are in…
A:
Q: Which atom below is sp2 hybridized?
A: During orbital hybridization, the mixing of atomic orbitals takes place to form new orbitals known…
Q: Give the starting material. (a) (b) a Ob C Br (c) Br 0⁰ (d) Mg MgBr MacBook Air
A: Alkyl halide reacts with metal(Mg) in presence of dry ether to give Grignard Reagent.
Q: (2Z,4Z,6Z)-2-chloro-7-methoxyocta-2,4,6-triene (2Z,6Z)-2-chloro-7-methoxyocta-2,6-diene-4-yne Draw…
A:
Q: 25. The mass of one molecule of a molecular compound with a molar mass of 176.3 g/mol is A) 2.928 ×…
A: 25) Given, molar mass of the compound = 176.3 g/mol mass of 1 molecule of a compound = ? Note: 1 mol…
Q: Choose all that apply. Incorrect answers count against you. A primary alcohol can be oxidized to a…
A: The information given is A primary alcohol can be oxidized to a carboxylic acid by
Q: Fill in the correct term for each of the following Here are the possible answers: Wax, Fats, Oils,…
A: Here, some statements about fats, lipids and protein are given with blanks. We need to fill in the…
Q: Question 9 AC 2.5 (1) The halogens can all act as oxidising agents. What is an oxidising agent? (ii)…
A: Oxidising agent, reducing agent ; these are the terms from redox reaction.
Q: When cobalt (II) chloride reacts with sodium hydroxide, the resulting products are cobalt (II)…
A: For the calculation of the mass of the reactant we use the following steps: Balance the given…
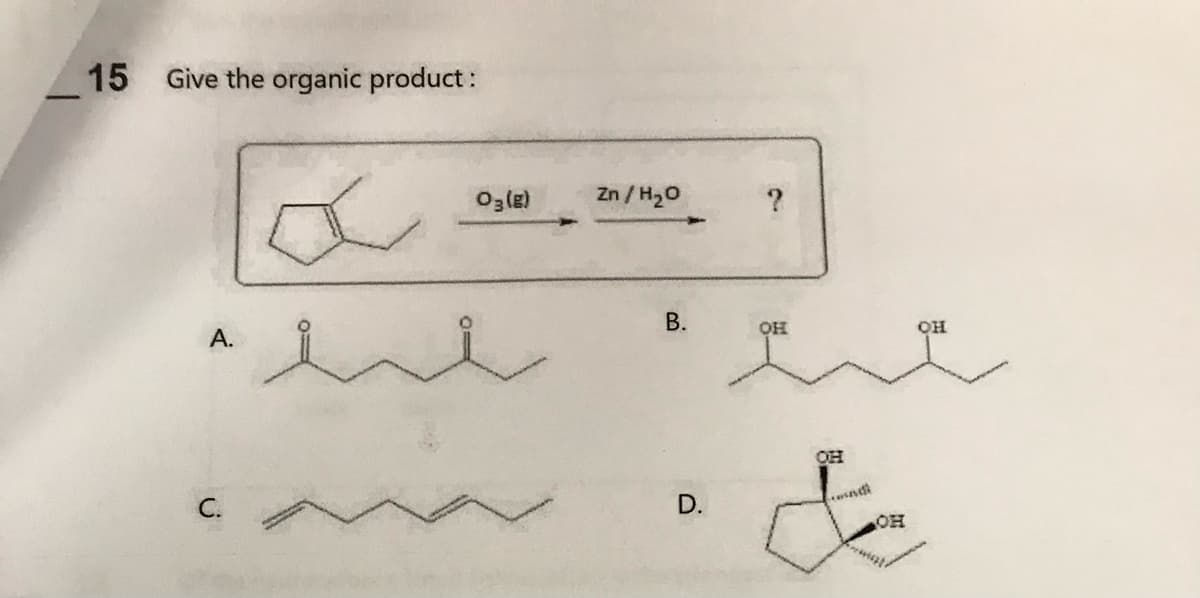
Q: Name the following compound. CH₂CH3 2-ethyl-1-cyclopentene 3-ethylcyclopentene ethylcyclopentene…
A:
Q: remaining Identify the diene used in the reaction shown below. H₂C- CN CH₂ + diene heat 0 H₂C -0 CN…
A: Diels-Alder reaction: The Diels–Alder reaction is a pericyclic reaction (cycloaddition) between a…
Q: Which species is the oxidizing agent in the following reaction? 2XO₂ + 2OH- + 3Na,ZO, 2XO + 3Na₂ZO,…
A: Oxidizing agent: The chemical species which oxidize the other chemical species by accepting…
Q: How do I find out how many protons an atom has?
A: We have to find out the number of protons in an atom.
Q: Select all that apply. Wrong answers count against you. Which of the following synthetic strategies…
A: The ether synthesis is mainly done by two processes. 1. dehydration of alcohol in presence of…
Q: Given the equation A(g) B(g) +2C(g). At a particular temperature, K-1.4 x 10³. Placing the…
A: Answer:- This question is answered by using the simple concept of Le Chatelier principle which…
Q: 7. A catalyst increases the rate of a reaction because a) provides another reaction pathway with…
A: We have to answer the following question
Q: What is the net ionic equation for the reaction below? 2 FeCl3(aq) + 3 Ca(OH)2(aq) → 3…
A: Given -> 2FeCl3(aq) + 3Ca(OH)2(aq) ----> 3CaCl2(aq) + 2Fe(OH)3(s)
Q: 3. A non-flow system contains 1 pound-mass of Ideal gas (cp=0.24; cv = 0.17, both in BTU/lb- R) the…
A: The given data: mass of the ideal gas = 1 pound, that is 1 lbm Cp = 0.24 Btu/lbm.R Cv = 0.17…
Q: The 121 howitzer refers to the bore size of the weapon in mm. Thus, the "121" has a 121 mm bore…
A: Given: Bore size = 121 mm and bore size in caliber = bore size in inches *100 since 1 in = 2.54 cm…
Q: -23 An oxygen atom has a mass of 2.66 x 10 g and a glass of water has a mass of 0.050 kg. Use this…
A:
Q: In the question, 'which set of three quantum numbers does not specify an orbital in the hydrogen…
A: A question based on atoms. Four choices relevant to quantum numbers of Hydrogen atom is prescribed,…
Q: Sample containing arsenic weighing 0.800 are titrated with a standard iodine soluton. what should be…
A: To solve this problem, you need to first determine the molar mass of arsenic trioxide (As2O3). The…
Q: NBS hv, CCl4 (a) (b) Br Br Br d (c) (d) Br (e)
A: We have to give the major product of the given reaction Introduction: Allylic bromination.
Q: How many signals would you expect in the ¹H NMR spectrum of this compound? Br Br
A: Numbers of signals is equal to chemically non- equivalent proton.
