19. Which statement about structural (constitutional isomers) is CORRECT? A) Structural (constitutional) isomers are two compounds with the same molecular formula and the same structure. B) Structural (constitutional) isomers are compounds which always have the same carbon chain length. C) Structural (constitutional) isomers are compounds in which the functional group will always be in the same position. D) Structural (constitutional) isomers are compounds that contain the same number of atoms of each element but the specific arrangement of the atoms differs. 20. The following two compounds are CH3 CH3 Br Br A) Constitutional isomers B) Conformational isomers C) Geometric isomers D) Enantiomers
19. Which statement about structural (constitutional isomers) is CORRECT? A) Structural (constitutional) isomers are two compounds with the same molecular formula and the same structure. B) Structural (constitutional) isomers are compounds which always have the same carbon chain length. C) Structural (constitutional) isomers are compounds in which the functional group will always be in the same position. D) Structural (constitutional) isomers are compounds that contain the same number of atoms of each element but the specific arrangement of the atoms differs. 20. The following two compounds are CH3 CH3 Br Br A) Constitutional isomers B) Conformational isomers C) Geometric isomers D) Enantiomers
General Chemistry - Standalone book (MindTap Course List)
11th Edition
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Chapter23: Organic Chemistry
Section: Chapter Questions
Problem 23.84QP
Related questions
Question
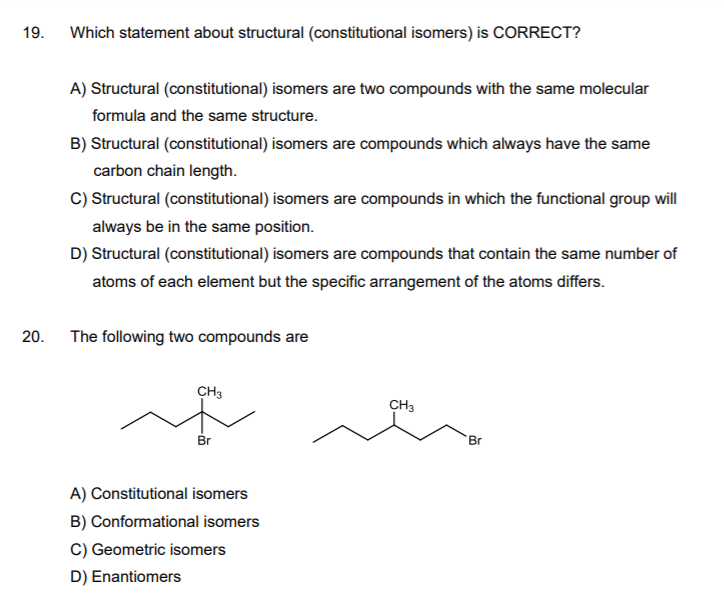
Transcribed Image Text:19.
Which statement about structural (constitutional isomers) is CORRECT?
A) Structural (constitutional) isomers are two compounds with the same molecular
formula and the same structure.
B) Structural (constitutional) isomers are compounds which always have the same
carbon chain length.
C) Structural (constitutional) isomers are compounds in which the functional group will
always be in the same position.
D) Structural (constitutional) isomers are compounds that contain the same number of
atoms of each element but the specific arrangement of the atoms differs.
20.
The following two compounds are
CH3
CH3
Br
Br
A) Constitutional isomers
B) Conformational isomers
C) Geometric isomers
D) Enantiomers
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning