15. How much water (in mL) must be added to 200 mL of a 1.25 M soldium chloride (NaCl) solution to decrease its concentration to 0.770 M? (answer; 125 mL)
15. How much water (in mL) must be added to 200 mL of a 1.25 M soldium chloride (NaCl) solution to decrease its concentration to 0.770 M? (answer; 125 mL)
Chapter7: Solutions And Colloids
Section: Chapter Questions
Problem 7.107E
Related questions
Question
100%
For question 15 I am certain I have the correct answer after using the V1C1=V2C2 formula but it’s not matching the key. Could you let me know if I’m correct or incorrect?
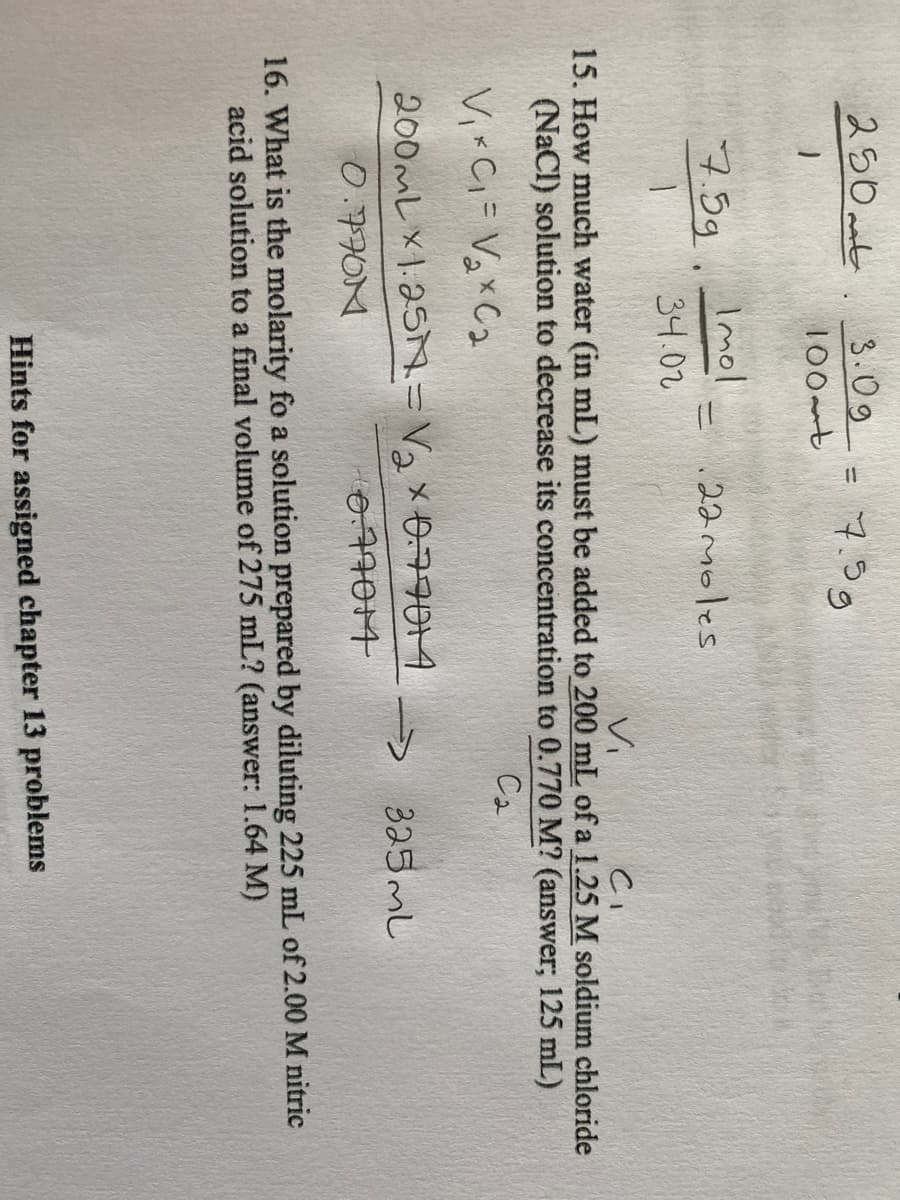
Transcribed Image Text:250 ant
3.09 - 7.39
%3D
100mt
Imol
7.5g.
22moles
34.02
Vi
15. How much water (in mL) must be added to 200 mL of a 1.25 M soldium chloride
(NaCl) solution to decrease its concentration to 0.770 M? (answer; 125 mL)
Ca
Vi Ci= Vgx C2
200mレ×1.25N-Va x679014
325ML
->
0.7770M
0,77014
16. What is the molarity fo a solution prepared by diluting 225 mL of 2.00 M nitric
acid solution to a final volume of 275 mL? (answer: 1.64 M)
Hints for assigned chapter 13 problems
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning