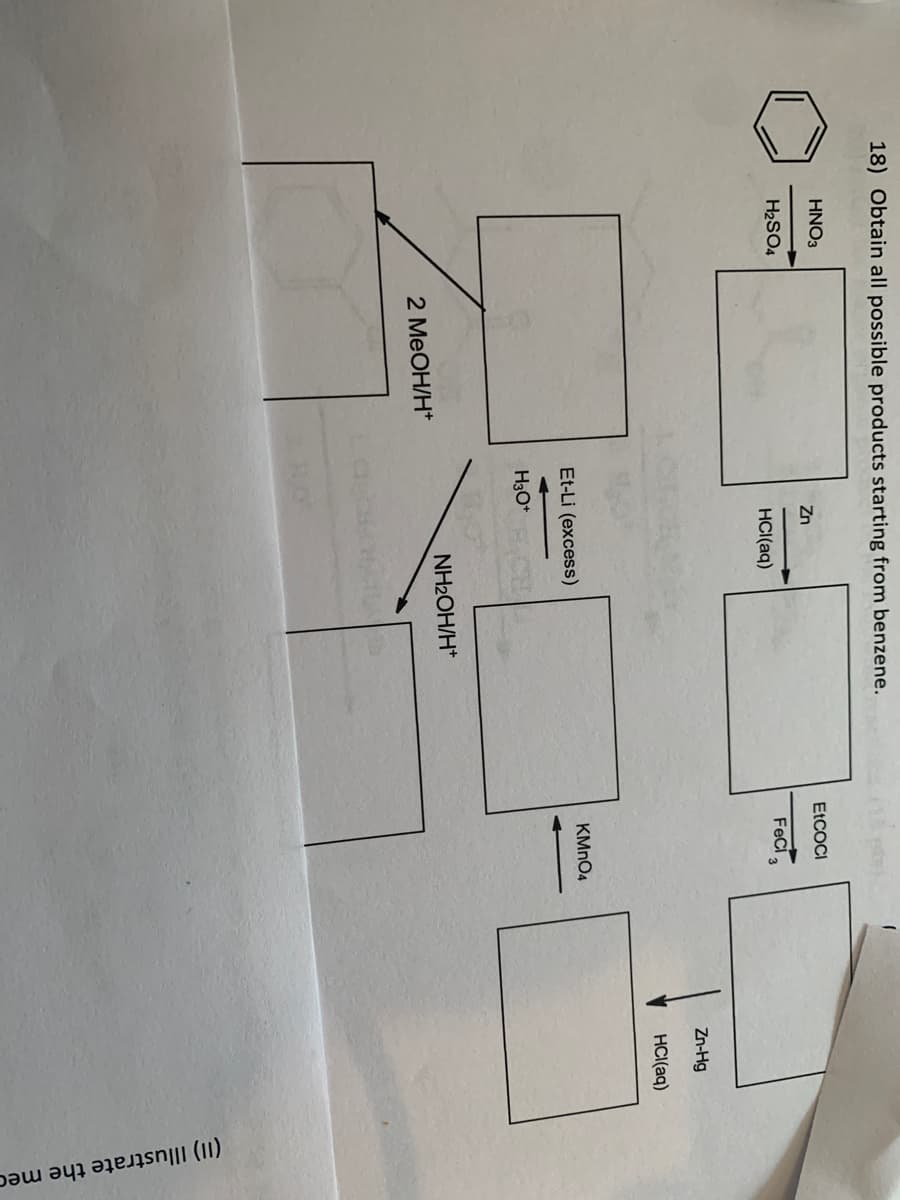
18) Obtain all possible products starting from benzene. HNO3 Zn H₂SO4 HCl(aq) Et-Li (excess) H3O+ 2 MeOH/H* NH₂OH/H* EtCOCI Feci 3 KMnO4 Zn-Hg HCl(aq) (II) Illustrate the mec
Q: Experiment No 24: Determine the relative strength of two Acids by studying the hydrolysis of ester…
A:
Q: 6. What is the relationship between A and B? HO OH II A) They are constitutional isomers They are…
A:
Q: A chemist s preparing hydrofluoric acid (HF) buffer of PH = 4.00 for an experiment. She has 125 ml…
A:
Q: What are the good titles for a community based projects?
A: There are many good titles as a community based projects some are listed below - - > Recycling…
Q: What is the pH of a solution prepared by dissolving 0.35 mol of solid CH3NH3Cl (methylamine…
A:
Q: Exercise 7 In a surface balance experiment involving myristic acid on water, 1.53 x 10-7 mole of the…
A:
Q: What is the molarity if 0.01 L of a 0.50 M HCI solution is diluted to a final volume of 0.02 L?…
A:
Q: at a given temperature. What is the rate of production e same temperature? Enter your response in…
A: Given, Rate of consumption of A = -dA/dt = 0.378 M/s Form rate of reaction, Rate of reaction =…
Q: Quantitative analysis student Jon analyzed an ore sample for its lead content and obtained a mean of…
A: Given: obtained value- 4.75% certified value- 4.50% To find: (a) percent error (b) percent accuracy…
Q: While performing the Dumas experiment, if the outside of the flask were not completely dry (Part I),…
A: Given: The outside of the flask was not completely dry in the Dumas experiment.
Q: 2. In the reaction CH4 +202 CO2 + 2H₂O a. How many grams of methane, CH4, is needed for the complete…
A: A. From the above reaction we see that 2 moles oxygen react with 1 mol methane Hence for 6 moles…
Q: Heat capacity) A bar of pure metal is heated to _61____°C The metal is placed into 300.0 ml…
A: Given : Mass of metal = 89.6 gm Initial temperature of metal = 61°C Volume of water = 300.0 ml…
Q: An empty steel container is filled with 2.50 atm of H₂ and 2.50 atm of F₂. The system is allowed to…
A:
Q: 2163 T(K) 1557 Mg₂SiO4 20 40 60 Mole % SiO₂ 80 1873 1543 SIO₂
A:
Q: Chemistry Give an example of Carbon bond cleavage reactions and ligation reactions
A: In following question calculate the carbon bond cleavage reaction and ligation reaction example
Q: what are advangtage and disadvantage of using an Atomic Force Microscopy and explain each
A: Atomic force microscopy is a technique by which we study the different type of surfaces like…
Q: Attempt 1 Calculate the cell potential for the galvanic cell in which the reaction Fe(s) + Au³+ (aq)…
A: we need to calculate cell potential for the given cell
Q: What product forms at each electrode in the aqueous electrolysis of th following salts? K3PO4 &…
A:
Q: CH3 Ill CH₂ H Br NC. NaOEt, EtOH CH3 NBS luz D NaOCH3 CH3OH ? H3O*, A E
A: Here we have to predict the major product formed in the following given reactions.
Q: 19) (1) Provide the majo Reduction, Clemme (A) Me2N (C) (E) (II) Provide the mechanism
A:
Q: Assuming equal concentrations of acid and its conjugate base, calculate the pH of NH,Cl and NH₂.…
A:
Q: Consider the following list of possible concentration cells. Which ones listed would have a positive…
A: for the concentration cell: Standard potential of the cell (E0cell) = 0V E0Cu2+/Cu = 0.34V E0Cu2+/Cu…
Q: 6. CH3 HNO3. H₂SO4 ?
A: In this question we have to tell the product of the reaction
Q: 5. Consider the following reaction mechanism. 2A + B→ с step I slow B C D step II fast 1) What is…
A: The speed of reaction is measured in terms of rate of reaction Here we are required to draw the…
Q: While performing the Dumas experiment, if the outside of the flask were not completely dry (Part I),…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: What is the expression for the solubility product constant, Ksp, for Ag2CO3? O Ksp=[Ag+1²[CO3²-]…
A:
Q: . If the 1st statement is false and the 2nd is true C. If the 1st statement is true and the 2nd is…
A: Thermodynamics is branch of chemistry in which we deal with amount of heat evolved or absorbed by…
Q: de (NaCl). The AHºd graph dd a File ous Page diss is +3.84 kJ/mol. BI UV A/ Record Audio Next Page…
A: Thermochemical reaction is equation which gives information about physical states of reactants and…
Q: Salt CsCH3CO2 NHaBr Acidic/basic/neutral What is produced in the reaction with water that confirms…
A: When cesium acetate is treated with water , then it gives cesium hydroxide and acetic acid. Here…
Q: If the peptide Ser-Pro-Ser-Arg-Ala-Lys-Gly- Trp-Gly-Phe-Leu-Tyr-Val-Ser-Ala-Ala is fully digested by…
A: Peptide bond cleavages by chymotrypsin
Q: ergy for the system to rea s supplied by a chunk of hat is the heat change for
A: From energy conservation Heat lost by metal = heat gain by water and calorimeter Heat gained by…
Q: Table 5. Ignition Testing Determining the Hydrocarbons Test Hydrocarbon Observation Inference…
A: Organic compound having carbon-carbon single bond are known as saturated compound. Organic compound…
Q: Consider this overall reaction, which is experimentally observed to be second order in X and first…
A: The rate equation for the given reaction is,…
Q: E 222/LECT/MT02-2022/SP/Traditional - Gene What is the standard cell potential for the reaction 2 Cr…
A: Cell potential is calculated for redox reaction as sum of potential of oxidation and Potential of…
Q: Give a complete explanation. Q.) What is a suppressor column and why is it employed?
A: Ans. Suppressor column: A Suppressor is a device placed between the column and the detector, and…
Q: Chemistry Draw a mirror image for each compound.label each compound as chiral or achiral . use…
A: chiral molecule nothing but the central carbon attached to four different groups or atoms called…
Q: Write a balance net ionic equation for the following redox reaction that takes place in a basic…
A: The reaction in which both oxidation and reduction reaction takes place is known as redox reaction.…
Q: 1. Predict whether the following processes are spontaneous, are spon in the reverse or are in…
A: Spontaneous process occurs by its own while Non-spontaneous process required external source to…
Q: en 4.70 and 6.70 en 3.82 and 5.82 en 1.82 and 3.82 en 2.82 and 4.82 en 5.70 and 7.70
A: It is Acidic buffer, So, pH range for buffer solution pH = pKa +/- 1 pKa = 2.82
Q: Mangrove bark is extracted by treating the finely ground wood with hot water. The original bark…
A: Given: original bark contains- 7% moisture 33% tannin 20% soluble non-tannin Residue…
Q: iven AH = +90.7 kJ/mol and AS = +221.0 J/mol K, determine AG for the reaction CH,OH(g) → CO(g) + 2…
A: Here we are required to find the Gibbs free energy for the given reaction at 250C
Q: 4. Show how to make each compound from an alkene (pay attention to regiochemistry) OH OH
A: 4. We have to give the structures of the Alkenes that give the desired alcohols.
Q: 21) Synthesize the following compounds from benzene by multi-step synthesis. 1) 3,5-dibromo benzoic…
A: Benzene undergoes electrophilic substitution reactions - 1- nitration 2- sulphonation 3-…
Q: Consider the reaction: 2 NO₂ (g) = N₂O4 (g) At equilibrium, the partial pressures of N₂O4 and NO2…
A: Equilibrium constant is defined as products concentrations divided by the reactant concentrations…
Q: What chemical would have to be in the same solution as CHO2- in order to form a buffer? Question…
A:
Q: What is the correct common name for the compound shown here?
A:
Q: Draw the major product formed when the compound shown below is mononitrated. CH3CH2 0 OCH₂CH3 Fill…
A:
Q: If a chemical reaction has a negative AH and a positive AS, then a. it will be spontaneous at low…
A:
Q: 30. The following spectrum is representative which of the following types of compounds? 100 500 1500…
A: Alcohol, OH stretching near 3300 cm-1Amide, NH 3250 cm-1 and C=O stretching at 1680 cm-1Carboxylic…
Q: Consider the titration of 150.0 mL of a 0.0040 M solution of the weak base caffeine (C8H₁0N4O2, K₂ =…
A:
Step by step
Solved in 2 steps with 1 images

- Why cis-Ru(II)Cl2(DMSO)4 reacts with pyridine, et cetera, to give substitution of the DMSO but not the chloride ligands, but trans-Ru(II)Cl2(py)4 react with suitable Na+ and K+ salts in aqueous pyridine to afford chloride-substituted derivatives. write the reactions equations.Propose a single synthesis method for cobalt(II) sulphate (CoSO4.7H2O) and describe in detail.Explain Ortho, para directors and activators ?
- (i) Tungsten hexacarbonyl can be prepared by reacting WCl6 with triethylaluminium in the presence of carbon monoxide. By-products of this reaction are aluminium trichloride and n-butane. Give the two relevant half-equations and then construct the full redox equation.(ii) Tetrarhodium dodecacarbonyl is formed upon treatment of Na+ [Rh(CO)2Cl2]- with H2O under a stream of CO. Give the two relevant half-equations and then construct the full redox equation for this reaction. Take into account that some of the CO acts as a reducing agent.I’m currently trying to write a lab report for the synthesis of dimolybdenum tetraacetate [Mo2(O2CCH3)4] from the reaction of molybdenum hexacarbonyl, Mo(CO)6 in glacial acetic acid and acetic anhydride under a nitrogen atmosphere, involving the difficult formation of a quadruple bond and requires high heat and long reaction time (approximately 20 hours). But these are the question I’m stumped on: 1. Why need the reaction be done under nitrogen? We also added dichlorobenzene and hexanes during the reaction. 2. Explain the purpose of dichlorobenzene and hexanes. 3. Why does the reaction take 20 hrs?Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides. One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(CzHs)3). 3SnCl4 + 4Al(C2H5)3 → 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.160 L of SnCl4 (d= 2.226 g/mL) was treated with 0.346 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). What is the theoretical yield in this experiment (mass of tetraethylstannane, Sn(C2H5)4)? If 0.257L of tetraethylstannane (d= 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?
- Please write out the mechanism for the acid (WHICH IS 3-BROMOBENZOIC ACID) reacting with anilide. PROVIDE THE MECHANISM AND BRIEFLY EXPLAIN THE STEPS OCCURING PLEASEAcid-catalyzed hydrolysis of a nitrile to give a carboxylic acid occurs by initial protonation of the nitrogen atom, followed by nucleophilic addition of water. Review the mechanism of base-catalyzed nitrile hydrolysis in Section 20-7 and then predict the products for each reaction below and write all of the steps involved in the acid-catalyzed reaction, using curved arrows to represent electron flow in each step.predict the stating materials, reagents, or products of the followinf reactions
- Taking Ka for acidic acid as 1.8 x 10-5, calculate H3O+ , using the known concentraions of acidic acid and soudium acecate contained in reaction mixtures 1 and 4. Reaction mixture time (s) Acidic acid concentration (mol/L) sodium acetate concentration (mol/L) calculated H3O+ concentration 1 237 0 1 x 10 -5 4 236 1 x 10 -4 1 x 10 -5Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides.One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(C2H5)3). 3SnCl4 + 4Al(C2H5)3 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.230 L of SnCl4 (d = 2.226 g/mL) was treated with 0.396 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). If 0.335 L of tetraethylstannane (d = 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?when H2O2 is added into acidified K2Cr2O7 which colour appears?

