Q: 27. Propose a mechanism for the following reaction. Use curved arrows!!! Explain each step. CH3 HCL…
A:
Q: Which enthalpy change is indicated by X in the enthalpy cycle shown? C(s) + 2H₂(g) + 20₂(g) AH ΔΗ…
A: The total enthalpy change for a chemical reaction is measured according to Hess's law. The summation…
Q: At 45.0 °C, a 14.0 L vessel is filled with 7.25 moles of Gas A and 8.45 moles of Gas B. What is the…
A: From ideal gas law equation P•V = n•R•T P = pressure of gas V = volume R = gad constant T =…
Q: Write down the common (not IUPAC) names of the organic molecules that would be released if this…
A: Triglycerides undergo hydrolysis to give glycerol and 3 fatty acids. Here the hydrolysis of ester…
Q: Ⓒ What is the major product of the following reaction sequence? 1 HBr, trace peroxides > Ⓡ+=N₂²Ⓡ Na…
A: Find out product of given organic transformation.
Q: Tamiflu, the most effective antiviral drug used to treat avian influenza, has the following…
A: Since you have posted multiple questions, we will provide the solution only to the first question as…
Q: CH₂ HCI 1) Li / éter 2) Cul 3) CH₂Cl H₂SO4
A: Given that, the reaction sequence is:We need to identify the product and explain its formation.
Q: Which of the following compounds, I, II, III, or IV do not have delocalized electrons, i.e. no…
A: Delocalisation of electrons occurs, when electrons are not associated with only one atom or bond,…
Q: 5. Using curved arrows please give the mechanism of this reaction, including any regioselectivity or…
A: The E1 mechanism (elimination unimolecular) is one of the two main pathways by which organic…
Q: 11. Using the Jahn-Teller theorem, predict which of the following should be distorted: Ti(H₂0)63+,…
A: In the intricate world of coordination chemistry, the behavior of transition metal complexes takes…
Q: A 6.00 L tank at 18.3 °C is filled with 4.33 g of carbon dioxide gas and 7.47 g of sulfur…
A:
Q: TRANSMITTANCE Spectrum G LOD TRANSHCTTANCEI 4000 Spectrum H 0.8 0.6 0.4 0.2 3000 Molecule 4 pump…
A: The question is based on the concept of organic spectroscopy. We need to analyse the spectral data…
Q: ECH CH3
A: Given are organic reactions. In reaction 1, reactant is alkyne.In reaction 2, reactant is alkene.
Q: Acidity: Complete the statement about the acidity/basicity of compounds due to aromaticity from the…
A: We will first draw all possible resonating structures that include the given anion. The more the…
Q: The boiling point elevation constant of water is Kb = 0.512°C/m. The maximum solubility of sucrose…
A:
Q: b) û Show how each of the following could be synthesized starting from an alkene that contains only…
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: 5.- Provide the product of the following reactions + Br + Mg 2 Li 1,4-Dioxane Na 1% mol Hexane
A: Alkyl halide reacts with metal Mg in ether solvent to produce alkyl magnesium halide. In the first…
Q: xes N N H CF3CO2H Q
A: This is deprotection reaction. While making peptide bonds, the amino group of an amino acid is…
Q: 3. Consider the two substitution reactions shown below (A and B): A B XOXO H₂ D Br H₂ D Br H O D a.…
A: => this is an nucleophilic substitution bimolecular reaction=> one step mechanism=>…
Q: 128 Identity of Oxalate Salt Solubility: mmciss OF C₂04² Solubility Product Constant Sr C₂O4 H₂0…
A: The question is based on the concept of solubility product principle.It states that when a weak…
Q: 18. What is the relationship between the following two compounds? (A) same compound (B) not isomers…
A: Organic molecules are the chemical compounds that consist of carbon and hydrogen, as well as other…
Q: 1. NaOH, Br₂ 2. H30* d. a. b. C. Br Br Br Br HO is to bly alf of Br- HO e.
A:
Q: will pair with Select one: by forming a. Guanine: cytosine: 3 O b. Thymine: cytosine: 2 Oc. Thymine:…
A: DNA is a polymer of nucleotides. A nucleotide is formed by three structural units:Nucleotide =…
Q: ((NH₂)₂CO) must be added to 495 g of water to give a solution with a vapor pressure 2.71 mmHg less…
A:
Q: МСРВА CH2N2, hv Br2, CC14
A: -> MCPBA (m-chloroperoxybenzoic acid) used for formation of epoxide when reacts with double…
Q: 1. For the reaction provided, the rate of disappearance of I (aq) is 2.4 x 10-3 mol/L.s. 103 (aq) +…
A: The rate of the formation of the product or the rate of disappearance of the reactants is known as…
Q: 53. Write correct mechanisms and specify the type of substitution or elimination reaction for the…
A: We have to predict the structure of the required products.
Q: Which of the following compounds does not have tetrahedral electron-pair geometry? NO₂™ ⒸH₂O O PC3…
A: From hybridization, we can determine the electron geometry.Formula for hybridization is:Where -V =…
Q: 5. Consider the 3 structures below (A, B, and C): "A"H "A"Br Br H H" Br A Homotopic Enantiotopic…
A: Constitutional isomer : Functional isomers are the constitutional isomers which share the same…
Q: Write the balanced net ionic equation for the reaction between K2CrO4 indicator and the AgNO3…
A:
Q: Hydrogen gas and nitrogen gas react form ammonia gas. what volume of ammonia would be produced by…
A: The given reaction is between hydrogen gas and nitrogen gas to give ammonia gas. The volume of…
Q: Predict the products of the following reaction. If no reaction will occur, use the NO REACTION…
A:
Q: Why will a gas at T=-100°C and P=4 atm exhibit less ideal behavior than a gas at T=+100°C and P=4…
A: Answer:Intermolecular attraction that is represented by van der walls coefficient aSize of the gas…
Q: 1. Complete the reactions. 1) H -CH3 Br₂ Acetic acid CH₂CH3 1.LDA 2.CH 3 CH ₂1 Pyridine Heat
A:
Q: What is the major product of the following reaction? дун он H 342 H 6) T چلو d) MCPBA? epoxic HH OH…
A:
Q: You have a solution of the three molecules below in a suitable organic solvent. NH₂ OH In addition…
A:
Q: Determine the absolute stereochemistry: O R. R S, S R. S S, R B A
A:
Q: K = 1.3 \times 10-10 for the reaction HC6H50(aq) =H+(aq) + C6H50-(aq). In which direction is the…
A: If equilibrium constant Kc is less than one, the equilibrium will favour the reactants.If Kc is…
Q: A. Starting from s-butyl alcohol: CHS, CH₂ 1.1,1-dimethylumicame 2. buta 3.2.2.3,3 intrundbybac
A: Reactions are the process by which a reactant is transformed into a product. During this…
Q: Give the major product for the following reaction. H&C. H3C CH3 OCH3 1. NaOCH3, CH3OH 2. H30¹, A
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: "rms' The root mean square velocity, expressed u is for a collection of particles and is directly…
A: The formula given is .The root mean square velocity is .The ideal gas constant is .The temperature…
Q: Acetic acid is a weak acid that ionizes in solution as follows: CH₂COOH(aq)CH₂COO (aq) + H+ (aq) If…
A: molality(m) = 0.106 mKf = 1.86oC/mn = 2 as acetic acid ionizes into acetate and hydrogen…
Q: The freezing point depression constant of water is Kf = 1.86°C/m. The maximum solubility of sucrose…
A:
Q: Complete the following reaction and write the IUPAC names of the two products. "NH₂ + H₂O + HCI heat
A: The question is based on organic reactions. we need to identify the product formed and explain…
Q: A compound with the molecular formula C4H8O2 gives a ¹H NMR spectrum with the following three…
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: A precious metal has an enthalpy of sublimation of 1640 J/mol and an enthalpy of fusion of 64 J/mol.…
A: The heat released when a substance changes from the gaseous state to the liquid state is the…
Q: C ?? D
A: This is an multi-step reaction
Q: Show your calculations for the Ksp of barium oxalate from the solubility you found in The CRC…
A: The question is based on the concept of solubility product principle.It states that when a weak…
Q: (14) which reaction sequence cen most efficiently bring about (N) the following transformation? CN H…
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: 11.- According to geometric isomerism, provide what isomer each molecule is: a) |||| b) FA FA
A: Constitutional isomers : Functional isomers are the constitutional isomers which share the same…
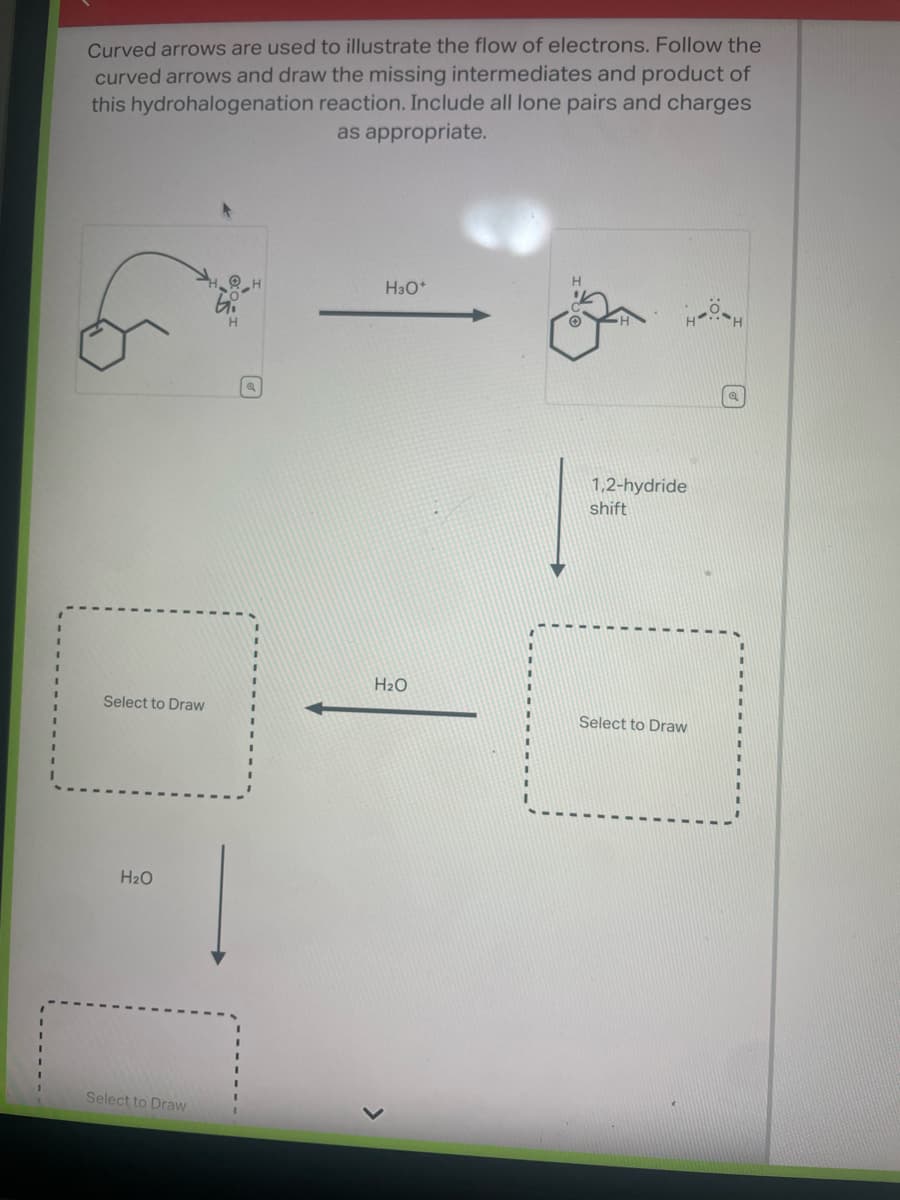
Step by step
Solved in 3 steps with 2 images

- The question is: "Draw the curved arrow mechanism for the reaction between (2S,3S)-3-methylhexan-2-ol and PCl3. Note the specific instructions for each box. Include nonzero formal charges and lone pairs of electrons on all appropriate atoms" I attached screenshots of the picture of the atoms below. What type of reaction (SN1, E1, SN2, or E2) would the reaction be, and how would these molecules interact? The question asks for multiple steps so I am guessing it is either SN1 or E1, but what is the reaction mechanism?for the Cl2 addition reaction with 1,2dimethylcyclohexene, draw the most reasonable curved arrow mecanism. based on the reaction products, explain why a carbocation intermediate cannot be proposedDraw the curved arrow mechanism for the reaction between (2R,3S)-3,5-dimethylhexan-2-ol and PCl3. Note the specific instructions for each box. Include nonzero formal charges and lone pairs of electrons on all appropriate atoms. Then answer the question about the mechanism.
- Chemistry please provide the flow of electrons aswell!! thank you! using the starting material (on the left) to determine the sythetic route which will be the most reaosnable and effective to theres none, you have to start witth the begin products to get to finish needed by using the minmium reagents and reactions needed to get to the final productwhy does this carbocation go through rearrangemnt by a hydride shift?Predict the product(s) of this reaction by interpreting the flow of electrons as indicated by the curved arrows.
- Please use curved arrows to show the electron movement, bond making and bond breaking in the concerted reaction below. Thank you!draw the potential products in the space below, for the halogenation (with heat/UW light) reaction:The quick rule which allows prediction of the major product for electrophilicaddition reactions to alkenes (such as shown in question 2) works mechanisticallybecause the electrophile adds to theC=C in the directionthat forms the (a) least stable carbocation, (b) most reactive nucleophile, (c) moststable carbocation,(d) lowest classification of halide.
- In the information provided,-see photo, we read that the axial attack is preferred for the reaction. Explain this preference for the axial attack and the trans product formedin Figure 1. Be sure to comment on the steric factors of the substituents, their placement onthe ring, and the hydride delivery agentDraw curved arrows to represent the flow of electron during the reaction for each of thefollowing reactions.Give a full curved arrow mechanism for the ring-opening reaction of an epoxide in aqueousacid shown below. Show ALL non-bonded electron pairs and all non-zero formal chargeswhere applicable. (Note: you will need to redraw the structures provided in order to showthe mechanism correctly; non-bonded electron pairs have not been shown, although nonzero formal charges are included.) Be sure to show any stereochemical elements in theproduct(s) correctly.

