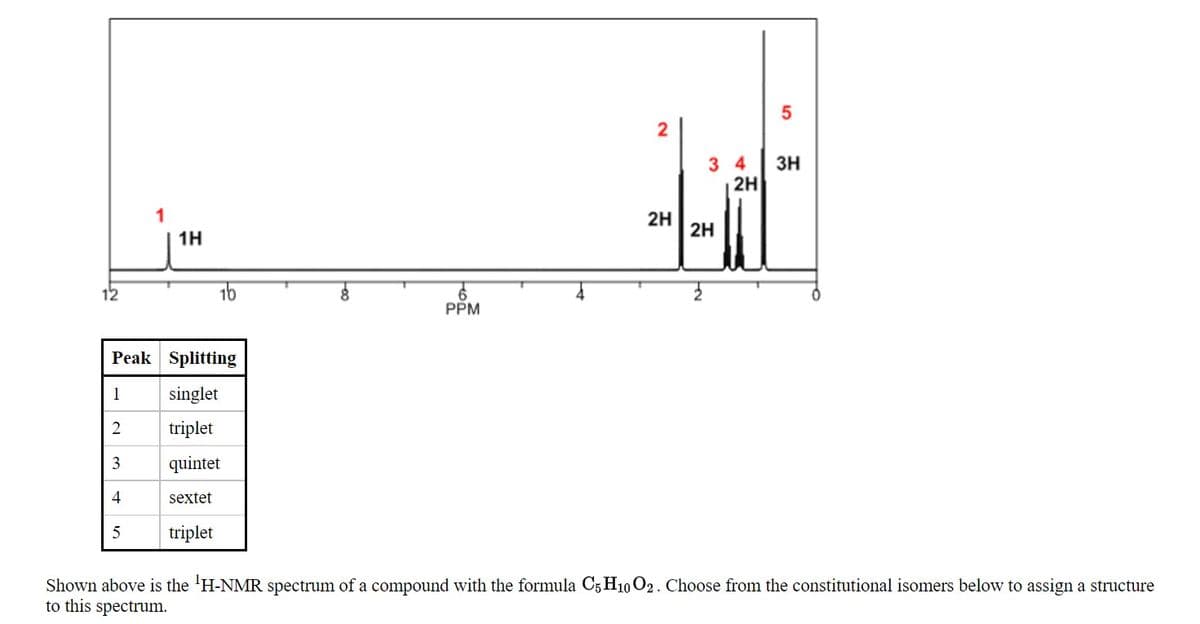
2 3 4 3H 2H 2H 2H 1H 12 16 PPM Peak Splitting 1 singlet triplet 3 quintet 4 sextet triplet Shown above is the 'H-NMR spectrum of a compound with the formula C5 H10 O2. Choose from the constitutional isomers below to assign a structure to this spectrum.
2 3 4 3H 2H 2H 2H 1H 12 16 PPM Peak Splitting 1 singlet triplet 3 quintet 4 sextet triplet Shown above is the 'H-NMR spectrum of a compound with the formula C5 H10 O2. Choose from the constitutional isomers below to assign a structure to this spectrum.
Chapter20: Carboxylic Acids And Nitriles
Section20.SE: Something Extra
Problem 56AP
Related questions
Question
i keep getting it wrong, i need help
Transcribed Image Text:3 4
3H
2H
1
2H
2H
1H
PPM
Peak Splitting
1
singlet
2
triplet
3
quintet
4
sextet
triplet
Shown above is the 'H-NMR spectrum of a compound with the formula C5 H10 O2. Choose from the constitutional isomers below to assign a structure
to this spectrum.
2.
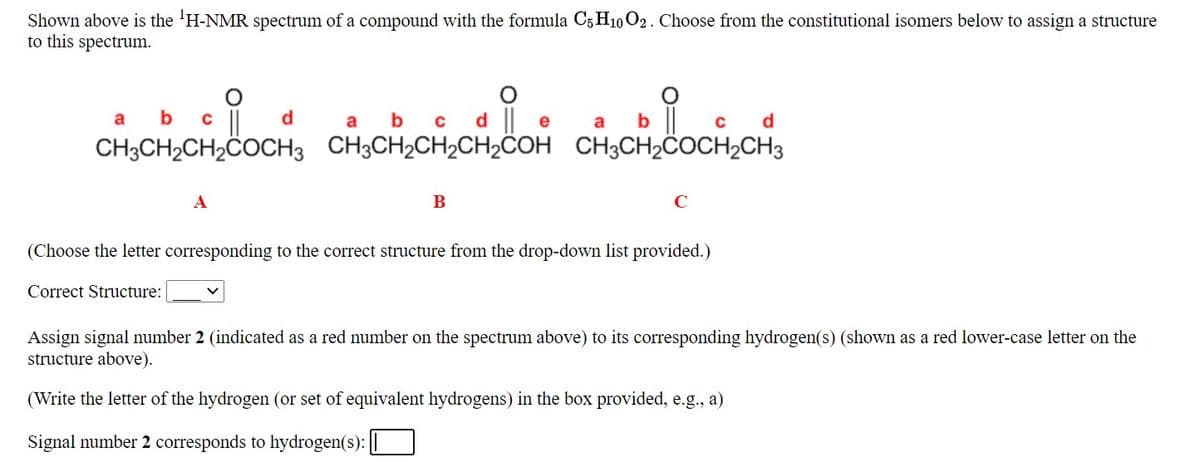
Transcribed Image Text:Shown above is the 'H-NMR spectrum of a compound with the formula C5 H10 O2. Choose from the constitutional isomers below to assign a structure
to this spectrum.
a b c ||
b c d e
CH3CH2CH2COCH3 CH3CH2CH2CH2COH CH3CH2COCH2CH3
d.
b||
d
a
a
A
B
C
(Choose the letter corresponding to the correct structure from the drop-down list provided.)
Correct Structure:
Assign signal number 2 (indicated as a red number on the spectrum above) to its corresponding hydrogen(s) (shown as a red lower-case letter on the
structure above).
(Write the letter of the hydrogen (or set of equivalent hydrogens) in the box provided, e.g., a)
Signal number 2 corresponds to hydrogen(s): ||
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

