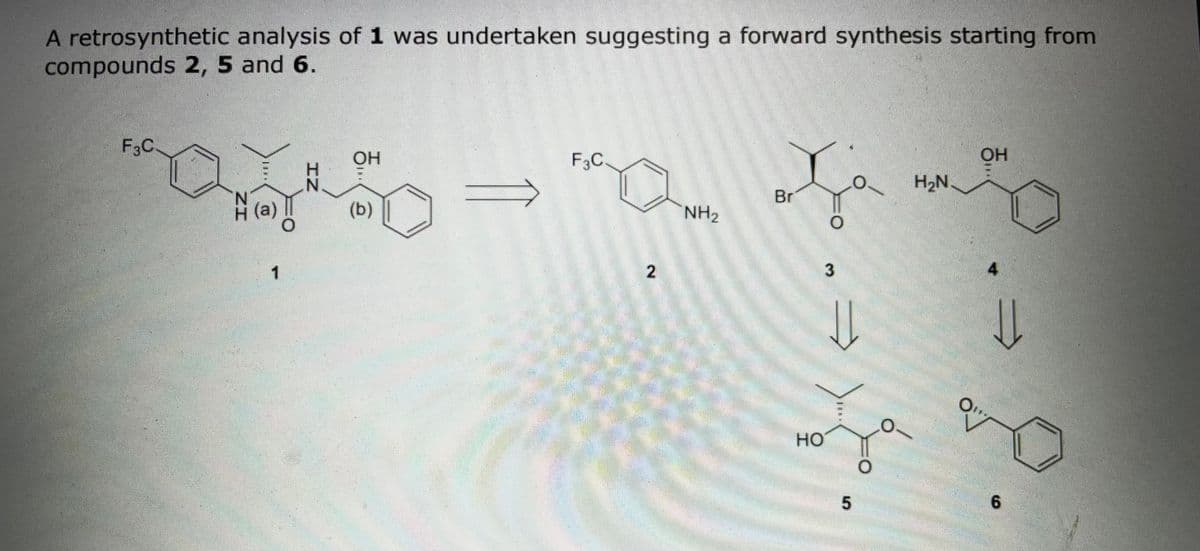
A retrosynthetic analysis of 1 was undertaken suggesting a forward synthesis starting from compounds 2, 5 and 6. F3C. OH F3C. OH H2N. Br (b) NH2 1 2 но
Q: Scheme 2: Synthesis of a common antihistamine from benzaldehyde. Mg, ether 1. Ether 2. 1M HCI HBr…
A: Diphenhydramine is an antihistamine. Synthesis of antihistamine from benzaldehyde:
Q: b) Pyrrole and indole both react with the reagents shown below to give compounds and F respectively.…
A:
Q: 1. Complete the disconnection of the compound given below and briefly explain the following terms…
A: Since there are total 3 question but as per the Bartley rules we can only answer one question. If…
Q: 8. From the given starting material provide a reasonable synthetic route to the following molecule.…
A: Here we have to synthesize the given compound from the starting material 2-methylbutane-1,3-diene.
Q: Waile product give mechani dm O HNO2 me Ph Cat TSOH THN IV HQ.
A: As per the rules, only the first three questions can be answered. Organic transformations.
Q: (a) Which is a stronger acid NH2 NH, b. If nitration is done on both the compounds what will be the…
A:
Q: What spectral features including mass spectra, IR spectra, proton spectra and carbon spectra allow…
A: In case of benzonitrile, a weak IR-absorption occurs at 2260-2222 cm-1 which is due to C-N triple…
Q: Treatment of Compound E with sodium amide and then methyl iodide does not produce the desired…
A: -> NaNH2 is a base hence, it can abstract most acidic hydrogen from compound and generate…
Q: Suppose you have compounds A-D at your disposal. Using these compounds, devise two different ways to…
A: (i) A + D -------> E ( major product) (ii) B + C -------> E ( minor product) Method (i) is…
Q: can be 4. (a) The following compound synthesised via Robinson annulation. Provide retrosynthetic…
A: Robinson annulation is a reaction which uses a ketone and a methyl vinyl ketone (α,β-unsaturated…
Q: Part C: Provide Products and Reagents C1) Provide the best reagents required to convert benzene into…
A: Clemmensen reagent is used to -CO- into -CH2... Mera directing group directs incoming group at meta…
Q: CH3 CH2 H3C CH3
A: A compound with carbon and hydrogens is known as hydrocarbons. These hydrocarbons can be synthesized…
Q: (b) Provide 1,3,5 Anazine product(s) : H3C the (ii) H3C- NH2 missing MeO MeO NH–NH NMC2 -R…
A:
Q: 2. THP (tetrahydropyranyl) is a commonly used protecting group for alcohols. As shown below,…
A: Sometimes more than one functional group is present in the compound. When a reagent is added to such…
Q: Propose a short synthesis f or ONE of the following molecules. You can start with acetylene, and…
A: The answer is given as follows
Q: Which of the following statements about terminal alkynes is FALSE?I I. A geminal dihalide is…
A: Which of the following statements about terminal alkynes is FALSE.,
Q: The product obtained when the compound below is treated with one mole of CH3OH is. Select one: OCH3…
A: Given compound: We have to find the structure of the compound when the above compound is treated…
Q: Which of the following reactions describes why Acetic Anhyhdride needs to stay dry in order for it…
A: The acetic anhydride is need to stay dry because acetic anhydride in the presence of water gets…
Q: 3. (a) Arrange the following compounds according to their reactivity towards electrophile. Justify…
A:
Q: NABH4 CH3CH2OH
A: In this question, we have to find out the correct answer of given problem by the help of the…
Q: In the following three compounds(1,2,3) arra
A: Benzene is having pi-electron excess, hence it undergo electrophilic substitution reactions ,…
Q: Explain the difference in reactivity between CH3OH2 and CH3OH in a nucleophilic substitution…
A: The alcohol contains a two lone pair and therefore, will be reactive for the attack on reagent to…
Q: To prepare butanoic acid from 1-propanol, which sequence of reagent(s) is/are best employed? a)…
A: Scheme 1: Step 1 : propanol is converted into bromo propane using PBr3. Step 2: Bromo propane is…
Q: Arrange the following compounds in order of increasing reactivity to nucleophilic addition. I.…
A: Nucleophilic addition reactions are the addition reactions in which a nucleophile attacks the…
Q: 1. Draw the retrosynthetic analysis of compound A by providing the appropriate disconnection and…
A: We have to suggest a complete synthetic pathway with the reagents and reaction conditions to…
Q: 13) Propose a synthesis of compound 47 using the chemicals in the box. 47 РСС HBr Mg H2O
A:
Q: CHM 1321EV –- Organic Chemistry I 15. Use a flow chart to illustrate how you could separate each of…
A:
Q: HC=CH Reagents available a. HCI e. На, Рd i. H2 / Lindlar's catalyst b. HBr f. OsO4 then NaHSO3 j.…
A:
Q: Which of the following reactions of benzene requires a Lewis acid? a Bromination b FC…
A: Given statement is : Which of the following reactions of benzene requires a Lewis acid? Options…
Q: -3. (a) Provide retrosynthesis corresponding forward reaction for the following compounds : OH (i)…
A:
Q: (d) . 1. `MgBr 1. O3 (2 equivalents) 2. Me2s 2. H20 K2Cr,O7 H2SO4 (2 equivalents) (d) Propose an…
A: Grignard reagent is mainly used to form new carbon-carbon bonds. Ozonolysis results in the cleave of…
Q: А B C D Br Br
A: We have to rank the order of the reactivity for the given compound
Q: Tertiary (3°) aromatic amines react with NaNO2 and HCl to afford products of electrophilic aromatic…
A: The electron withdrawing /electron releasing groups have important role in the activation and…
Q: 19 4. Devise a synthesis for the following compound 30-0-00
A:
Q: 1. Reaction of excess methyllithium with ethyl acetate (CH3CO2C2H5) produces, after acid work-up,…
A: Methyl lithium and magnesium bromide are very reactive and nucleophilic in nature.
Q: Explain the difference in reactivity between CH3OH2 and CH3OH in a nucleophilic substitution…
A: The alcohol contains a two lone pair and therefore, will be reactive for the attack on reagent to…
Q: Select the expected product of the following reaction sequence. NaCN 1. LIAIH, THF DMSO 2. Hао Br…
A: For the given reaction in the first step SN2 reaction take place to form alkyl cyanide with…
Q: Provide the reagents necessary to carry out the following conversion OH OH A 1) HCN; 2) PCC B 1)…
A:
Q: 15. Outline a synthetic route, with no more than three steps, to accomplish the following…
A: Lactones can be reduced to diols using reducing agents like LiAlH4, LiBH4, DIBAL-H, etc.
Q: 7. Suggest a reasonable synthesis for the following molecule, starting from benzene and using any…
A: First, we have to prepare p-methozybenzene diazonium chloride, p-CH3O-C6H4-N2Cl as shown in Step-2.…
Q: 2. Consider the following scheme. NH2 NH2 Reagent A Но ÓH Compound T HO Compound S KOH, NH,NH2…
A: a) The IUPAC name of the given compound T is, 1-amino-1-(4-hydroxy phenyl) propan-2-one b)…
Q: 4. Suggest a synthesis for the following from the indicated. starting material and any necessary…
A: “Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: 6. Propose a route to Compound 2 from only bromobenzene and methyl formate as carbon sources, plus…
A: Let us talk about the grignard reagent. Here firstly the bromobenzene is used. The we have to…
Q: 9. Provide the following: aniline i) the major organic product of the reaction shown, and ii) the…
A:
Q: 1. Propose a stepwise process for the synthesis of the aromatic compound from benzene. C₂H5 CH3 a.…
A: Given compounds are : Propose a stepwise synthesis of the given compounds = ?
Q: 15. In general, Grignard reagents RMgX react rapidly in anhydrous solvents with ethylene oxide…
A: Grignard reagent belongs to the family of organometallic compounds. This reagent is widely used for…
Q: 4 Suggest the retrosynthetic analysis of Decane CH-(CH))s-CHs
A: It is a technique ofchemical synthesis by involving deconstructing a target molecule into readily…
Q: Show the structures of the missing substance(s) in each of the following acid-base equilibria. a.…
A: Nitrogen is lewis base which can accept hydrogen from acid and form positive charge on it.
Q: Do a retrosynthetic analysis on the following compound, ending withavailable starting material.
A: Do a retrosynthetic analysis on the following compound, ending with the available starting material.
Suggest and explain two synthetic methods for the conversion of compound 6 to compound 4. Include side reactions
Step by step
Solved in 2 steps with 1 images

- what is the retrosynthetic analyiss and synthesis of this?Sketch and explain the IR, UV, mass, and NMR (1H and 13C) spectroscopy data of Benzonitrile[Grignard Synthesis of Benzoic Acid] Characterization 1. Explain melting point determination as a characterization technique 2. Explain IR spectroscopy as a characterisation technique.
- Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.Propose structural formulas for compounds (2) and (3).Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.Name the types of functional groups in estroneOver the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.How many chiral centers are present in estrone?
- Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.Show how your proposals for compounds (2) and (3) can be converted to compound (1). (Note: In the course of developing this synthesis, Tietze discovered that vinylic bromides and iodides are more reactive in Heck reactions than are aryl bromides and iodides.)Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q. In the course of the double Heck reactions, two new chiral centers are created. Assume in compound (3), the precursor to rings C and D of estrone, that the fusion of rings C and D is trans and that the angular methyl group is above the plane of the ring. Given this stereochemistry, predict the stereochemistry of…For butenafine, develop a synthesis that starts with napthalene and benzene. (aromatic methyl groups can be converted to alkyl bromides using Br2/light, and aldehydes by heating the alkyl bromide in DMSO, eg. ArCH3 to ArCH2Br then to ArCHO).
- When the nitrogen-containing aromatic heterocyclic compounds 1 and 2 are treated with HCl, only 1 forms the hydrochloride salt, whereas compound 2 is unreactive. Provide an explanation for this observed reactivity.Propose a mechanism and use stability factors to account for the formation of a major product in the reaction of pyrrole with Br2, FeBr3.2-methoxypyrrole is brominated exhaustively by treatment with Br2, while 2-methoxypyridine is selectively brominated at the 5-position. Explain thedifferences in reactivity, and the regioselectivity of the pyridine bromination,using resonance structures as part of your explanation.

