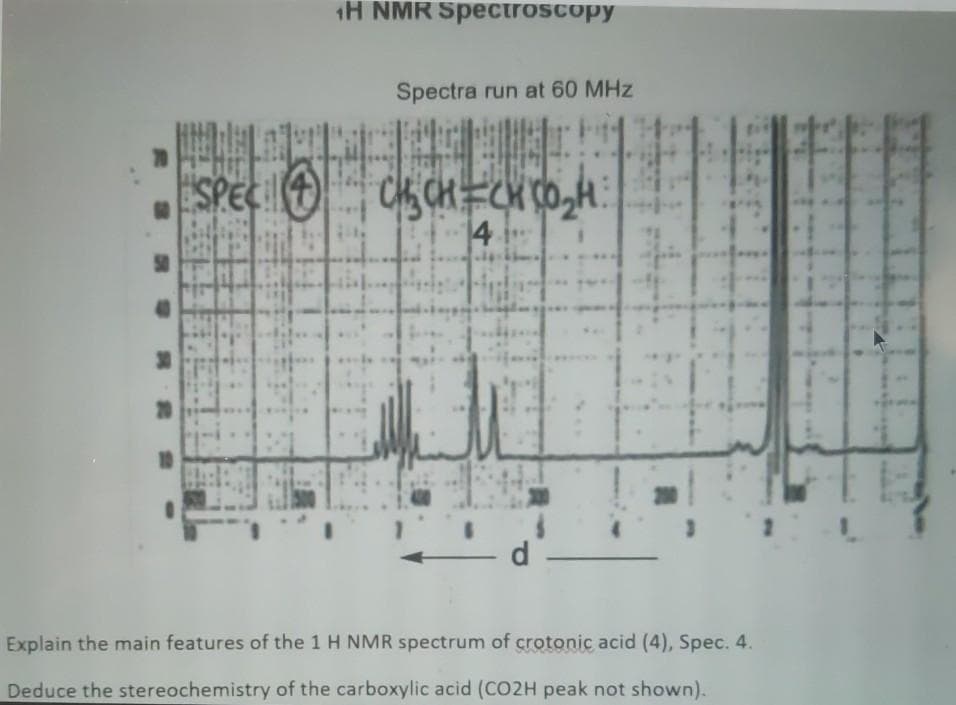
Explain the main features of the 1 H NMR spectrum of crotonic acid (4), Spec. Deduce he stereochemistry of the carboxylic acid (CO2H peak pot sbown)
Q: 2. MF Relative Intensity 100- 80 40 20 0-|rrrrrr 25 -5 50 75 m/z 100 125 150
A:
Q: (a) Predict the values of m>z and the structures of the most abundant fragments you would observe…
A: Since we are entitled to answer up to one question, we’ll answer the first question as you have not…
Q: 8 Give logical fragmentation reactions to account for the following ions observed in these mass…
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: (b) An organic compound (A), having MF CgH12O reduces Fehling solution and Tollens reagent. The…
A: A question based on mass spectrum that is to be accomplished.
Q: An IR spectrum shows a medium absorption band at 2962 cm1 and a strong absorption band at 1764 cm1.…
A: IR spectroscopy is used to identify the presence of the different functional groups in compounds.…
Q: Give logical fragmentation reactions to account for the following ions observed in these mass…
A: (a)
Q: How can 1H NMR be used to prove that the addition of HBr to propene follows the rule that says that…
A:
Q: Primary (1°) alcohols often show a peak in their mass spectra at m/z = 31. Suggest a structure for…
A: Given information, Primary alcohols often show peak in their mass spectra at m/z = 31.
Q: molecular weight of H2O is 18 but his boilling point 100C, WHY? *
A: A liquid boils when its vapor pressure becomes equal to the atmospheric pressure. Vapor pressure…
Q: The carbonyl absorptions of esters X and Y differ by 25 cm−1. Which compound absorbs at higher…
A: Stretching frequency or frequency number : It is also called vibrational frequency. Every molecule…
Q: Calculate the IHD of C7H6XNO and explain (elucidate) the structure using the 1H and 13C NMR data.
A: IHD is is index of hydrogen defficiency .It tells about how many hydrogens(H2) are required to add…
Q: Sketch the form of the 19F-NMR spectra of a naturalsample of 10BF4- and 11 BF4
A: 19F is NMR-sensitive nucleus that gives signals when placed in an external magnetic field. The…
Q: The carbonyl absorption of an amide typically occurs in the 1630–1680 cm-1 range, while the carbonyl…
A: The absorption frequency of carbonyl group of amide is less than esters.
Q: Treatment of ketone A with ethynyllithium (HC≡CLi) followed by D3O+ afforded a compound B of…
A:
Q: (b) An organic compound (A), having MF C6H₁2O reduces Fehling solution and Tollens reagent. The…
A: According to the demand, Question No. b) and d) are answered as follows:
Q: N-propylbenzene, C6H5CH2CH3, contains C (sp3) -H and C (sp2) -H bonds. Its IR spectrum shows strong…
A: A multiple choice question based on IR spectroscopy, which is to be accomplished.
Q: Why is the C¬O absorption band of 1-hexanol at a smaller wavenumber (1060 cm-1) than the…
A: In IR spectroscopy, the absorption band of C-O for carboxylic acid is higher than the absorption…
Q: Comment on the general features of the predicted (extremely simplified) 1H-NMR spectrum of lycopene…
A: 1H NMR analysis:
Q: 9. The HNMR spectrum of acetic acid is shown below. Explain why the downfield singlet is much…
A: NMR spectroscopy is a technique used to investigate the properties of compounds. It is applicable…
Q: Which of the following is used as an 1 I. internal standard in in 1H-NMR spectroscopy? O…
A: NMR spectroscopy is a very important tool for the determination of the structure of the organic…
Q: The UV-spectrum of cyclohexanone shows absorption maxima at 189 nm and What type of transition is…
A: UV spectroscopy is absorption spectroscopy which covers ultraviolet and visible regions of the…
Q: (b) Ozonolysis of 2,7-dimethyl-4-octyne gave compound E. 0 Show the reaction involved for the…
A:
Q: Compound 67 is a high-boiling liquid (boiling point 180° C) that reacts with I, in aqueous base to…
A: The structure of a chemical compound is determined by using spectroscopy. Various spectroscopy are…
Q: (b) An organic compound (A), having MF C,H120 reduces Fehling solution and Tollens reagent. The…
A: Compound A reduces the Tollens reagent. Therefore the compound is aldehyde ( see the fragmentation…
Q: hic acid absorb at 272 n. Identify the isom- ation.
A: According to the question, the UV-spectra of two isomers of cinnamic acid absorb at 272 nm and 268…
Q: 3. Sketch the ¹H-NMR spectra for the following compounds: (a) for H (b) (c) Cl oto
A: 1H- NMR spectra occurs due to no of different type of Hydrogen present in compound.
Q: 3. Deduce the structure of the molecule with the formula CaHuo with adsorption in the FTIR spectrum…
A: In this question, we have given the values of infrared spectroscopy and NMR spectroscopy. We have to…
Q: ClCH₂CO₂H , elucidate the following mass spectra and give atleast 5 fragmentations
A: A question based on mass spectrum that is to be accomplished.
Q: How could 1H NMR spectroscopy be used to distinguish among isomers A, B, and C?
A: NMR- It is type of spectroscopy called as Nuclear Magnetic resonance spectroscopy. It is…
Q: (b) An organic compound (A), having MF C,H12O reduces Fehling solution and Tollens reagent. The…
A: A question based on mass spectrum that is to be accomplished.
Q: A student adds caustic soda to some acetone and leaves it overnight. The next day they filter off…
A: Carbonyl compound (aldehyde or ketones) having α hydrogens undergo Aldol condensation reaction in…
Q: The position of UV absorption maxima of aniline in aqueous solution are different from those of…
A: Aniline contains an -NH2 group.
Q: 1) what is the struture of B, mechanism for its formation and assigment of the IR and 13C spectra…
A: Please find the attachment.
Q: Show how the Cahn-Ingold-Prelog sequence rules are used to assign the geomet either E or Z. COOH но
A: According to " Cohn ingold prelog " rule Rank the atoms directly attached to the olefinic carbon…
Q: 5. Discuss peak splitting, and draw H(proton) NMR spectra for CH3CH2CH2F structure, label and…
A: Splitting of a proton is based on (n+1) rule where n is the number of adjacent hydrogens. Chemical…
Q: Reaction of BrCH2CH2CH2CH2NH2 with NaH forms compound W, whichgives the IR and mass spectra shown in…
A: The mass and the IR spectrum acts as an aid in the quantitative as well as the qualitative analysis…
Q: A compound strong absorption in infrared spectrum near 1700 cm" and molecular ions at m/z 166 and…
A: The question is based on the concept of Spectroscopy. we have to analyse the spectra provided and…
Q: Explain how you derive the structure of curcumin by 1H NMR and 13C NMR. (Explain in detail how many…
A: Every nuclei will have spin and if an external magnetic field is applied, there will be an energy…
Q: A student performed a reaction using 2-methoxyethanol as solvent under acid catalyst. When the…
A: Given that, a student performed a reaction using 2-methoxyethanol as solvent under acid catalyst.…
Q: Propose the 1H and 13C NMR spectrum, including DEPT-90, DEPT-135 and Decoupled, of the following…
A:
Q: 1H-nmr signal at 4.2 (d) is due to * "H NMR Spectrum 200 MHz. CDCI, solution) exchanges with D,0…
A: The NMR signal of -OH occurs at around 2-3 ppm. The aromatic C-H occurs in the range of around 7-8…
Q: Find the A and T% of a 6.5X10-4 mol L-1 solution of a drug (Molecular weight 200) whose absorptivity…
A:
Q: Carboxylic acids often give a strong fragment ion at m/z (M – 17). What is the likely structure of…
A: The carboxylic acid often give a strong fragment ion at m/z(M- 17). In this statement loss of 17 it…
Q: Oxidation of citronellol, a constituent of rose and geranium oils, with PCC in the presence of added…
A:
Q: 2815 ex pesto d p NmR specba Doad and explain the
A: Let us try to understand tge spectra.
Q: Find the A and T% of a 6.5X104 mol L1 solution of a drug (Molecular weight 200) whose absorptivity…
A:
Q: Provide c mechgnism dit > Br HO Br TOOH It
A: This reaction is used to protect aldehyde and ketone from some reaction . This type of reaction is…
Q: कना १०मaडडापल श्ुल्कांवेर ( EcN ) vrल्वलङ्ड ज्क वतंवेe , व वटववाय P० 3cnu&4& भp8, सुdसमा पूुकलांवर…
A: Amount of KCN= 0.410 gram KCN(aq)+HCL(aq) ->HCN(g) + KCl(aq)
Q: Reaction of BrCH2CH2CH2CH2NH2 with NaH forms compound W, which gives the IR and mass spectra shown…
A:
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

- The carbonyl absorption of an amide typically occurs in the 1630–1680 cm-1 range, while the carbonyl absorption of an ester occurs at much higher wavenumber, typically 1735–1745 cm-1. Account for this difference.Fully interpret the 1H-NMR spectrum of 4-vinylbenzoic acid providedPropose a structure for the molecule with the formula of C5H10O from the H NMR, C NMR, IR and mass spectrometry spectrums.
- Based on the 13C NMR data provided with multiplicity, deduce the structures of each of the benzenoid aromatic compounds below, where the molecular formula for compound is C10H14. a. 134(s), 131(d), 19(q)3 The 1H- and 13C-NMR data of an ester of molecular formula C6H10O2 are given below. Also shown are the COSY and HETCOR NMR spectra of the ester. Identify the ester, explaining how you reach your conclusion. 1H-NMR: 7.20-6.90 (1H), 5.85 (1H), 4.16 (2H), 1.88 (3H), 1.31 (3H) ppm 13C-NMR: 166.7, 144.5, 123.0 , 60.2, 18.0, 14.3 ppmTreatment of butan-2-one (CH3COCH2CH3) with strong base followed byCH3I forms a compound Q, which gives a molecular ion in its massspectrum at 86. The IR (> 1500 cm−1 only) and 1H NMR spectra of Q aregiven below. What is the structure of Q?
- Propose the 1H and 13C NMR spectrum, including DEPT-90, DEPT-135 and Decoupled, of the following structures.Following is a 1H-NMR spectrum of2-butanol, Explain why the CH2 protons appear as a complex multiplet rather than as a simple quintet.(a) The 'H-NMR spectrum of cyclobutanone shows two signals - signal A at 3.00 ppm and signal B at 1.95 ppm. Give the multiplicity of each signal. cyclobutanone (b) When cyclobutanone is treated with D20 and NaOD, the only signal observable in the 1H-NMR is a singlet at 2.00 ppm. Explain why this is the case. [Note: Deuterium atoms do not display signals in the TH-NMR spectrum]
- Predict the 13C-NMR spectrum of methyl orange in DMSO-d6. How many signals will beobserved and in which range?ClCH₂CO₂H , elucidate the following spectra bellowAn organic compound B with formula C6H14O has the following: IR Spectroscopy 2974 cm-1, 1080 cm-1 Mass Spectrometry 102 (M+), 87, 73 1H NMR Spectroscopy Eight signals at δ 1.10 (d, 3H), 1.13 (dd, 3H), 1.14 (dd, 3H), 1.59 (ddq, 1H), 1.60 (ddq, 1H), 3.19 (ddq, 1H), 3.51 (dq, 1H), 3.50 (dq, 1H). Compound B is obtained by the reaction of compound A with NaH followed by CH3CH2Br. The stereochemistry of A is "S" Using this information, deduce a plausible structure for Compound A with correct stereochemistry.

