4. The following compound possesses a molecular formula of C5H100 and the indicated 13C spectrum. A proton NMR was taken, but unfortunately the data was lost except for the fact that it showed three distinct signals: a singlet, a doublet, and a septet (7 peaks), all of which appear between 0 and 3 ppm. Please identify the compound, and then draw what would be its expected 'H NMR picture. 13C Spectrum 220 200 180 160 140 120 100 80 60 40 20 0
4. The following compound possesses a molecular formula of C5H100 and the indicated 13C spectrum. A proton NMR was taken, but unfortunately the data was lost except for the fact that it showed three distinct signals: a singlet, a doublet, and a septet (7 peaks), all of which appear between 0 and 3 ppm. Please identify the compound, and then draw what would be its expected 'H NMR picture. 13C Spectrum 220 200 180 160 140 120 100 80 60 40 20 0
Chapter13: Structure Determination: Nuclear Magnetic Resonance Spectroscopy
Section13.12: Dept 13c Nmr Spectroscopy
Problem 22P
Related questions
Question
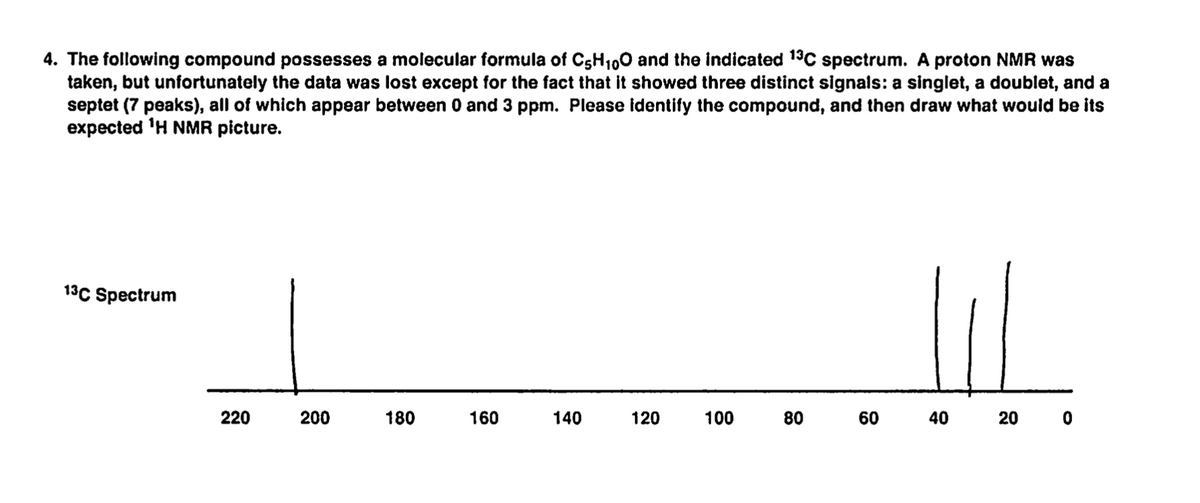
Transcribed Image Text:4. The following compound possesses a molecular formula of C5H100 and the indicated 19C spectrum. A proton NMR was
taken, but unfortunately the data was lost except for the fact that it showed three distinct signals: a singlet, a doublet, and a
septet (7 peaks), all of which appear between 0 and 3 ppm. Please identify the compound, and then draw what would be its
expected 'H NMR picture.
13C Spectrum
220
200
180
160
140
120
100
80
60
40
20 0
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

