Q: There are only two naturally occuring stable isotopes of chlonne, the masses of which are listed in…
A: Given that, the chlorine atom is naturally has only two isotopes 35Cl and 37Cl. Also, the mass of…
Q: Chemistry Give an example of Carbon bond cleavage reactions and ligation reactions
A: In following question calculate the carbon bond cleavage reaction and ligation reaction example
Q: professor
A: A professor rab this experiement (colligative properties) and found that despite precise weighing…
Q: प्रश्न 1 एवं 2 के लिये अनुच्छेद The isomerism which is due to change in position of linking atoms in…
A: Let us discuss the question step by step.
Q: Quiz 4 a. What orbitals are overlapping to form each of the following bonds? b. Classify whether the…
A:
Q: Calculate the amount of air required to completely burn 1 kg of Octane (C8H18). If the amount of air…
A:
Q: Choose the required reagents necessary to prepare the following compound in presence of catalytic…
A: These reactions are the example of the formation of imine from ketone and amide, and formation of…
Q: MISSED THIS? Read Section 15.6 (Pages 656 - 661); Watch KCV 15.6, IWE 15.9.Consider this overall…
A: Rate law depend on the concentration of reactant raised to order of the reactant.
Q: For the electrochemical cell, Cd(s) | Cd2+ (aq) || Co2(aq) | Co(s), determine the equilibrium…
A: Given,
Q: de (NaCl). The AHºd graph dd a File ous Page diss is +3.84 kJ/mol. BI UV A/ Record Audio Next Page…
A: Thermochemical reaction is equation which gives information about physical states of reactants and…
Q: Draw the most stable form of the major product in the reaction. 1. NaOC₂H5 2 2. H3O*, C2H5OH
A:
Q: A student burns a small snack food item and finds that it releases 40,800 J of energy. How many…
A: The conversion of quantity from one unit to another unit is known as unit conversion. During the…
Q: Which conservation law is violated in the strong nuclear reaction p + n → p + \overline{p}p + n
A: In nuclear reaction, atomic mass and atomic number are balanced. In nuclear reaction mass is not…
Q: work If 5.50 L of water vapor at 50.2 °C and 0.121 atm reacts with an excess of iron to produce iron…
A:
Q: What are the good titles for a community based projects?
A: There are many good titles as a community based projects some are listed below - - > Recycling…
Q: 13.1. Hydrogen sulfide (H₂S) boils at -61 H,Se at -42°C, and H₂Te at -2°C. Based on this trend, at…
A: In this question, we have to explain why H2O boil at a much higher temperature than the other…
Q: Identify the data set with the highest (a) precision and (b) accuracy. Data set Certified true Mean…
A: Accuracy refers to closeness of mean value of data set to the actual value. Precision refers to…
Q: +qcal 1927 J = qunknown 192 hermal energy is supplied by a 36 le of unknown material at 125.0 s…
A: Given qUnknown = -1927 J Initial Temperature = 125 °c Final Temperature = 23.8 °c Mass of sample =…
Q: Interaction of the reagent K. Fisher with water leaks Choose one answer: a. in one stage O b. in two…
A: We have to tell the interaction of the reagent K. Fisher with the water leaks a. In one stage b. In…
Q: Which statements are true regarding ligands? Select all that apply. Ligands donate all electrons…
A: A,B,E
Q: ion Calculate the value of K, for the reaction 2N₂(g) + O₂(g) 2N₂O(g) 1 at 298.15 K and 1773 K.…
A:
Q: The rate of a 2nd order decomposition reaction was measured at 700 and 1000 K with the following…
A:
Q: When performing the extraction process for eugenol, what are the most common problems that can be…
A: Ans. Two most common problems in extraction process for eugenol: (i) Eugenol does not extract from…
Q: Label the components of a hydrogen‑oxygen fuel cell.
A: Hydrogen-oxygen fuel cell is an electrochemical cell that convert chemical energy ( energy produced…
Q: HVALI AGE Based only on intermolecular forces, which of the following would be the most soluble in…
A: Solubility is depend on nature of subject. Polar solutes are soluble in polar solution and non polar…
Q: vill be the final temperatur of the two systems? 400. K 250. K
A: Using energy conservation Heat loss by sample A = heat gain by sample B (m.c.□T)A = (m.c.□T)B
Q: In thin layer chromatography (TLC), explain how the pigments separate on a piece of paper. 2. If…
A: The mixture of pigments should be applied to the silica gel in a very thin line, so that the…
Q: For this assignment you will be comparing acids and bases. The chart below will help you organize…
A: The properties and examples are -
Q: Give the IUPAC systematic name(s) of an alkane or cycloalkane with the formula C8H18 that has only…
A: Firstly, a primary hydrogen atom maybe designated by: Primary = a hydrogen on a carbon attached to…
Q: 1. Give the IUPAC name and the common name of the following: CH3 -N-CH CHg - N - CH, 0 7. CH₂-C-OH…
A: Multiple question given here.
Q: 10. How many grams of silver oxide are needed to react with 7.9 g of hydrochloric acid to produce…
A: Ag2O + 2HCl = 2AgCl + H2O Molar mass of ; Ag2O : 107.87×2 + 16 = 231.7 g/mol HCl : 1 + 35.45 =…
Q: What is the pOH of a solution that has pH = (8.69x10^0)? Enter your answer in scientific notation…
A:
Q: How many liters will 1.36 g of helium, He, occupy at 371 K and 705 mmHg? Report answer to one…
A: The ideal gas equation shows the relation between pressure, volume, temperature and no of moles of…
Q: To study the kinetics of iodination of acetone, why starch is used as an indicator just before the…
A: To study the kinetics of iodination of acetone, starch is used as an indicator just before the…
Q: Every Bronsted-Lowry acid donates its proton to a species which is its conjugate base. Every…
A: Given reactions are; HCl aq + NH3 (aq)→ NH4+ (aq) + Cl-(aq) and NH3 (aq) + H2O (l) → NH4+(aq) +…
Q: How much energy is in 1500 micrograms of gold? A. 1.35 × 10⁸ J B. 1.35 × 10²³ J C. 1.35 × 10¹¹ J D.…
A:
Q: and eg t2g d. An analogous compound, [Cu(H₂O)6]2+ has a different arrangement of the Draw the…
A: Given that, a metal complex is [Cu(H2O)6]2+. We have to tell the different arrangement of the…
Q: What is the concentration of X2- in a 0.130 M solut Ka2 = 7.5 x 10-11. O 1.6 x 10-8 M 6.2 x 10-4 M O…
A: H2A H+ HA- I 0.130 0 0 C -x +x +x E 0.130-x x+y x+y Further dissociat is HA- H+ A2,-…
Q: -) Give reagents and conditions that will allow ALL of the following synthetic transformations to be…
A:
Q: Calculate the standard potential, E, for this reaction from its equilibrium constant at 298 K. X(s)…
A:
Q: To do: Identify the oxidizing agent and reducing agent in the reactions. 1. 2Na2S2O3+l2-Na2S4O6+2Nal…
A: Here we have predict the substance which act as oxidizing agent and reducing agent.
Q: f. The H-X-H bond angle in H₂O is 104.5°, while in H₂Te the explanation.
A:
Q: Salt CsCH3CO2 NHaBr Acidic/basic/neutral What is produced in the reaction with water that confirms…
A: When cesium acetate is treated with water , then it gives cesium hydroxide and acetic acid. Here…
Q: What is a calorimeter constant? ) Amount of energy lost out of or absorbed by a calorimeter )…
A: In this question, we have to choose the correct option for the given question.
Q: O oxygen O chlorine O barium Oboron
A: Multiple choice question is given here.
Q: 3H₂S(aq) + 2HNO3(aq) → 3S(s) + 2NO(g) + 4H₂0) 6. What is the percent yield if 23.6 g of…
A: Given, 3H2S(aq) + 2HNO3(aq) ➝ 3S(s) + 2NO(g) + 4H2O(l) mass of hydrosulfuric acid (H2S) react = 23.6…
Q: (c) Give reagents and conditions that will allow the following synthetic transformation to be…
A:
Q: What is the pH of 0.1000 M Na₂HPO4? (K₁2=6.2x10-8, K3=4.8x10-13) What is the pH of 0.1000 M Na3PO4?…
A: The solutions considered are 0.1000 M Na2HPO4 and 0.1000 M Na3PO4. The question requires to…
Q: 1. What are [H3O+], [OH-], and pOH in a solution with a pH of 8.59 at 25 oC? Full Solution 2. The…
A: Since we know that, At 25°C , pH +pOH = 14 pH = - log10[H3O+] pOH = -log10[OH-]
Q: If 25.0 mL of water are added to this system, indicate whether the following statements (Note the…
A: If we add water then concentration of acid and salt is changed due to volume increases. a) True b)…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

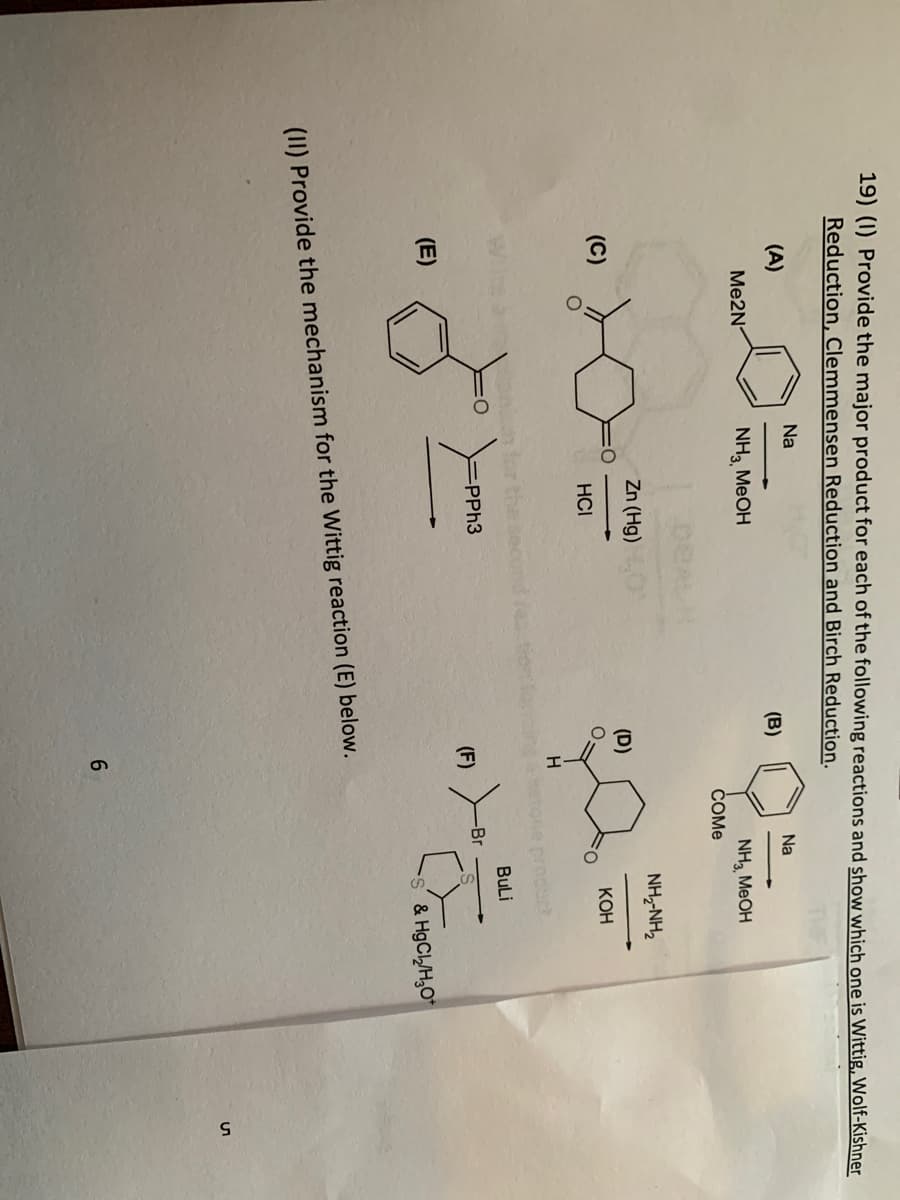
- Describe a synthetic plan with suitable justification of the sequence of reactions you would understate to complete synthesis of compound 1 from compounds 2,3,4. Suggest suitable reagents1. Discuss a disadvantage of Friedel - Crafts acylation reaction 2. Propose a mechanism for the formation of (ii)(a) Illustrate the following name reactions by giving example :(i) Cannizzaro’s reaction(ii) Clemmensen reduction(b) An organic compound A contains 69.77% carbon, 11.63% hydrogen and rest oxygen. The molecular mass of the compound is 86. It does not reduce Tollen’s reagent but forms an addition compound with sodium hydrogen sulphite and gives positive iodoform test. On vigorous oxidation it gives ethanoic and propanoic acids. Derive the possible structure of compound A.
- Draw/outline a reasonable and detailed mechanism for the dehydration of 2-methylcyclohexanol catalyzed by phosphoric acid. Use curved arrows to show the flow of electrons and draw the structures of all intermediates and byproducts formed in the reaction. Predict and rationalize the expected product distribution based on thermodynamic aspectsSuggest reactivity of compound A, B and C in increasing order of E2 reactionSuggest and explain two synthetic methods for the conversion of compound 6 to compound 4. Include side reactions
- Wolff-Kishner reduction of compound W gave compound A. Treatment of A with m-chloroperbenzoic acid gave B which on reduction with LiAH4 gave C. Oxidation of compound C with chromic acid gave D (C9H14O). Suggest the structures for A, B, C, and D.A student was given from the list of the compounds below A, B and D blindly and asked to identify them all. He treated each of them with Brady's reagent (2,4-ditrophenylhydrazine) and isolated a bright yellow compound for one of them, but the other two gave false negatives. The student reasoned that the false negatives may be due to sterics and, on further thinking, it dawned on him that he might be able to rule out one of the false negatives with the haloform test. What compound did he find compatible with the haloform test? That compound did indeed give a false negative in the Brady test. Which of the other two was positive in the Brady test? A = haloform B = Brady A = haloform D = Brady B = haloform A = Brady B = haloform D = Brady D = haloform A = Brady D = haloform B = Brady3. Obtain acetophenone and acetaldehyde by reaction of glycols with periodic acid. Justify your answer with the reaction mechanism.
- Please show the electron-flow mechanism of the general synthesis of fluorobenzene from aniline, this involves predicting major and by-products using electronic and structural effects. The arrow push mechanism must be shown.The following equation shows the bromination of compound 1. Propose a structure for product D and a curved arrow mechanism that accounts for its formation featuring the initiation, propagation and termination steps. can you also discuss the stereochemical outcome.Compound A was prepared by sequential Suzuki cross-coupling reactions.First, compound 1 was treated with an aryl iodide to give compound 2, which was then treated with an aryl boronic acid to produce compound A. How wood the structures for the aryl iodide, boronic acid, and compound 2 look like.

