19) Consider the table of allosteric effectors and their effect on the following metabolic processes.
19) Consider the table of allosteric effectors and their effect on the following metabolic processes.
Biochemistry
6th Edition
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Reginald H. Garrett, Charles M. Grisham
Chapter18: Glycolysis
Section: Chapter Questions
Problem 18P: Distinguishing the Mechanisms of Class I and Class I Aldolases Fructose bisphosphate aldolase in...
Related questions
Question
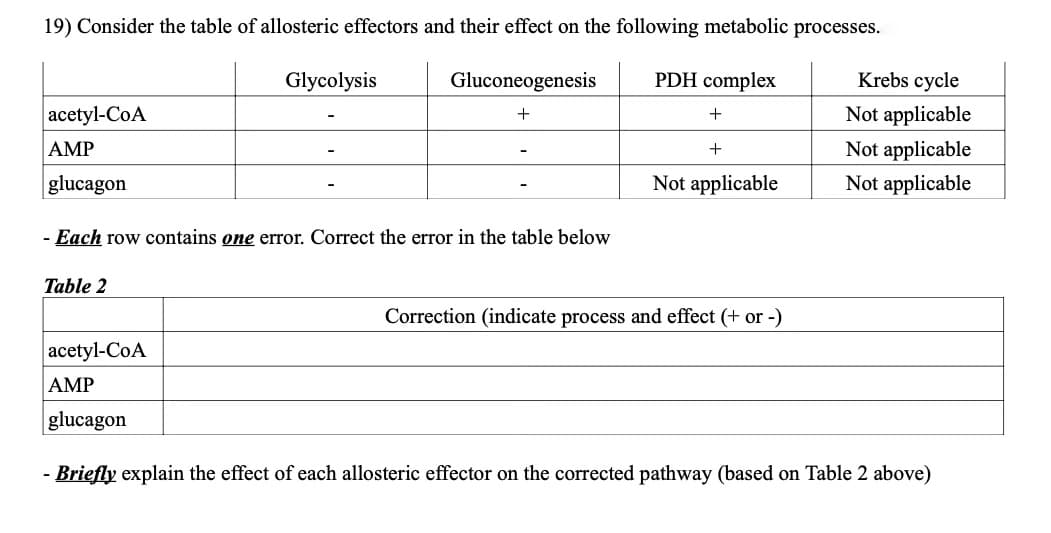
Transcribed Image Text:19) Consider the table of allosteric effectors and their effect on the following metabolic processes.
Glycolysis
Gluconeogenesis
PDH complex
Krebs cycle
acetyl-CoA
Not applicable
AMP
Not applicable
+
glucagon
Not applicable
Not applicable
- Each row contains one error. Correct the error in the table below
Table 2
Correction (indicate process and effect (+ or -)
acetyl-CoA
AMP
glucagon
- Briefly explain the effect of each allosteric effector on the corrected pathway (based on Table 2 above)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning