Introduction to General, Organic and Biochemistry
11th Edition
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Chapter5: Gases, Liquids, And Solids
Section: Chapter Questions
Problem 5.68P
Related questions
Question

Transcribed Image Text:PART B: Predict the product: Please answer 3 of the following 5 questions (4, 5, 6, 7,
8). If all questions are attempted, only the first 3 will be araded for marks.
Please indicate the questions you would like graded
Provide the products that complete the following reaction equations. Note: unless
otherwise indicated, provide the major organic products of the reactions. Please include
stereochemistry (R/S, EIZ) where applicable.
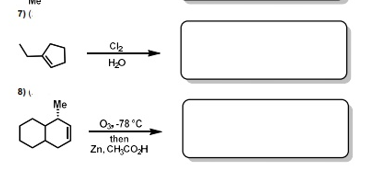
Transcribed Image Text:7) (
Ch
HO
8)
Me
O-78 °C
then
Zn, CH,COH
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning