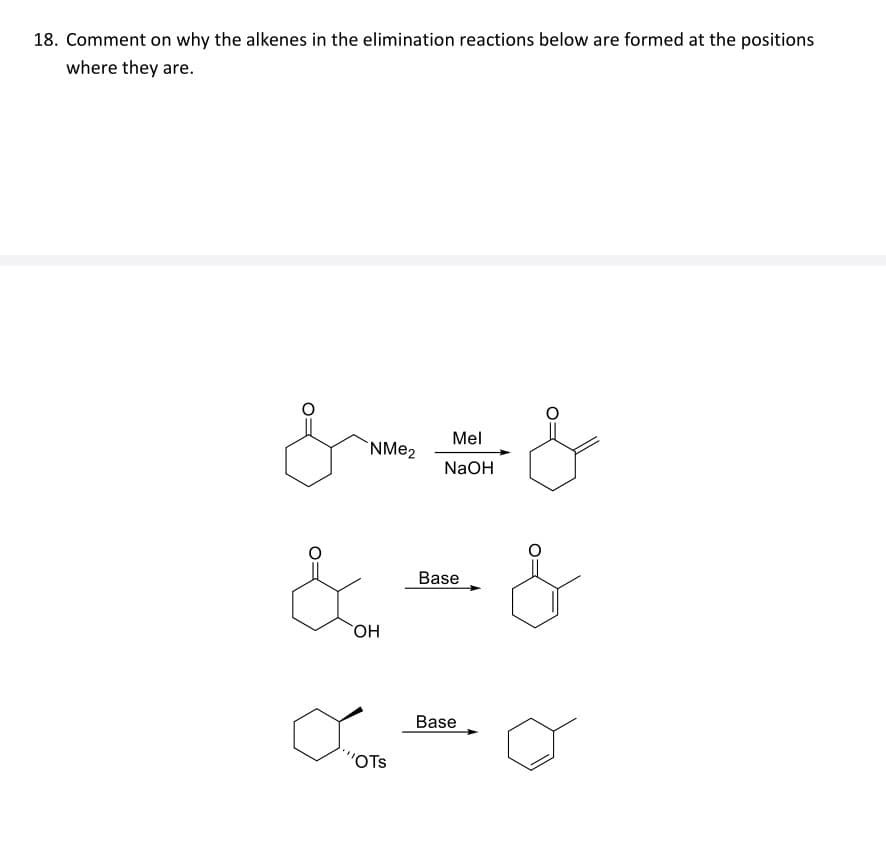
18. Comment on why the alkenes in the elimination reactions below are formed at the positions where they are. &&& & OH NMe2 Base Mel NaOH & OTS Base
Q: Draw the major product of the following sequence of reactions. If the reaction is likely to give you…
A: In the given reaction sequence the substrate is toluene and it is reacted with two sets of reagents.…
Q: Three atoms (I, II, and III) are indicated in the molecule shown below. || III What is the oxidation…
A:
Q: Don't use hand raiting and step by step answer please
A: To calculate the pKa and Ka of an indicator such as phenolphthalein, we need to know the pH at which…
Q: Draw the structure of the organic product of the following transformation. CNO Br X AICI 3 NEW R +…
A: Step 1:As we see that in the given reaction condition Lewis acid is given and the product formed is…
Q: Please correct answer and don't use hend raiting
A: Step 1: Primary Alcohol :-- Primary alcohols are those alcohols where the carbon atom of the…
Q: None
A: Step 1: Step 2: Step 3: Step 4:
Q: Make a synthesis with 4 steps. Use addition, subtraction, and elimination reactions. Try to change…
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: synthesis question . . . what mechanisms should i use to get to the product? =N د C
A: Here is a general approach: Start with a precursor molecule that has a correct carbon chain. You…
Q: is it appropriate to write the equation for standard enthalpy of formation of Fe) as shown? 2Fe (s)…
A: Standard enthalpy of formation is the enthalpy change when one mole of compound is formed form it's…
Q: When a 18.6 mL sample of a 0.340 M aqueous hydrofluoric acid solution is titrated with a 0.405 M…
A: The objective of this question is to calculate the pH at the midpoint of a titration between…
Q: 14 Homework Unanswered Due Apr 11th, 11:59 PM Select the correct retrosynthesis to obtain the…
A: To answer this query, permit's observe the stairs of retrosynthesis. 1. Identify the target…
Q: The mole fraction of neon in dry air near sea level is 0.00001818, where the molar mass of neon is…
A: Step 1: Use the Raoult's Law to solve the problem. Refer to the solution below.
Q: TRANSMITTANCEI Compound A has 4 carbons and one heteroatom (not C or H). a. Please give appropriate…
A: The objective of the question is to identify the structure of compound A, its reaction scheme with…
Q: A chemist is studying the following equilibirum, which has the given equilibrium constant at a…
A: The objective of the question is to calculate the equilibrium pressure of O3 in the given reaction.…
Q: Please correct answer and don't use hend
A: Step 1:Conjugate Addition (1,4-Addition): One of the most notable reactions involving Gilman…
Q: 2. Draw the appropriate structure from the given IUPAC name. a. 7-iodo-2-heptyne b.…
A: The IUPAC nomenclature of organic compounds follows this naming format:Drawing an organic compound…
Q: Draw an alkyl halide that would undergo an SN2 reaction to yield this product under the conditions…
A: Step 1:As we know that in SN2 reaction, there occurs nucleophilic attack from back side,at the…
Q: 6. (13 pts) A solution of 0.1000 M HCl was used to titrate 25.00 mL of Na2CO3 (0.0800 M). Calculate…
A: Given data:Reaction: (A - {Products})Rate law: ({Rate} = -{d[A]}/{dt} = k[A]^x)Ka1 = (4.46 *…
Q: 791 Problem 15 of 35 Submit Draw the major product of this reaction. Ignore inorganic byproducts. 1.…
A:
Q: Devise a synthesis for your assigned compound using organic reagents no larger than two or three…
A: Given is organic synthesis reaction.The given compound is amide.
Q: A 415 g sample of water absorbs infrared radiation at 1.01 × 10 nm from a carbon dioxide laser.…
A: The values are provided in questionmass of water = 415 g specific heat of water c = 4.186 J /…
Q: Step by step answer please and don't use hand raiting please
A: The R/S configuration is a method used to assign the spatial arrangement of substituents around a…
Q: 6. A solution of 0.1000 M HCl was used to titrate 25.00 mL of Na2CO3 (0.0800 M). Calculate the pH of…
A: The objective of this question is to calculate the pH of the solution at different stages of the…
Q: write the balanced for the acid hydrolysis of ethyl propanoate, showing line-angle or condensed…
A: Ester on hydrolysis in the presence of acid produces carboxylic acid and alcohol.
Q: der these chemical species by increasing pH of an 0.1 M aqueous solution of each. That is, imagine…
A: In a 0.1 M aqueous solution, the objective is to arrange the chemical species by raising pH. We'll…
Q: When a 0.013 M aqueous solution of a certain acid is prepared, the acid is 0.73% dissociated.…
A: To calculate the acid dissociation constant (Ka), we can use the equation for percent…
Q: 1. Write IUPAC name for each structure below. a. b. ہو Br d. می شماره e.
A: Rules for the IUPAC name of organic compounds:1. Identify the longest carbon chain.2. Identify all…
Q: Question 3 What mass of NiCO, will dissolve in 1.4 L of 0.502 M NaCO₂ solution? K of NICO, is…
A: Given: [CO32−]=0.502M;V=1.4L;Ksp=6.6x10−9;massNiCO3=???g;MNiCO3=118.702g/molStep 1: Write the…
Q: PLEASE, provide the missing reactants, reagents or products to complete the reaction.
A: Since you have posted multiple questions, according to Bartleby Q&A guidelines we are supposed…
Q: Please correct answer and don't use hend raiting
A: Step 1:This reaction is known as E2 elimination reaction,as we see that the given base is sterically…
Q: (CH21/23) [References] Draw the structure of the major organic product(s) of the reaction. H₂C 1. 2…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: URGENT! Please draw the full reaction mechanism with arrow pushing of N-Acetylserine reacting with…
A: The objective of the question is to draw the reaction mechanism for the conversion of N-Acetyl…
Q: A 1000 mL solution contains 0.500 M K,CrO, and 0.500 M Na₂SO. A concentrated Ba(NO3)2 solution is…
A: The question is based on the concept of solubility product principle.It states that when a weak…
Q: What is the solubility of Zn(PO4)2 in a solution that contains 0.0300 M Zn⁺2 ions? (Ksp of Zn(PO4)2…
A:
Q: Part A At equal concentrations, how would the freezing point of an electrolyte (e.g. NaCl)…
A: ∆Tf = iKmWhere i = van't Hoff factor for NaCl it is 2 while for glucose it is 1.∆Tf = Tsolution -…
Q: Please draw curved arrows for all the orts
A: Step 1: Step 2: Step 3: Step 4:
Q: Should the mole be included in the SI System of units? Why or Why not.
A: What Is the Mole?The mole (symbol: mol) is a fundamental unit in chemistry. It refers to a specific…
Q: In the reaction A → B + C the concentration of A decreases exponentially with time. Why can't the…
A: The objective of the question is to understand why the rate equation for the reaction A → B + C…
Q: None
A: Step 1: Step 2: Step 3: Step 4:
Q: (b) Ir(I) ions can form octahedral, tetrahedral or square planar complexes depending on the ligands…
A: For transition metals, the geometry of the complex (octahedral, tetrahedral, or square planar)…
Q: Draw major E2 ELIMINATION product. Hint: Review Chapter 10.3, the elimination can only occur if the…
A:
Q: The base protonation constant X of ammonia (NH Calculate the pH of a 0.16 M solution of ammonia at…
A:
Q: 13. Which of the following could be the quantum numbers (n, I, m₁, ms) for the valence electron in a…
A: Step 1: Step 2: value of ml can not be greater than the value of l. Step 3: Step 4:
Q: Recognizing different skeletal structures How many different molecules are drawn below? X G
A: The objective of the question is to identify the different molecules in the given sample.
Q: 7.8 g of calcium fluoride is dissolved in 100 L of water at 25\deg C. Will a precipitate be formed?
A: Molar mass of CaF2 = 78.078 gMass of CaF2 = 7.8 gTo determine if a precipitate will form, we need to…
Q: If enough of a monoprotic acid is dissolved in water to produce a 0.0111 M solution with a pH of…
A: The objective of the question is to find out the equilibrium constant for the given acid.The…
Q: Draw the major product of this elimination. Consider regiochemistry and stereochemistry. Ignore…
A:
Q: The electron transport chain (ETC), or respiratory chain, is linked to proton movement and ATP…
A: The statements that accurately describe the electron transport chain (ETC) are:Electron carriers are…
Q: What is the product? Br CN 1. NaOEt 2. DIBAL 3. H3O+
A: NaOEt is a strong base,DIBAL-H is a reducing agent.H3O+ is acidic water workup.
Q: Question 5: A solution is prepared by adding 375 mL of 5.16 x 10-3 M silver nitrate and 125 mL of…
A: Step 1: Step 2: Step 3: Step 4:
Step by step
Solved in 2 steps

- Why do you suppose ketone halogenations in acidic media are referred to as being acid-catalyzed, whereas halogenations in basic media are base-promoted? In other words, why is a full equivalent of base required for halogenation?In light of your answer to Problem 30-40, explain why a mixture of products occurs in the following reaction:please explain why this happends, im having trouble in this section. Part A Chymotrypsin, an enzyme that hydrolyzes peptide bonds in proteins, functions in the small intestine at an optimum pH of 7.7 to 8.0. How is the rate of the chymotrypsin-catalyzed reaction affected by each of the following conditions?
- Given the information pictured, The final step of the reaction sequence uses PhLi. Provide a reason and possible mechanism by which this final elimination and hydrolysis reaction would occur.10.14 Complete the following reactions: Show the step-by-step process. Do not use shortcut methods. Make it as detailed as it can be. Encode (not hand-written)!Can you pls assist me with the mechanism of Benzyl alcohol being oxidised to Benzyl aldehyde and to Benzoic acid using KMnO4 as an oxidant. Showing arrows and everything.
- 3-metyl-2-butanol react with H₂SO₄ and continue with reductive ozonolysis process. What would become the product(s)?w how enols, enolate ions, andenamines act as nucleophiles. Predictthe products of their reactions withhalogens, alkyl halides, and otherelectrophiles. Show how they areuseful in synthesis.Study the curved arrows in the reactions below and provide the structures of theproduct(s) formed. One block may represent more than one product/by-product.
- One possible way of determining the identity of an alkene, is to let itundergo an oxidative cleavage reaction in the presence of hot basicpotassium permanganate. You are given two containers said to containdifferent alkenes. Container A is marked as cis / trans‐2‐butene andcontainer B as 2‐methyl‐1‐butene. Explain by referring to the formation ofproducts, how you would verify the identity of the alkenes.What is /are organic product(s) expected when 1-butyne is first treated with sodium amide and the resulting acetylide anion reacts with tert-butyl chloride before aqueous acid workup?Can you explain (D). Please Draw out the mechanisms for the reactions to include the arrow pushing.

