Q: Calculate the equivalence point of a titration of 0.3326 g of THAM (pKa of conjugate base is 8.08)…
A: The objective is to determine the pH at the equivalence point for the neutralization titration of…
Q: Write down the common (not IUPAC) names of the organic molecules that would be released if this…
A: Given question is related to identifying the products of the given compound (triglyceride )…
Q: Two mechanisms are among those that have been postulated for decomposition of aryl diazonium salts…
A: The objective of the question is to understand how kinetic studies can be used to determine the…
Q: Write de complete (not UIPAC) name of each molecule below. Note: If a molecule is one of a pair of…
A: Orgnaic compounds having two functional groups namely, amine and carboxylic acids are called amino…
Q: С H H30* H нео (a)
A:
Q: Draw all five resonance structures of the intermediate of the following reaction. CI :O: HOỘ: :O:…
A: Given a reaction, draw the resonance structures of intermediate.
Q: What is the product of the following reaction? 2 CH₂CH₂CH₂ OH OH CH3CH₂CH₂CHCH CH3CH₂CHCH OH H…
A: aldol condensation is a condensation reaction in organic chemistry in which two carbonyl moieties…
Q: Draw the products of the two step reaction sequence shown below. Ignore inorganic byproducts. If the…
A:
Q: A precious metal has an enthalpy of sublimation of 1640 J/mol and an enthalpy of fusion of 64 J/mol.…
A: The heat released when a substance changes from the gaseous state to the liquid state is the…
Q: Silicon reacts with gaseous fluorine to form a fluoride. Type in the formula of the silicon fluoride…
A:
Q: 14. Which compound will be most soluble in water? (A) CH-CH₂-CH₂-CH₂-CH₂-OH (B) CH-CH₂-CH₂-CH₂-COOH
A: Carboxylic acids are more soluble in water than alcohol having ame molar mass because acid form…
Q: Use the balanced equation, 2NO+ O₂ 1 2 NO₂, to answer the following questions. Part 1 of 3 Determine…
A:
Q: e melting point of copper metal is 1083 °C. The vapor pressure of solid copper is reported to be…
A: At given temperature (1060 °C + 273 = 1333 K) and pressure (3.56 × 10-7 atm), the number of copper…
Q: 1) Predict the major product for the reaction shown. (CH3)2CHCH₂Cl AICI 3 ODE
A: Alkyl halide in presence of Lewis acid give electrophilic aromatic substitution reaction on benzene…
Q: The product(s) from CH₂CH=CHCO₂CH₂CH₂ C E CH₂CH=CHCOH CH₂CH=CHCH₂OH + CH₂CH₂OH + CH₂CH₂OH…
A: We have given the organic reaction and we have to find out the product of the reaction. the given…
Q: Predict the relative boiling points for the following molecules, putting the highest at the top of…
A: As the intramolecular forces increase, the boiling point also increases.
Q: hich of the following is the correct wedge and dash conformation for the following Newman…
A:
Q: جد OEt 1. NaOEt 2. 0 3. HO H
A: This is an example of Michael addition
Q: The atmospheric pressure at the top of Mount Everest (29035 feet) is about 256 mmHg. Convert this…
A: To convert from one unit to another unit, a conversion factor has to be used. The unit to which it…
Q: Dichloroacetic acid has a dissociation constant of K₁3.32 x 10-². Part A Calculate the degree of…
A: Given,Concentration=0.0625 m
Q: The proton with the lowest delta value (the most shielded) is The proton with the highest delta…
A: The NMR spectroscopy is an abbreviated form of Nuclear Magnetic Resonance Spectroscopy. Each kind of…
Q: Suppose a 500. mL flask is filled with 0.30 mol of Br₂, 1.2 mol of OCI, and 0.60 mol of BrCl. The…
A: The objective is the computation of equilibrium molarity of Br2, i.e., [Br2]. Equilibrium molarity…
Q: Classify each chemical reaction: Reaction Cl₂(g) + 2KI (aq) → 2KCl(aq) + 1₂ (s) H₂SO₂ (aq) H₂O(l) +…
A: The objective of the question is to classify each given chemical reaction into one of the main types…
Q: 5. C7H100 %T 9 HR200500646NS 100 80 60 40 20 2000 8 1 1:1 7 لار 3000 6 5 : ppm 2000 4 Waussu بلد 3…
A: The question is based on the concept of organic spectroscopy. We need to analyse the spectral data…
Q: Draw the products of the two step reaction sequence shown below. Ignore inorganic byproducts. If the…
A: When the benzene reacts with an alkyl halide in the presence of a Lewis acid catalyst alkyl benzene…
Q: Ⓒ What is the major product of the following E2 reaction? ---82 LiTMP E2 ) و مالي مل مار مار
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: The critical temperature and critical pressure of a gas obeying van der Waal's equation are 30oC and…
A: Given that TC = 30°C = 303.15K PC = 73 atm b = ?
Q: X Incorrect. Modify the given copies of the starting material to draw the two condensation products.…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: Which explains why methoxyethene readily undergoes cationic polymerization? Choose one: O stabilizes…
A: A polymer is a large molecule that is formed by the repetition of small units called monomers, and…
Q: Calculate the amount of heat needed to melt 194. g of solid octane (CH₁) and bring it to a…
A:
Q: Determine the entropy change for the reaction given. Enter your answer in J/K. CO2(g) + 2NH3(g)…
A: Entropy of CO2(g) = 213.74 J/molKEntropy of H2O(l) = 69.91 J/molK Entropy of H2O (aq) = 188.83…
Q: b) û Show how each of the following could be synthesized starting from an alkene that contains only…
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: Draw Intermediate Aqueous workup Draw Product Aqueous workup Draw Product
A: KMnO₄ a oxidizing reagent it reacts with alkene and gives syn diol.
Q: =) Starting from compounds containing fewer than six C atoms, propose a synthesis of
A: Given reaction is an example of the Michael addition reaction In Michael addition reaction is the…
Q: Use the standard reduction potentials located in the 'Tables' linked above to calculate the standard…
A: For a electrochemical cell E⁰cell can be calculated by using the following formula: E⁰cell = E⁰red(…
Q: A voltaic cell is constructed from a standard Ni²+ |Ni half cell (E° red = -0.250V) and a standard…
A: The electrode which is having a higher reduction potential gets reduced at the cathode while the…
Q: 1. (a) HO -CH₂CH=CH₂ Draw the most stable chair conformation for the compound above.
A: The basic principle is; the bulky group should always come at equatorial position.The higher the…
Q: a) to c) show the formulas of rubidium oxides and these are repeated in d) to f). From the options…
A: Answer:
Q: Energy Identify the groups of orbitals in the molecular orbital energy level diagram of…
A: Note: Since you have posted multiple questions, we will provide the solution only to the first…
Q: Use the standard reduction potentials located in the 'Tables' linked above to calculate the standard…
A: In the given question solve by below steps.Decide oxidised and reduced elements and take the…
Q: Shown below is a galvanic cell with anode compartment b containing anode a and cathode compartment d…
A:
Q: Which molecule contains an atom with an unhybridized p orbital in its valence shell? (a) (b) (c) (d)…
A: Find the hybridissation of suspected element to know about participation of P orbital in…
Q: Considering this, watch the rotation of 2-methylbutane in 3-D and explain why the following…
A: Write the IUPAC name of both .
Q: For the reaction shown, which of the compounds listed below would be the expected major, and final,…
A: Alkynes on hydroboration-oxidation reaction, forms aldehydes or ketones as products. The product…
Q: The electron density solutions for the molecule H₂ in question 1 are different sizes. Why is that?
A: We have been given different orbital diagram and we have to find the correct answer for the given…
Q: A 50.0 mg sample of an unknown radioactive substance was placed in storage and its mass measured…
A: Radioactive decay is a first order reaction. Half-life ( t1/2 ) formula for first order reaction or…
Q: The image at right shows electron density solutions for the molecule H₂ in panels a-e and an energy…
A: We have been given the electron density with pictorial representation at different points.
Q: Give the major product for the following reaction. H&C H₂C CH3 CH3 HO, H₂O, A
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: 3.) A 100.0 mL portion of 0.250 M CaCl₂ is mixed with 150.0 mL of 1.00 × 10³ M NaF. Will a…
A: Solubility product quotient, Qsp is the product of the concentrations of the products, each raised…
Q: what is the pH of a 1.20 M solution of C2H5NH3I
A:
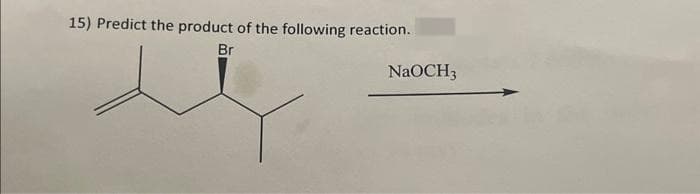
Step by step
Solved in 3 steps with 1 images