Q: a citrate buffer of pH 4.5, if you have 500.0 mLs of 0.750 M citric acid (Kay-7/ M NaOH, and…
A:
Q: for the galvanic cell at 298 K Zn(s) + 2In2+(aq) Zn2+(aq) + 2In+(aq) Ecell = 0.36V what is the…
A: Equilibrium constant(Kc) and Ecell are related as Ecell=0.0592nlogKc at 298KOn rearranging the…
Q: Consider the ZERO-ORDER reaction: A —> product. (a) Write the rate law for the reaction. (b) What…
A:
Q: Identify an unknown compound with the following mass spectrum data: M* at m/z of 106, intensity of…
A: Given, The molecular ion peak(M+) = 106 m/z with intensity = 82 The M+1 peak= 107 m/z with intensity…
Q: Industrial reactions are mostly carried out under standard-state conditions. it is also possible…
A: Since you have asked multiple questions, we will solve the first question for you. For the remaining…
Q: Previous Page 3 of 3 What main type of intermolecular forces must be overcome in converting each of…
A: A) In the case of bromine (Br₂), the main intermolecular force that must be overcome is the London…
Q: 8.31c x Your answer is incorrect. KM-O, NOH Cold Modify the given carbon skeleton to draw the major…
A: Organic reactions are those in which organic reactant react to form organic products. In the given…
Q: Given the two reactions 3. PbCl₂ (s) 4. AgCl(s) Pb²+ (aq) + 2Cl(aq), K3 = 1.85x10-10, and Ag+ (aq) +…
A: Characteristics of equilibrium constant are : - If the equation (having equilibrium constant K) is…
Q: Question 2 Give the starting material. (a) (b) O a (c) (d) 1. Hg(OAc)₂, H₂O 2. NaBH4 OH MacBook Air
A:
Q: 7 Give the IUPAC name for the following compound. LOH A. 3-methyl-1-pentenyl alcohol B.…
A: 7) IUPAC nomenclature: In IUPAC nomenclature a molecule's longest chain of carbons is taken as…
Q: Balance the Redox Reaction given below: MnSO4 + NaBiO3 + H2SO4 ⟶ NaMnO4 + Bi2(SO4)3 + H2O + Na2SO4
A:
Q: b) e) 2 d) ethylbenzene the correct reagents for these transformations: nitrobenzene acetic acid…
A: A reagent is a substance that is added to a reaction mixture to bring the chemical reaction.
Q: -, Compound X, C46H75BrO4, reacts with excess H2/Pd to give C46H79BrO4. Compound X has rings and…
A: Index of hydrogen deficiency = It is the measure of degree of unsaturation of a compound 1 IHD…
Q: the drawings Include any stereochemical terms for any stereochemistry indicated on the structure
A: Solution: Cyclohexane substituted compound can be made chiral by substituting at least one H with…
Q: What is the total combined volume of all the products if 30 L of propane gas, C3H8(g), is…
A: To find out the combined value, first write the balanced reaction for a combustion of propane gas.…
Q: What is the approximate Rf value of the spot in lane A of the TLC plate below? ● ● -F-- A B
A: The Rf value of a compound is equal to the distance traveled by the compound divided by the distance…
Q: A steel rod having a cross-sectional area of 300 mm² and a length of 150 m is suspended vertically…
A: To calculate the total elongation of a rod under a tensile load, we can use the formula: ΔL = PL /…
Q: formic acid HCOOH is a weak acid with Ka equal to 1.8 x10^-4 what is the pH of a 0.0115 M aqueous…
A:
Q: Given the following unbalanced redox reaction: Fe2+ + MnO4- → Fe3+ + Mn2+ Which is the oxidizing…
A: The addition of electrons or decreases in the oxidation state is known as a reduction reaction and…
Q: If a mixture of Glu (pI=3.22) and Arg (pI=10.76) is passed through a cation exchange column at…
A: Given that, The isoelectric point of Glu is 3.22. And the isoelectric point of Arg is 10.76. We…
Q: The combustion of propane, C3H5, is found to have a heat of combustion of -332 0 kJ/mol of propane…
A: The water has to be heated from 15.00oC to 90.00oC . Change in temperature needed is ∆T…
Q: Q1 (Conformation & Structure) a) When the molecule below is placed in the most stable chair…
A: Given that, a compound is We have to tell how many hydrogens are at axial position on the most…
Q: what are the bronsted lowry bases in this reaction?
A:
Step by step
Solved in 2 steps with 1 images

- For 20.7 the solutions for part c and d dont seem right. Shouldn't reacting with 1 eq of H2 just take away the C=C bond and not the C=O bond. (H2 reduces C=C selectively to form a ketone according to page 738. Also the both the C=C and C=O bond should be reduced if excess H2 is usedIn extraction of caffeine from tea, CaCO3 (pKa of HCO3- is 10.3) is used to convert tannin to glucose and calcium salt of gallic acid. Explain why glucose is not in its salt form but gallic acid is. Assuming pKa of -OH of glucose is 12, and pKa of -OH of gallic acid is 9.5 and pKa of -COOH of gallic acid is 4.5.The data below give the concentration of thiol (mM) in the blood lysate of the blood of two groupsof volunteers, the first group being ‘normal’ and the second having rheumatoid arthritis (Banford, J.C.,Brown, D.H., McConnell, A.A., McNeil, C.J., Smith, W.E., Hazelton, R.A. and Sturrock, R.D., 1983,Analyst, 107: 195): Normal: 1.84, 1.92, 1.94, 1.92, 1.85, 1.91, 2.07Rheumatoid: 2.81, 4.06, 3.62, 3.27, 3.27, 3.76 Verify that 2.07 is not an outlier for the ‘normal’ group.
- Acetylene is a very weak acid; however, it will react with moist silver(I) oxide and form water and a compound composed of silver and carbon. Addition of a solution of HCl to a 0.2352-g sample of the compound of silver and carbon produced acetylene and 0.2822 g of AgCl. (a) What is the empirical formula of the compound of silver and carbon? (b) The production of acetylene on addition of HCl to the compound of silver and carbon suggests that the carbon is present as the acetylide ion, C22-. Write the formula of the compound showing the acetyl ide ion.Relationships among E° cellv G°, and KWhen BF3 behaves as a Lewis acid, the stability of the adducts with (CH3)2O, (CH3)2S, and (CH3)2Se decreases in the order in which the Lewis bases are listed. However, when the Lewis acid is B(CH3)3, the most stable adduct is that formed with (CH3)2S and the adducts with (CH3)2Se ad (CH3)2O are about equally stable. Explain the difference in stability of the adducts
- Benzoic acid (C6H5C02H) has Ka =6.3 x 10-5 a) What is the value of Kb for the benzoate ion (C6H5C02-)? b) What is the h30+ concentration of a O, 10 mol/dm3 solution of benzoic acid? c) What is the pH of the solution in (b) above?Sodium hypochlorite (NaOCl, the oxidant in household bleach) in aqueous CH3COOH is also touted as a “green” oxidizing agent. For example, oxidation of (CH3)2CHOH with NaOCl forms (CH3)2C = O along with NaCl and H2O. (a) What advantages and/or disadvantages does this method have over oxidation with HCrO4- –Amberlyst A-26 resin? (b) What advantages and/or disadvantages does this method have over oxidation with CrO3, H2SO4, H2O?outline a separation scheme for isolating pure aspirin. lab video: https://www.youtube.com/watch?v=Y4NMpO1xI8U
- Compute for the appropriate amount (mL) of the compostions below of CTAB precitpitation solution used for DNA extraction. a. 1% w/v CTAB b. 10 mM EDTA c. 50 mM Tris-ClA 0.9056 g sample of KBrO3 (MM=167.0) was dissolved in dilute HCl and treated with an unmeasured excess of KI. The liberated iodine required 37.21 mL of Na2S2O3. Calculate the molar concentration of sodium thiosulfate. 1 mol BrO3- = 3 mol I2 = 6 mol S2O32- 0.8744 M 2.623 M 0.8791 M 0.2915 Ma. For this acid-base reaction: C⊖H3 + H3O+ −−→ CH4 + H2O, the conjugate acid of carboanion (C⊖H3) is _________________ b. Complete the equation: HC≡N + NaOH −−→ ___________+__________ c. Rank the acidity of these: NH3, HF, H2O, HCl, NaH Most acidic______>_______>_______>_______>_______least acidic d. The Ka of acetic acid (found in vinega) is 1.8×10-5. Then the pKa of formic acid is







