2. If we plot the first ionization energies vs. atomic number for the main group elements, we would have the following trend: 2500 Periodicity of 1st lonisation Energy for Periods 14 (and start of Period 5) 2250 (c) doc b 2000 1750 1500 1250 1000 750 500 250 Atomic number and symbol 123 45 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 37 38 H He i Be BCN O F Ne Na Mg Al SI PSaAr K Ca Sc Ti v G Mn fe Co NCu Zn Ga Ge As Se Br Kr Rb Sr Figure 2.7 Plot of the first ionization energies of the first 38 elements against its atomic s oft number Source: https://socratic.org/questions/how-does-atomic-number-affect-ionization-energRY By referring to Figure 2.7, answer the following questions: 1. What group of elements corresponds to the peaks of ionization energy on your graph? Where is this group located on the periodic table? 2. What is the trend in the first ionization energies as the atomic number increases within a group of elements? 3. What is the trend in the first ionization energies as the atomic number increases across a period or row of elements? D Focus 1st IE in kl/mol
2. If we plot the first ionization energies vs. atomic number for the main group elements, we would have the following trend: 2500 Periodicity of 1st lonisation Energy for Periods 14 (and start of Period 5) 2250 (c) doc b 2000 1750 1500 1250 1000 750 500 250 Atomic number and symbol 123 45 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 37 38 H He i Be BCN O F Ne Na Mg Al SI PSaAr K Ca Sc Ti v G Mn fe Co NCu Zn Ga Ge As Se Br Kr Rb Sr Figure 2.7 Plot of the first ionization energies of the first 38 elements against its atomic s oft number Source: https://socratic.org/questions/how-does-atomic-number-affect-ionization-energRY By referring to Figure 2.7, answer the following questions: 1. What group of elements corresponds to the peaks of ionization energy on your graph? Where is this group located on the periodic table? 2. What is the trend in the first ionization energies as the atomic number increases within a group of elements? 3. What is the trend in the first ionization energies as the atomic number increases across a period or row of elements? D Focus 1st IE in kl/mol
Chapter28: Atomic Spectroscopy
Section: Chapter Questions
Problem 28.4QAP
Related questions
Question
100%
1.What group of elements corresponds to the peaks of ionization energy on your graph? Where is this group located on the periodic table?
2.What is the trend in the first ionization energies as the
3.What is the trend in the first ionization energies as the atomic number increases across a period or row of elements?
Transcribed Image Text:AutoSave O ff
Periodic-Properties-of-Elements (1).docx - Protected View - Saved to this PC
P Search
Sophia Marie Pineda SM
File
Home
Insert
Draw
Design
Layout
References
Mailings
Review
View
Help
QuillBot
A Share
P Comments
i) PROTECTED VIEW Be careful-files from the Internet can contain viruses. Unless you need to edit, it's safer to stay in Protected View.
Enable Editing
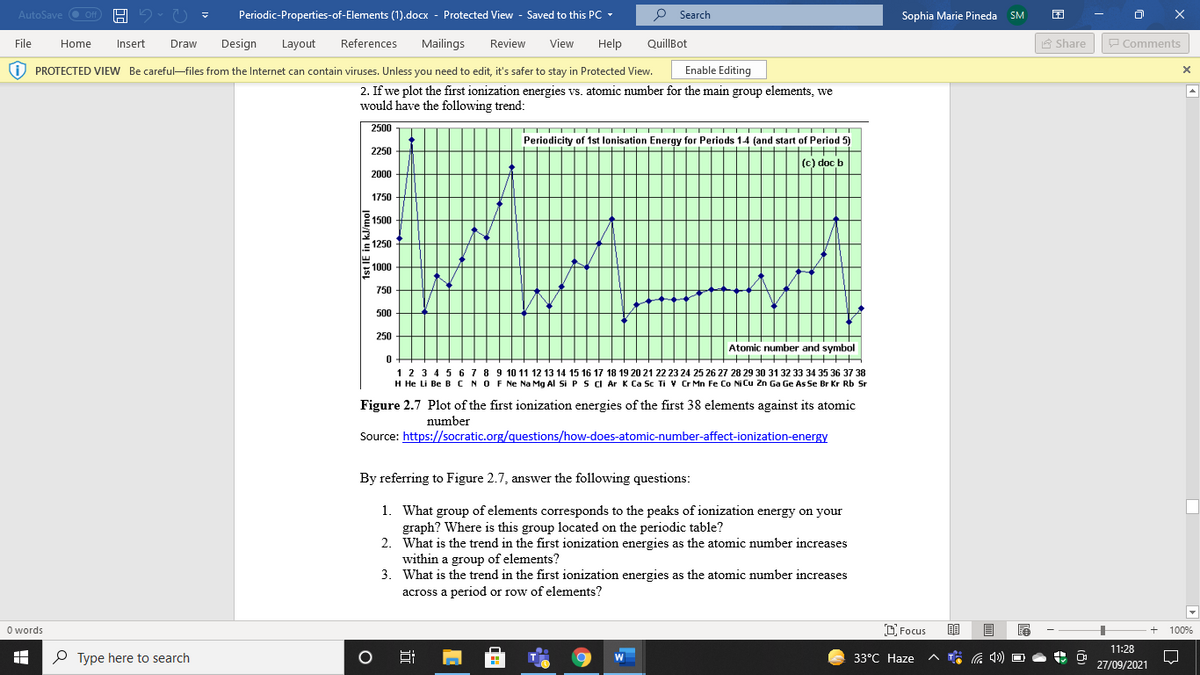
2. If we plot the first ionization energies vs. atomic number for the main group elements, we
would have the following trend:
2500
TITT TTTTT TTT TTITTTIIT TTITT
Periodicity of 1st lonisation Energy for Periods 14 (and start of Period 5)
++++
(c) doc b
2250
2000
1750
1500
1250
1000
750
500
250
Atomic number and symbol
12 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 37 38
H He Li Be B CNO FNe Na Mg Al SIPS CI Ar K Ca Sc Ti V Cr Mn Fe Co NiCu Zn Ga Ge As Se Br Kr Rb Sr
Figure 2.7 Plot of the first ionization energies of the first 38 elements against its atomic
number
Source: https://socratic.org/questions/how-does-atomic-number-affect-ionization-energy
By referring to Figure 2.7, answer the following questions:
1. What group of elements corresponds to the peaks of ionization energy on your
graph? Where is this group located on the periodic table?
2. What is the trend in the first ionization energies as the atomic number increases
within a group of elements?
3. What is the trend in the first ionization energies as the atomic number increases
across a period or row of elements?
O words
D Focus
100%
11:28
P Type here to search
33°C Haze
27/09/2021
1st IE in k/mol
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

