2. What are the mystery solutions? (remember mystery solutions 1-5 correspond to A9NO3, OCOC12; Cu(NO3)2, NaBr, KOH. In your answer write which one compound is in mystery solution #1, #2 etc. There are no repeats.) BIU E E E E 8 O fx # 1 #2 AGNO3 # 3 # 4 KOH
2. What are the mystery solutions? (remember mystery solutions 1-5 correspond to A9NO3, OCOC12; Cu(NO3)2, NaBr, KOH. In your answer write which one compound is in mystery solution #1, #2 etc. There are no repeats.) BIU E E E E 8 O fx # 1 #2 AGNO3 # 3 # 4 KOH
Chapter28: Atomic Spectroscopy
Section: Chapter Questions
Problem 28.13QAP
Related questions
Question
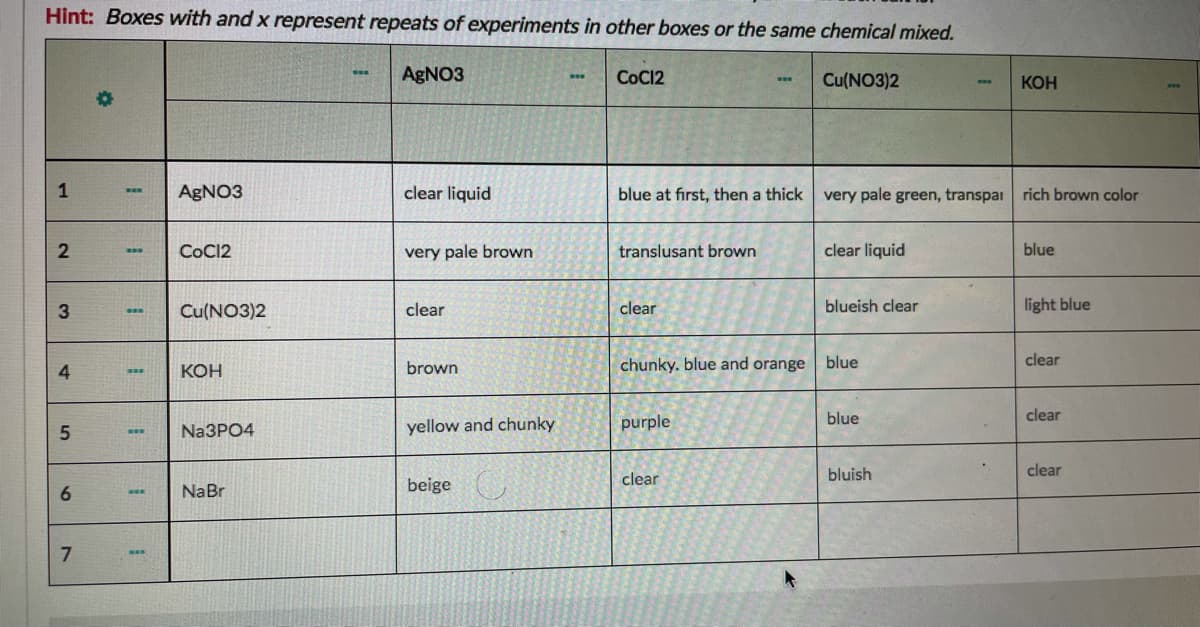
Transcribed Image Text:Hint: Boxes with and x represent repeats of experiments in other boxes or the same chemical mixed.
AGNO3
COC12
Cu(NO3)2
...
КОН
...
1
AGNO3
clear liquid
blue at first, then a thick
very pale green, transpai rich brown color
COCI2
very pale brown
translusant brown
clear liquid
blue
...
3
Cu(NO3)2
clear
clear
blueish clear
light blue
brown
chunky. blue and orange blue
clear
КОН
yellow and chunky
purple
blue
clear
Na3PO4
bluish
clear
clear
6.
NaBr
beige
7
2.
4,
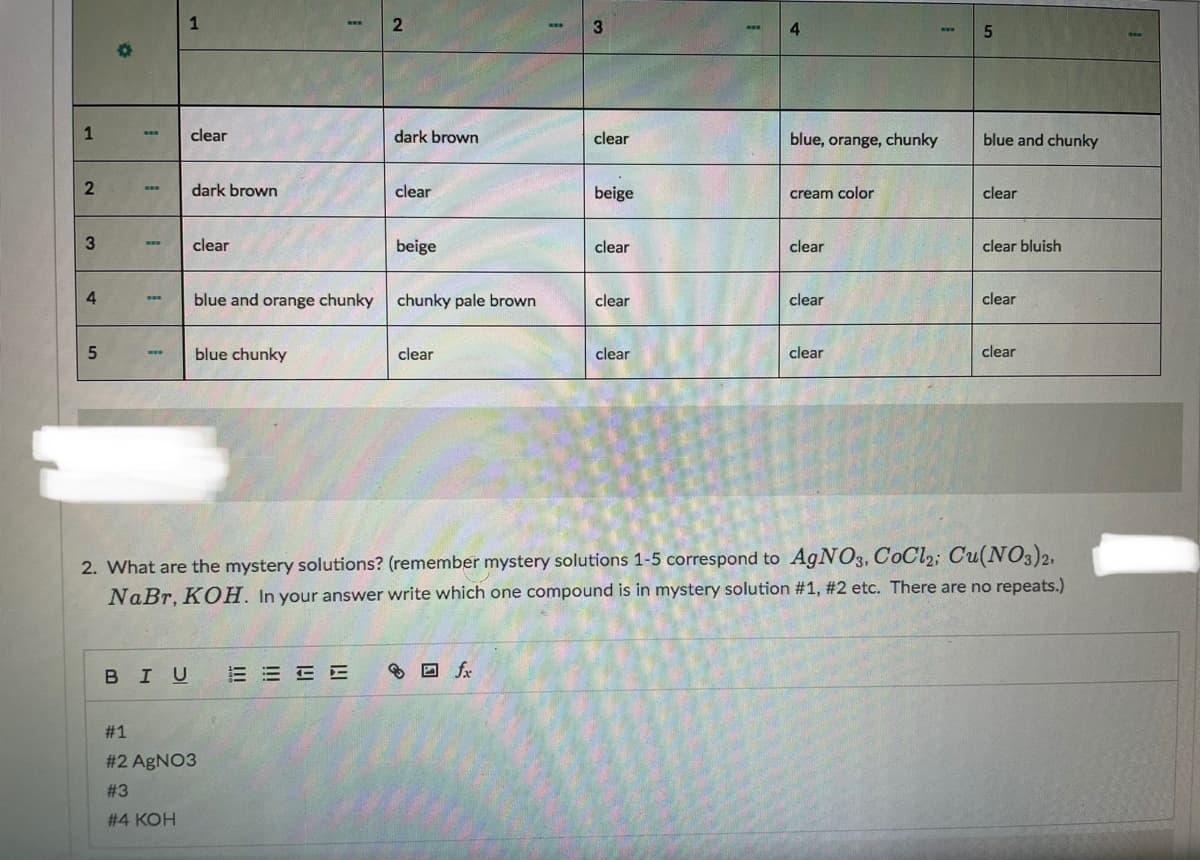
Transcribed Image Text:1
4
clear
dark brown
...
clear
blue, orange, chunky
blue and chunky
dark brown
clear
beige
cream color
clear
3.
clear
beige
clear
clear
clear bluish
4
blue and orange chunky
chunky pale brown
clear
clear
clear
ww.
blue chunky
clear
clear
clear
clear
2. What are the mystery solutions? (remember mystery solutions 1-5 correspond to A9NO3, CoCl2; Cu(NO3)2,
NaBr, KOH. In your answer write which one compound is in mystery solution #1, #2 etc. There are no repeats.)
BIU
E = E E
8 D fx
# 1
# 2 ABNO3
#3
# 4 KOH
1.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning