4. Draw the formula of Ala-Cys at pH = pl a) b) pH > pl c) pH < pl (Ala: pK.(COOH)= 2,34, pK.(NH3") = 9,69; Cys: pK.(COOH) = 2,0, pK.(NH;") = 8,2, pK.(SH) = 10,3)
4. Draw the formula of Ala-Cys at pH = pl a) b) pH > pl c) pH < pl (Ala: pK.(COOH)= 2,34, pK.(NH3") = 9,69; Cys: pK.(COOH) = 2,0, pK.(NH;") = 8,2, pK.(SH) = 10,3)
Anatomy & Physiology
1st Edition
ISBN:9781938168130
Author:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Chapter26: Fluid, Electrolyte, And Acid-base Balance
Section: Chapter Questions
Problem 6RQ: A cation has a(n) ________ charge. neutral positive alternating negative
Related questions
Question
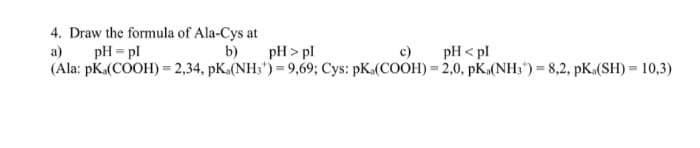
Transcribed Image Text:4. Draw the formula of Ala-Cys at
a)
pH = pl
b)
pH > pl
c)
pH < pl
(Ala: pKa(COOH) = 2,34, pK.(NH;") = 9,69; Cys: pK.(COOH) = 2,0, pK.(NH;") = 8,2, pK.(SH) = 10,3)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Anatomy & Physiology
Biology
ISBN:
9781938168130
Author:
Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:
OpenStax College

Anatomy & Physiology
Biology
ISBN:
9781938168130
Author:
Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:
OpenStax College