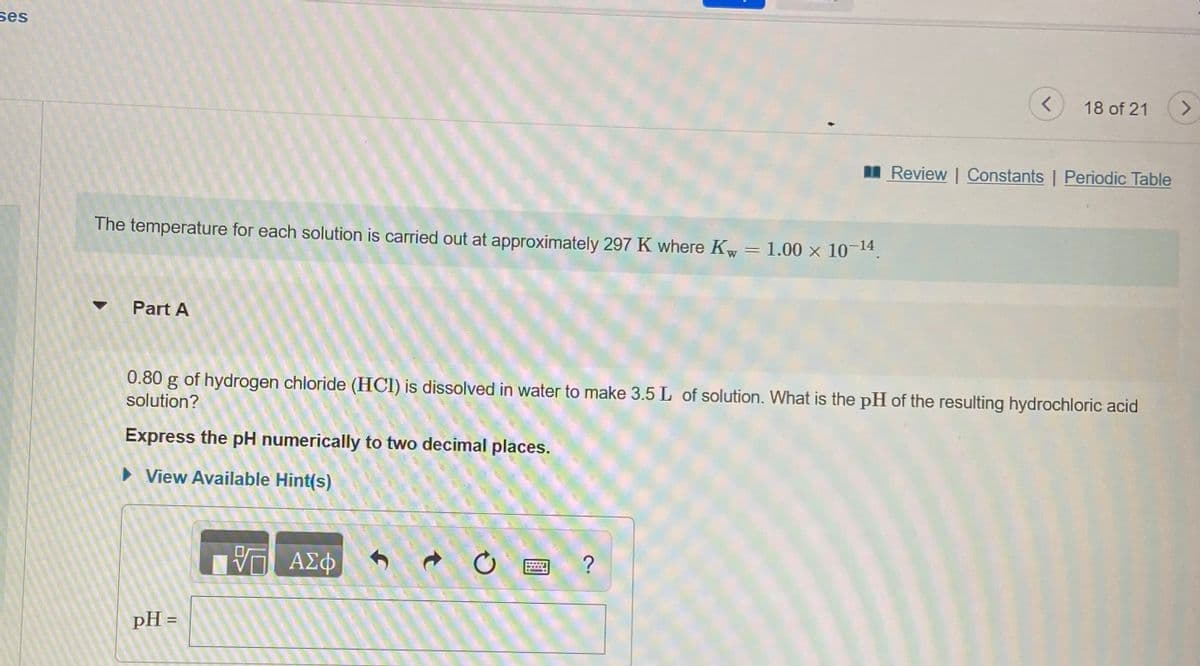
18 of 21 I Review | Constants | Periodic Table The temperature for each solution is carried out at approximately 297 K where Kw 1.00 x 10-14 Part A 0.80 g of hydrogen chloride (HCI) is dissolved in water to make 3.5 L of solution. What is the pH of the resulting hydrochloric acid solution? Express the pH numerically to two decimal places. • View Available Hint(s) pH = 国
18 of 21 I Review | Constants | Periodic Table The temperature for each solution is carried out at approximately 297 K where Kw 1.00 x 10-14 Part A 0.80 g of hydrogen chloride (HCI) is dissolved in water to make 3.5 L of solution. What is the pH of the resulting hydrochloric acid solution? Express the pH numerically to two decimal places. • View Available Hint(s) pH = 国
Chapter21: Potentiometry
Section: Chapter Questions
Problem 21.22QAP
Related questions
Question
Transcribed Image Text:ses
18 of 21
<>
I Review | Constants | Periodic Table
The temperature for each solution is carried out at approximately 297 K where Kw = 1.00 x 10-14.
Part A
0.80 g of hydrogen chloride (HCI) is dissolved in water to make 3.5 L of solution. What is the pH of the resulting hydrochloric acid
solution?
Express the pH numerically to two decimal places.
> View Available Hint(s)
ΑΣφ
pH =
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning