2. Fermentation is a complex chemical process of wine making in which glucose is converted into ethanol and carbon dioxide: CgH1206- → 2C,H;OH + 2CO, Starting with 500.4 g of C,H,,0, what is the maximum amount of ethanol (in grams) that can be obtained by the process? How much of C,H,„O, would produce 100 g of C,H;OH?
2. Fermentation is a complex chemical process of wine making in which glucose is converted into ethanol and carbon dioxide: CgH1206- → 2C,H;OH + 2CO, Starting with 500.4 g of C,H,,0, what is the maximum amount of ethanol (in grams) that can be obtained by the process? How much of C,H,„O, would produce 100 g of C,H;OH?
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter3: Chemical Reactions
Section: Chapter Questions
Problem 81QRT: Write the balanced chemical equation for the complete combustion of adipic acid, an organic acid...
Related questions
Question
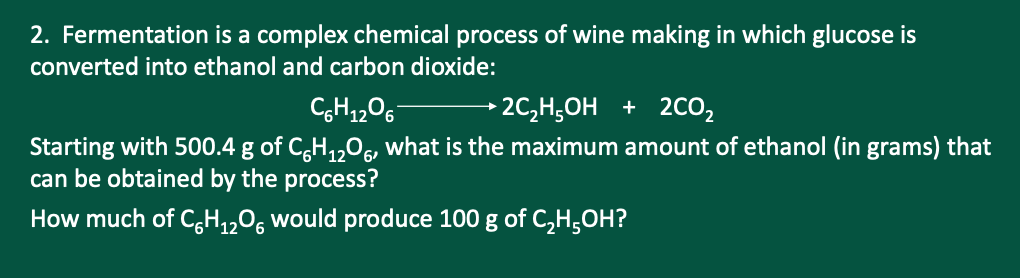
Transcribed Image Text:2. Fermentation is a complex chemical process of wine making in which glucose is
converted into ethanol and carbon dioxide:
→2C,H;OH + 2CO2
Starting with 500.4 g of C,H,,0, what is the maximum amount of ethanol (in grams) that
can be obtained by the process?
How much of C,H,,0, would produce 100 g of C,H,OH?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning


Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning


Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning