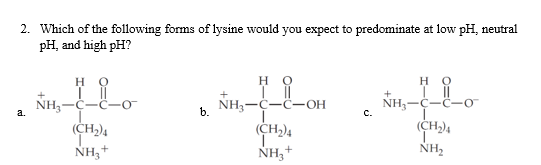
2. Which of the following forms of lysine would you expect to predominate at low pH, neutral pH, and high pH? но но NH,-C-C-o NH3-C-C-OH NH,-C-C (CH)4 a. b. с. NH,+ NH,+ NH,
Q: OH HO HO A NH2 Но. HO R-O. HO. HO HO R-O, OH Но HO `OH OH Но R-O, D HO. но но HO Но `OH OH B но. `OH…
A: Glycosidic bond is a type of covalent bond formed after reaction between hydroxyl group of…
Q: 1. What is the distinguishing structural feature of O-glycans? 2. What other prominent class of…
A: O-Linked glycoproteins are typically big proteins with molecular masses more than 200 kDa.…
Q: 2. | Calculate the overall charge (pH 7) on the following three polypeptides and answer the…
A: Proteins or peptides are composed of twenty standard amino acids that differ from each other on the…
Q: 5. Which of the following names best describes the molecule? CH2OH C=0 но- H- H- -OH ČH,OH A.…
A: We'll answer the first question since the exact one wasn't specified. Please submit a new question…
Q: 6) What is the cognate a-keto acid of alanine? You may have to look at alanine.
A: Hello. Since your question has multiple parts, we will solve the first question for you. If you want…
Q: 1. Indicate whether each of the following molecules is an alpha amino acid or not and explain why.…
A: Proteins are defined as the polymers of amino acids. It is a macronutrient that plays a very…
Q: 4. Draw the formula of Ala-Cys at pH = pl a) b) pH > pl c) pH < pl (Ala: pK.(COOH)= 2,34, pK.(NH3")…
A: Ala-Cys is a dipeptide. Alaninne has R= CH3 but cysteine has R= CH2-SH.
Q: [2]6. If the mobile phase was changed from 7:3 hexane/acetone to 1:1 hexane/acetone how would you…
A: RF value is the ratio of distances moved by the solute and solvent.
Q: 3. Describe the function of each of the following molecules/cofactors:
A: a. ATP is made up of adenosine and three phosphate. b. NAD+ is nicotinamide adenine dinucleotide. It…
Q: 7) Place the following in correct sequence from simplest to most complex: 1. molecules
A: Atoms are the smallest particles, two or more of which combine to form molecules. Many molecules…
Q: Polypeptides beginà end are: a) 5’ à 3’ b) 2’ à 5’ c) CO2H à NH2 d) NH2 à CO2H
A: Peptides are short chains of two and fifty amino acids, linked by peptide bonds. Chains of fewer…
Q: 1. Give the properties of a colloidal system. What makes them different compared to suspensions and…
A: Colloids are formed when one substance is dispersed through another but does not combine to form a…
Q: 3. Draw the chemical structure of a triacylglycerol with lauric acid, palmitoleic acid and stearic…
A: Fats are an important nutrient for healthy cell and normal cellular functioning. The fats are stored…
Q: 2. Classify the following alkaloid according to ring system, draw the structure, then give the…
A:
Q: 5. Which of the following structure is the not stable structure of hexoses? 6 Он ОН4 HO HO. 5 HO- 2…
A: In the hexose structure or any structure when the position of OH group is in equatorial postion it…
Q: sub= 18 help
A: The reaction given in the question involves the conversion of dihydroxyacetone phosphate to…
Q: 1. For this compound to cyclize, an alcohol group must attack a carbonyl group. Which carbon is…
A: A monosaccharide often switches from the acyclic form to a cyclic through a nucleophilic addition…
Q: (9 Why does DNA contain thymine instead of uracil? Justify.
A: Given: DNA contain Thymine instead of Uracil. DNA is a long polymer of polynucleotide chain. It…
Q: 4. Lipids can also be detected by a preliminary translucent spot test, also called the paper bag…
A: A translucent spot test is a qualitative assay for the determination of fats and oils. When the…
Q: 16. Which of the following choices correctly describes the molecular structure of a..ova A. There…
A: Biomolecules are organic molecules made up of mainly carbon and hydrogen, but other elements are…
Q: CHO CH,OH HO H. OH OH H- OH CH,OH HO H- но H. OH H- OF OH OH CH-OH CH,OH III Which of these are…
A: Sugar also known as carbohydrates in biochemistry are the polyhydroxy compounds of aldehydes and…
Q: 1. Draw the condensed structural formulas for A and B. [0] [0] CH3 A B CH,-C I-CH,-C-OH
A: We are authorized to answer one question at a time since you have not mentioned which question you…
Q: 1. Discuss the difference between an ⍺-D-glucose and a β-D-glucose. How can the structure of a…
A: Monosaccharides having 5 or 6 carbons in the chain gives cyclic structure in aqueous solution via…
Q: 3. Which of the following is a disaccharide? A. CH12O6 B. CH1O5 C. C12H22O11 D. C3H,O3
A: Carbohydrates are classified into monosaccharides, disaccharides, and polysaccharides.…
Q: 6. Which of the following acts as lipid-soluble radical trapping antioxidants? A. Ascorbate B. Uric…
A: There are two types of radical trapping antioxidants:- Water soluble lipid soluble
Q: Why do the pH and protease rankings differ between fresh fruits but are similar for cooked fruits?
A: Protease : It is an enzyme that catalyses the breakdown of proteins into smaller polypeptides or…
Q: 4. What is the total charge of lysine in the strong acidic medium? A) +2 B) -2 C) +1 D) 0 E) -1
A: Proteins are known as the muscle-building substances of the body. They are very complex…
Q: Average net charge of +2 predominates: а. b. The predominant species is…
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the question and…
Q: One of the triacylglycerols found in corn oil contains palmitic acid, linoleic acid, and linolenic…
A: Fatty acids are the simplest form of lipids with a long hydrocarbon chain and a carboxylic acid…
Q: 10. Write the chemical description of the disaccharide shown ? (composition, glycosidic bond,…
A: Disaccharides: Two Monosaccharides when joined together it is called Disaccharides. They are joined…
Q: 2. Draw the structure of pentapeptide Trp-Asp-Cys-Lys-Gln that would predominate in aqueous solution…
A: .
Q: 2. Which of the following molecules contain beta glycosidic linkage? A. Amylose B. Glycogen C.…
A: Cellulose is a structural polysaccharide in plants. It is made up of glucose monomers.
Q: 3. What are the effects of the following solvents when mixed with lipids? What type of hydrolysis is…
A: The lipids are the chemical substances or macromolecules that are usually insoluble in water but…
Q: 1. Write the reversible reactions with its corresponding enzymes of Dehydration via Hydrolysis…
A: Biomolecules are the biological molecules that are present inside the living organisms. These…
Q: Explain why lipids are insoluble in polar solvents. 2. Aside from their sources and physical states…
A: Lipids: Lipids are a heterogeneous group of naturally occurring organic compounds that include…
Q: 8. What kind of linkage is shown in the disaccha ОН ОН OH HO НО OH ÓH Α. α (1> 4) B. a (1 → 6) С.…
A: Saccharide is the basic structural unit of complex sugars or carbohydrates. These sugars or…
Q: 3. Which statement(s) correctly describe this disaccharide? CH2OH CH2OH A. It's the alpha anomer,…
A: Carbohydrates are classified into different types based upon the no of carbon atoms and they are…
Q: Which of the following compounds would form Azo-dye from primary aromatic amines? Indicate YES or NO…
A: Azo dyes are generally synthetic compounds that has an azo bond and Primary aromatic amines are…
Q: 1. For the test for sulfur in egg albumin, would you conclude that sulfur is present on it? Explain.…
A: Introduction Egg albumin consists of major water with proteins followed by minor amounts of…
Q: 3. Fill in the missing molecules or required cofactors. PLP b) @NH₂ OH
A: Hi! Thank you for the question. We are authorized to answer three subparts at a time, since you have…
Q: 1. At pH 7, the net charge of lysine is А. -2 В. -1 С. 0 D. +1 Е. +2
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: 6. What functional groups are present in the following molecul HạN. OH NH. A) amine and carboxylic…
A: There are several functional groups that can be found in a molecule. Some examples of functional…
Q: 2. How is the solubility of albumin in each organic solvent?
A: Solubility of albumin in each organic solvent is mentioned below:- The strongly polar,protic…
Q: Differentiate the following in terms of proteins' structural features in living systems? - primary…
A: Introduction: Proteins are biopolymers made up of twenty L-alpha amino acids linked by peptide…
Q: 1. What are the differences between terpenes and terpenoids? 2. Classify the following terpenes,…
A: The plants synthesize a variety of organic molecules. They are of two types, primary and secondary…
Q: 4. Which of the following formulae of fatty acids represents saturated fatty acid? (i) Palmitic…
A: Fatty acids are the components of lipids. Fatty acids are composed of a hydrocarbon chain and a…
Q: Which of the following alters proteins and membranes? Alcohols O lodine O All other choices O…
A: Many critical roles in the body are performed by certain complex molecules called proteins. For the…
Q: 9. Write the tripeptide structure for: val-ser-cys. 10. Write the reactions for: phe + ser
A: Amino acids are the building blocks of proteins. whereas each amino acid consists of an amino group,…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

- 1. The main sweetener in Mackies Honeycomb is _______________1. Draw the structures of the 20 biologically active AA. Under each structure, write the name, one-letter code, and three-letter code followed by the AA classification based on polarity.1. What is the distinguishing structural feature of O-glycans? 2. What other prominent class of glycoproteins besides mucins is known for the presence of O-linked glycans?
- 4. What is the region/point where AA is predominantly present as a (-2)-charged species?5. The effective buffering range for the amino acid in the acidic region is?6. What is the region/point where the solution has a 50:50 percent mixture of the (0) and the (-1) species?1. Which of the following forms of lysine would you expect to predominate at low pH, neutral pH and high pH? 2. One of the 20 amino acids is unusual in that its side chain contains a ring that incorporates the amine functional group of the amino acid. What is the name of this amino acid? Write the chemical structure of this amino acid at physiological pH.1. What is the possible identity of the AA? a. Aspartic acid b. Lysine c. Alanine d. Proline 2. What is the isoelectric point of AA? 3. What is the pKa responding to the dissociation of the alpha-carboxylic group?
- 1. The chromatography solvent is very polar as it contains alcohol, an acid and water. Based on this information, list all the polar amino acids and arrange them from most polar to least polar.3. One of the triacylglycerols found in corn oil contains palmitic acid, linoleic acid, and linolenic acid. During the production of spreadable margarine, all of the double bonds in this triacylglycerol are converted from cis to trans isomers. Linoleic acid: CH3(CH2)4CH=CHCH2CH=CH(CH2)7COOHLinolenic acid: CH3CH2CH=CHCH2CH=CHCH2CH=CH(CH2)7COOH Palmitic acid: CH3(CH2)14COOH a. Identify the unsaturated fatty acid(s) listed above and provide the associated omega designation. b. Draw an accurate representation of the structure of the triacylglycerol present in spreadable margarine. Circle all ester bonds in the structure you have drawn.1. a. Explain why the melting point of palmitic acid (16 carbons, no double bonds) is slightly lower thanthat of stearic acid (18 carbons, no double bonds). Explain why the melting point of oleic acid (18carbons, one double bond) is lower than that of stearic acid b. A mixture of lipids containing phosphatidic acid, cholesterol, testosterone, phosphatidylserine, andphosphatidylethanolamine was applied to a hydrophobic interaction chromatography column. Thecolumn was washed with a high salt buffer, and the lipids were then eluted with decreasing saltconcentrations. In what order would the lipids be eluted from the column? Explain your answer.
- 1. If Ser 80 was replaced with Arg what kind is the change in the bond type between these two molecules? Will this change be more or less stable. ExplainSuppose you add too much acid to your milk and the ph drops to 2.0 . would you expect casein to precipitate at that pH level ? what is the net charge ?( positive , negative , or neutral) at that pH?8. Match each of the following amino acids with the intermediate needed for its synthesis.(a) 3-Phosphoglycerate 1. glutamate 2. serine 3. asparagine (b) oxaloacetate 1. glutamate 2. serine 3. Asparagine

