2 steps BU4NF C9H1602 A + B ÓSIME,tBu (i) Enone C can be prepared from alkyne A and aldehyde B in two steps via a gold-catalysed reaction. Identify A and B and provide reagents/conditions for a synthesis of C. (ii) Give the structure of D and classify the ring-closing reaction for its formation from C according to Baldwin's rules
2 steps BU4NF C9H1602 A + B ÓSIME,tBu (i) Enone C can be prepared from alkyne A and aldehyde B in two steps via a gold-catalysed reaction. Identify A and B and provide reagents/conditions for a synthesis of C. (ii) Give the structure of D and classify the ring-closing reaction for its formation from C according to Baldwin's rules
Chapter16: Chemistry Of Benzene: Electrophilic Aromatic Substitution
Section16.SE: Something Extra
Problem 45MP
Related questions
Question
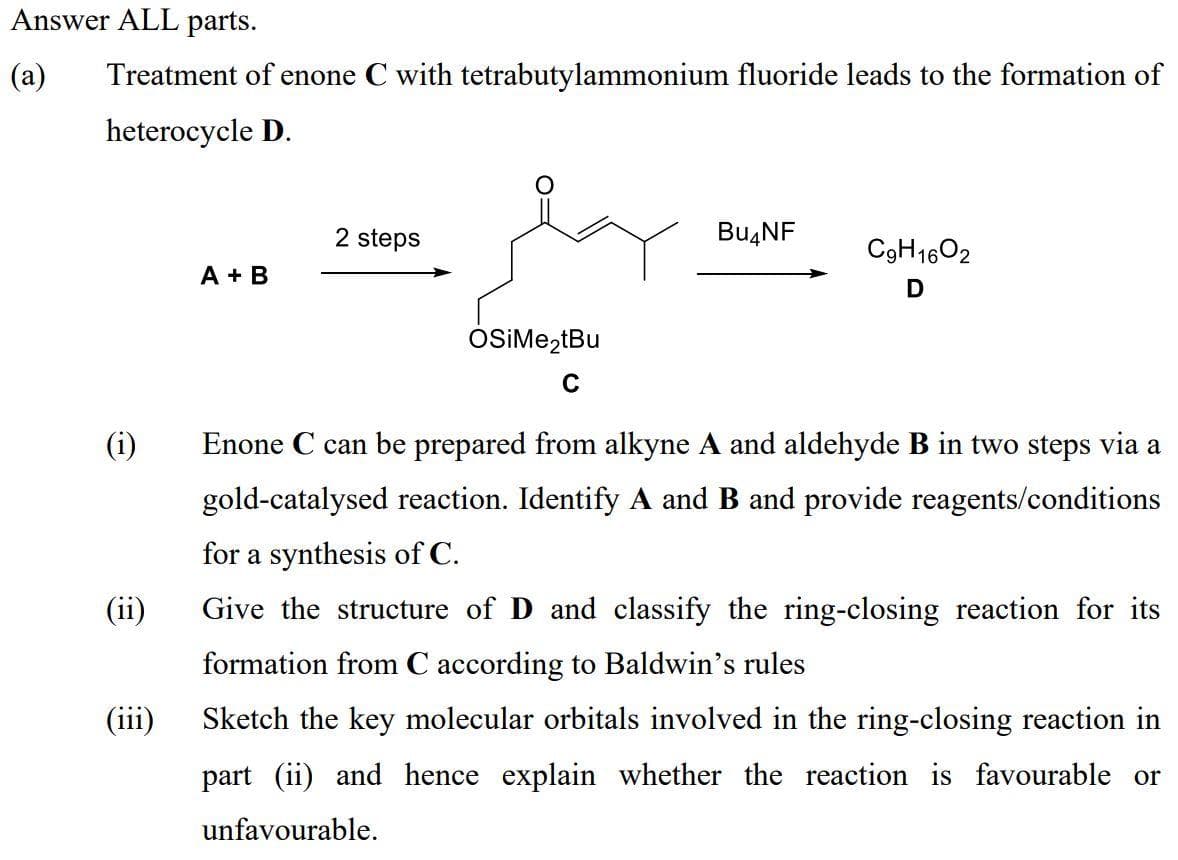
Transcribed Image Text:Answer ALL parts.
(a)
Treatment of enone C with tetrabutylammonium fluoride leads to the formation of
heterocycle D.
2 steps
BU4NF
C9H1602
A + B
D
OSIME,tBu
(i)
Enone C can be prepared from alkyne A and aldehyde B in two steps via a
gold-catalysed reaction. Identify A and B and provide reagents/conditions
for a synthesis of C.
(ii)
Give the structure of D and classify the ring-closing reaction for its
formation from C according to Baldwin's rules
(iii)
Sketch the key molecular orbitals involved in the ring-closing reaction in
part (ii) and hence explain whether the reaction is favourable or
unfavourable.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

