2. The next few questions will ask you to explain the following properties for these three different elements: Na(s), Si(s), and Br2(1). Helium (He) Nitrogen (N2) Sodium (Na) Silicon (Si) Phase at Diamond Graphite room temp (298K). mp mp 370 K grey, shiny, malleable and mp 1687 K mp about 4300K Hard, brittle, translucent, does not conduct mp 3873 K Soft, slippery, grey, shiny, conducts Other nitrogen is liquid between 63 K and 77 K properties (if any) grey, shiny, hard and ductile and brittle, can be chipped. conducts electricity electricity electricity Bonding present Would a
2. The next few questions will ask you to explain the following properties for these three different elements: Na(s), Si(s), and Br2(1). Helium (He) Nitrogen (N2) Sodium (Na) Silicon (Si) Phase at Diamond Graphite room temp (298K). mp mp 370 K grey, shiny, malleable and mp 1687 K mp about 4300K Hard, brittle, translucent, does not conduct mp 3873 K Soft, slippery, grey, shiny, conducts Other nitrogen is liquid between 63 K and 77 K properties (if any) grey, shiny, hard and ductile and brittle, can be chipped. conducts electricity electricity electricity Bonding present Would a
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter9: Liquids, Solids, And Materials
Section9.9: Metals, Semiconductors, And Insulators
Problem 9.16CE: Look in Appendix D and compare the electron configurations shown there with the fusion enthalpies...
Related questions
Question
Please help answer this
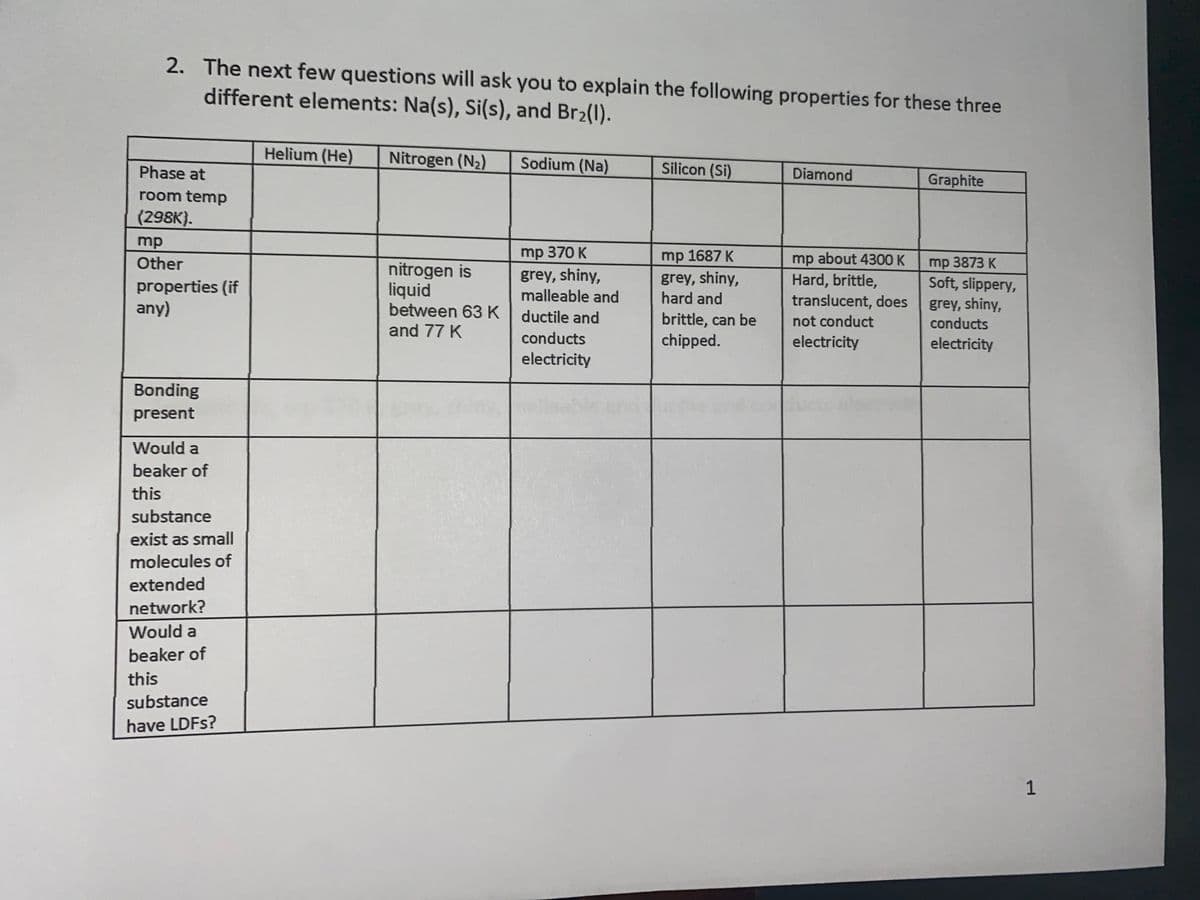
Transcribed Image Text:2. The next few questions will ask you to explain the following properties for these three
different elements: Na(s), Si(s), and Br2(1).
Helium (He)
Nitrogen (N2)
Sodium (Na)
Phase at
Silicon (Si)
Diamond
Graphite
room temp
(298K}.
mp
Other
mp 370 K
grey, shiny,
malleable and
between 63 K ductile and
mp 1687 K
about 4300 K
mp
Hard, brittle,
translucent, does
mp 3873 K
Soft, slippery,
grey, shiny,
conducts
properties (if
any)
nitrogen is
liquid
grey, shiny,
hard and
brittle, can be
chipped.
not conduct
and 77 K
conducts
electricity
electricity
electricity
Bonding
present
Would a
beaker of
this
substance
exist as small
molecules of
extended
network?
Would a
beaker of
this
substance
have LDFS?
1
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning