2 of 2 G H un acid catalyst [H+], or pyridine, is present if needed. OH HO, CHOH OH HO AA BB DD EE FF GG HH Ketones, Aldehydes and Epoxides: Assume "then H,0" is included if a protonation step is needed J K A A A A U V Acid Chlorides: Assume AICI, or pyridine is present if needed w Other Reagents: 11 PCC in CH2C2 12 Na,Cr207, H2S04. H20 13 BH3 THF or 9-BBN, then H2O2, NaOH 14 Hg(OAc)2, H2O, then NaBH, 15 O3, then Zn, HCI or DMS 16 MCPBA or CH;CO,H 17 Br2, light or NBS, heat 1 H30* (dilute H2SO,) or H3O*, heat 2 conc. H2SO4, heat 21 Br2, FeBr3 22 Mg, Etz0 23 Cl2, AICI, 24 SOCI2, pyridine 25 HNO3, H2SO, 26 fuming H2SO4 27 Fe, HCI; then NaOH 3 NaOEt 4 t-BUOK 5 H2, Pt 6 H2, Lindlar's catalyst 7 Na, NH3 8 LAH or xs LAH, then H20 9 NABH4, CH,OH 10 NABH, 18 HBr 28 Zn(Hg). НCI 29 KCN, or KCN + HCN 30 CO2, then H3o* 19 HBr, ROOR pH 5 20 PBr3 OH (-H2O) 31 (H'). HOʻ 32 NH3 (1 or 2 equiv.) 33 CH3NH2 (1 or 2 equiv) 34 (CH3)2NH (1 or 2 equiv) 35 EINH2 (1 or 2 equiv) 36 PHCH,NH2 (1 or 2 equiv). 37 LDA, -78 °C 38 NaH, 25 °C Grignard, Wittig and Gilman Reagents: Assume "then H,0" is included if a protonation step is needed „MgBr MeMgBr EtMgBr PhMgBr G1 G2 G3 G4 CuLi Me,CuLi Et,CuLi (PHCH2),CuLi 39 LIAI(OR),H, then H,0 40 DIBAH, then H20 41 Brz. (H3O"] 42 Br2, NaOH 43 Pyridine G5 G6 G7 G8 MePh,P=CH2 PhyP=CHCH, PhyP=CHCO,Et PhyP=CHPH W1 W2 W3 W4
2 of 2 G H un acid catalyst [H+], or pyridine, is present if needed. OH HO, CHOH OH HO AA BB DD EE FF GG HH Ketones, Aldehydes and Epoxides: Assume "then H,0" is included if a protonation step is needed J K A A A A U V Acid Chlorides: Assume AICI, or pyridine is present if needed w Other Reagents: 11 PCC in CH2C2 12 Na,Cr207, H2S04. H20 13 BH3 THF or 9-BBN, then H2O2, NaOH 14 Hg(OAc)2, H2O, then NaBH, 15 O3, then Zn, HCI or DMS 16 MCPBA or CH;CO,H 17 Br2, light or NBS, heat 1 H30* (dilute H2SO,) or H3O*, heat 2 conc. H2SO4, heat 21 Br2, FeBr3 22 Mg, Etz0 23 Cl2, AICI, 24 SOCI2, pyridine 25 HNO3, H2SO, 26 fuming H2SO4 27 Fe, HCI; then NaOH 3 NaOEt 4 t-BUOK 5 H2, Pt 6 H2, Lindlar's catalyst 7 Na, NH3 8 LAH or xs LAH, then H20 9 NABH4, CH,OH 10 NABH, 18 HBr 28 Zn(Hg). НCI 29 KCN, or KCN + HCN 30 CO2, then H3o* 19 HBr, ROOR pH 5 20 PBr3 OH (-H2O) 31 (H'). HOʻ 32 NH3 (1 or 2 equiv.) 33 CH3NH2 (1 or 2 equiv) 34 (CH3)2NH (1 or 2 equiv) 35 EINH2 (1 or 2 equiv) 36 PHCH,NH2 (1 or 2 equiv). 37 LDA, -78 °C 38 NaH, 25 °C Grignard, Wittig and Gilman Reagents: Assume "then H,0" is included if a protonation step is needed „MgBr MeMgBr EtMgBr PhMgBr G1 G2 G3 G4 CuLi Me,CuLi Et,CuLi (PHCH2),CuLi 39 LIAI(OR),H, then H,0 40 DIBAH, then H20 41 Brz. (H3O"] 42 Br2, NaOH 43 Pyridine G5 G6 G7 G8 MePh,P=CH2 PhyP=CHCH, PhyP=CHCO,Et PhyP=CHPH W1 W2 W3 W4
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter10: Alcohols
Section: Chapter Questions
Problem 10.56P: Using your reaction roadmap as a guide, show how to convert 2-methylpentane into...
Related questions
Question
hw help 10
![Alkyl Halides (X = CI, Br or I): Assume AICI, is present if needed
2 of 2
G
un acid catalyst [H+], or pyridine, is present if needed.
он
OH
OH
CHJOH
AA
BB
DD
EE
FF
GG
HH
Ketones, Aldehydes and Epoxides: Assume "then H,0" is included if a protonation step is needed
K.
P
R
V
Acid Chlorides: Assume AICI, or pyridine is present if needed
YY
zZ
Other Reagents:
11 PCC in CH2C2
21 Br2, FeBr3
22 Mg. Et,0
23 Cl2, AICI3
24 SOCI2, pyridine
25 HNO3, H2SO4
26 fuming H2SO4
1 H3O* (dilute H2SO4) or H3O*, heat
2 conc. H2SO4, heat
3 NaOEt
12 NazCr207, H2S04, H2O
13 BH3•THF or 9-BBN, then H2O2, NaOH
14 Hg(OAc)2, H2O, then NaBH4
15 O3, then Zn, HCI or DMS
16 MCPBA or CH;CO3H
17 Br2, light or NBS, heat
4 t-BUOK
5 H2, Pt
6 H2, Lindlar's catalyst
7 Na, NH3
8 LAH or xs LAH, then H20
9 NABH4, CH3OH
10 NABH,CN, pH 5
27 Fe, HCI; then NaOH
28 Zn(Hg). НCI
29 KCN, or KCN + HCN
30 CO2, then H30*
18 HBr
19 HBr, ROOR
20 PB13
31 (H*]. HOʻ
32 NH3 (1 or 2 equiv.)
33 CH,NH2 (1 or 2 equiv)
34 (CH3)½NH (1 or 2 equiv)
35 EINH2 (1 or 2 equiv)
36 PHCH,NH2 (1 or 2 equiv).
37 LDA, -78 °C
(-H20)
Grignard, Wittig and Gilman Reagents:
Assume "then H,0" is included if a protonation step is needed
MgBr
MeMgBr
EtMgBr
PhMgBr
G1
G2
G3
G4
CuLi
Me,Culi Et,Culi (PHCH2),CULI
38 NaH, 25 °C
39 LIAI(OR);H, then H20
40 DIBAH, then H2O
41 Br2. [H3O*]
42 Br2, NaOH
43 Pyridine
G5
G6
G7
G8
MePh,P=CH2 PhyP=CHCH3 PhyP=CHCO,Et PhyP=CHPH
W1
w2
W3
W4](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2Fbc96294a-6867-4c53-8945-7b1c26d22cf9%2F66a21c83-bbc6-40e5-9f4e-7d77e3526aad%2Fayieiod_processed.jpeg&w=3840&q=75)
Transcribed Image Text:Alkyl Halides (X = CI, Br or I): Assume AICI, is present if needed
2 of 2
G
un acid catalyst [H+], or pyridine, is present if needed.
он
OH
OH
CHJOH
AA
BB
DD
EE
FF
GG
HH
Ketones, Aldehydes and Epoxides: Assume "then H,0" is included if a protonation step is needed
K.
P
R
V
Acid Chlorides: Assume AICI, or pyridine is present if needed
YY
zZ
Other Reagents:
11 PCC in CH2C2
21 Br2, FeBr3
22 Mg. Et,0
23 Cl2, AICI3
24 SOCI2, pyridine
25 HNO3, H2SO4
26 fuming H2SO4
1 H3O* (dilute H2SO4) or H3O*, heat
2 conc. H2SO4, heat
3 NaOEt
12 NazCr207, H2S04, H2O
13 BH3•THF or 9-BBN, then H2O2, NaOH
14 Hg(OAc)2, H2O, then NaBH4
15 O3, then Zn, HCI or DMS
16 MCPBA or CH;CO3H
17 Br2, light or NBS, heat
4 t-BUOK
5 H2, Pt
6 H2, Lindlar's catalyst
7 Na, NH3
8 LAH or xs LAH, then H20
9 NABH4, CH3OH
10 NABH,CN, pH 5
27 Fe, HCI; then NaOH
28 Zn(Hg). НCI
29 KCN, or KCN + HCN
30 CO2, then H30*
18 HBr
19 HBr, ROOR
20 PB13
31 (H*]. HOʻ
32 NH3 (1 or 2 equiv.)
33 CH,NH2 (1 or 2 equiv)
34 (CH3)½NH (1 or 2 equiv)
35 EINH2 (1 or 2 equiv)
36 PHCH,NH2 (1 or 2 equiv).
37 LDA, -78 °C
(-H20)
Grignard, Wittig and Gilman Reagents:
Assume "then H,0" is included if a protonation step is needed
MgBr
MeMgBr
EtMgBr
PhMgBr
G1
G2
G3
G4
CuLi
Me,Culi Et,Culi (PHCH2),CULI
38 NaH, 25 °C
39 LIAI(OR);H, then H20
40 DIBAH, then H2O
41 Br2. [H3O*]
42 Br2, NaOH
43 Pyridine
G5
G6
G7
G8
MePh,P=CH2 PhyP=CHCH3 PhyP=CHCO,Et PhyP=CHPH
W1
w2
W3
W4
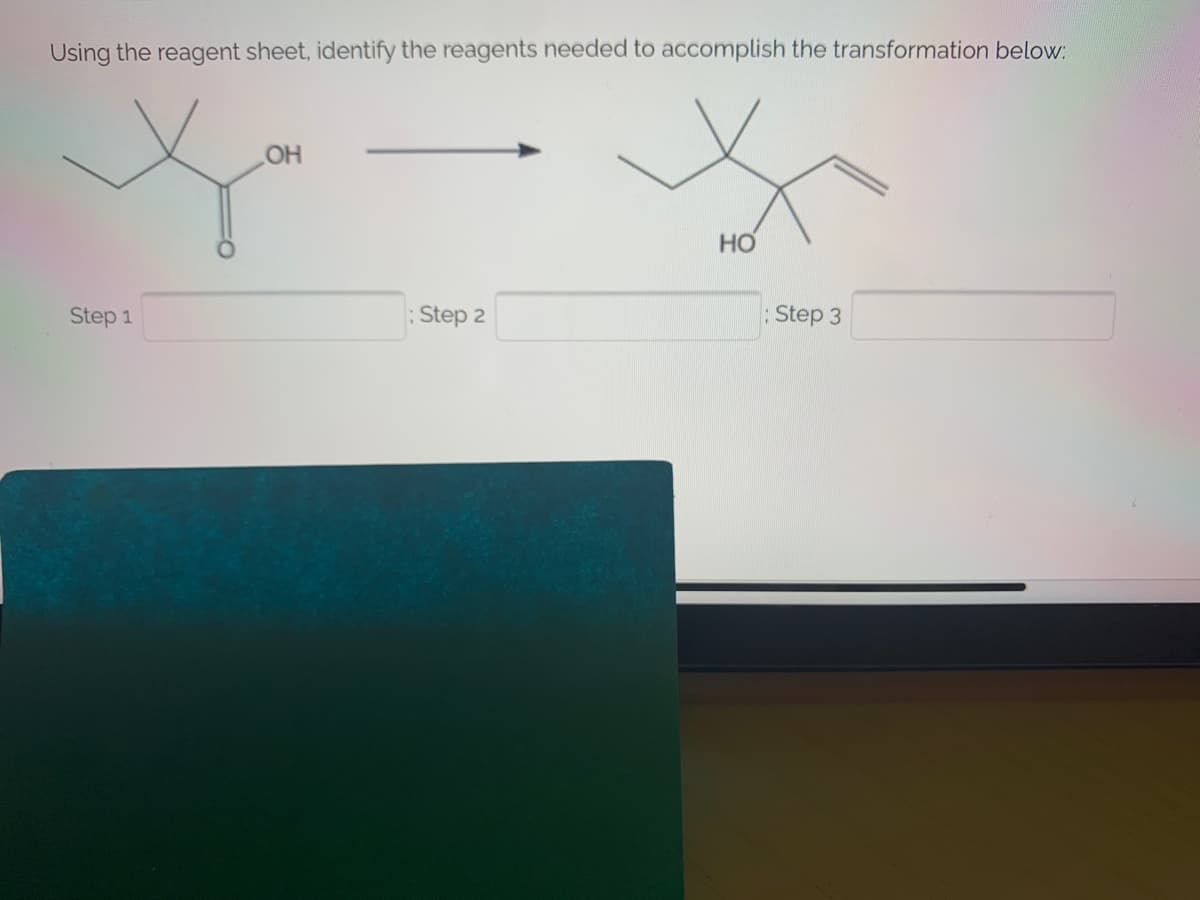
Transcribed Image Text:Using the reagent sheet, identify the reagents needed to accomplish the transformation below:
OH
HO
Step 1
Step 2
: Step 3
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning
