Q: Consider the protons that are in the position ortho- to the amine group in the structure shown…
A: Downfield means proton at higher chemical shift value and upfield means proton at lower chemical…
Q: OH dil. H,SO, (with hydride shift)
A:
Q: lease explain the answer Chlorine is an CIRCLE one ortho-para or meta director. Please include,…
A: Solution - According to the question - Given - Chlorine is an CIRCLE one ortho-para or meta…
Q: Which compound best fits this IR?
A: The compound is the 3 -methyl butanoic acid
Q: Below are two possible resonance drawings of methylvinyl ketone (MVK). In part 1 adding the missing…
A: There are two possible resonance drawings of methylvinyl ketone.
Q: Which would you predict to be more shielded, the inner or outer protons of [24]annulene?
A: NMR spectroscopy is used to detect the organic molecules on the basis of the environment of the…
Q: S n the blanks with the corect structures, Also, for each product label them syn or anti when…
A:
Q: 1, in the following compound? A Homotopic В Enantiotopic Diastereotopic D Not related at all
A:
Q: a) Draw all curved arrows that show the entire transformation of 1 into 6, and identify the entire…
A:
Q: 2. Note in the NMR spectrum above that there are two D signals and two F signals. This is because…
A: - Given compound consist of two chiral centers. - Carbons directly bonded to phenyl ring…
Q: CI, H 'N NH но.
A: Basic site is one which can donate lone pair easily. This site should be less electron-negative and…
Q: с.
A: Resonance structures are madeup via flow of electrons.
Q: (1) Explain hour Spectroscopy con disting win between (1) Benzyl Alcohol enal Methoxybenzora; (11)…
A: IR AND HNMR spectroscopy can be used to distinguish the Compounds
Q: In the following molecule, the directing effects of the two substituents are a. reinforcing b.…
A: -NH2 group is ortho & para directing and increases the electron density at ortho and para…
Q: "NH₂ NH₂ CH₂Br DMF CH₂Br (excess)? DMF ?
A:
Q: Which of the highlighted bonds absorbs at higher in an IR spectrum?
A: a.
Q: a) Aspirin Exp: H2SO4 HO.
A: A question based on IR spectroscopy that is to be accomplished.
Q: To determine whether the compound and its mirror image ar superimposable. What steps to be taken ?
A: When the mirror image and the original molecule are placed one above the other, if both the…
Q: Which highlighted bond in each pair absorbs at higher wavenumber?
A: Absorbance at a particular wave number depends upon the strength of the bond.More stronger the bond…
Q: The McLafferty rearrangement of the molecule below will produce an ion that will be detected at a…
A: McLafferty rearrangement: It is a cleavage of the α-β sigma bond by the transfer of γ-H from the…
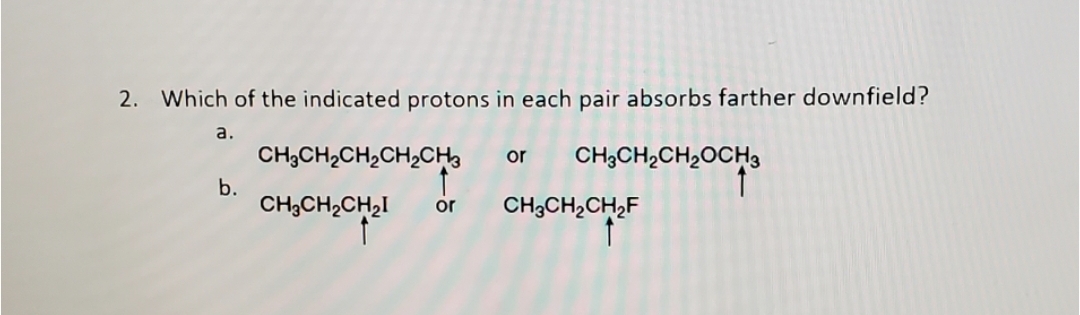
Q: Which of the labeled protons in each pair absorbs farther downfield: (a)CH3CH2CH3 or CH3OCH3; (b)…
A: NMR spectroscopy is a technique used to study the properties of organic compounds. It is a…
Q: Rank the labeled protons in the following molecule in order of increasing pKa.
A: pKa value is inversely proportional to acidity. Therefore, greater the acidity of a hydrogen less…
Q: Using only the information in the pK, table below, write an estimated pK, value for each labeled H…
A: Acidic strength of a weak acid is measured interms of Ka value , Ka is called acid dissociation…
Q: Br show homotopic protons by numbering the homotopic groups. show homotopic carbon nuclei by…
A:
Q: Which of the indicated protons will be more downfield? Why? Consider the lecture slide titled…
A: the quality of exhibiting properties with different values when measured along axes in different…
Q: Pls help me asap answer the following question, make sure your asner is correct
A:
Q: Which position(s) on the ring will be most likely to be sulfonated? Explain your reasoning for…
A: In the sulfonation Reaction, we can used sulfonating agent like SO3, py or SO3 / H2SO4. This reagent…
Q: Please help! And what is the direction of the molecular dipole moment in the chloroform molecule?…
A: NOTE: please note according to our policies , we can only answer one question at one time. you have…
Q: Which highlighted bond in each pair absorbs at higher wavenumber?
A: Stronger bonds vibrate at higher frequency. As the frequency increases wave number also increases.
Q: Which of the following reactions is a non-markonikov's? a. CH3CH=CH2 to CH3CH2CH3 b. CH3CH=CH2 to…
A: A. CH3CH=CH2 +H2 ------> CH3CH2CH3 This is hydrogenation reaction this is non markonikovs…
Q: What species is the most likely to undergo a 1,2-hydride shift? OA. В. OC. D. ÇH3 „CH CHCH3 „CHCH3…
A: The tertiary carbocation is the most stable than the secondary carbocation and primary carbocation .…
Q: Which of the following Nitrogens circled in the molecule Ranitidine is the most basic?
A: Green circled nitrogen ( left to the furan ring) will be most basic.
Q: When determing the R/S for the stereocenter, which bond do I look at next where it is circled?…
A: While giving R/S notation to a chiral carbon, we assign numbers from 1 to 4, where #1 is assigned to…
Q: Which hydrogen is the easiest to remove by homolytic bond cleavage? H2 H, all are equivalent O H1 O…
A:
Q: ou say in this picture's order and basis of the elution? Left spectra is medazepam while right is…
A: NMR is a physical phenomenon in which the nuclei are exposed to an external magnetic field and…
Q: You are labeling the H on borneol in preparation for doing 1H-NMR analysis. Would you expect the two…
A:
Q: 19. For Ha, how many non-equivalent protons are on the adjacent carbon atoms (that is, how many…
A: 19. For Ha, the number of non-equivalent protons on adjacent carbon is two(Hb ). 20 . n is the…
Q: Which ketone you think has the most stable nsystem? Explain your answer clearly based on the…
A: Aromatic compounds are stabilized very much by resonance . They follow (4n+2)π Huckel rule which…
Q: r of decreasing carbonyl stretching frequency.
A: We’ll answer the first question since the exact one wasn’t specified. Please submit a new question…
Q: 2) Which of the following compound have lower Rf value? HO,
A: 2. Rf is the Retention factor and is defined as the distance travelled by the sample divided by the…
Q: Rank the protons in the labeled CH2 groups in order of increasing acidity, and explain why you chose…
A: First consider the following compound. In this compound, CH2 is directly bonded to 2 electron…
Q: Knowing that the stereocenter is S, why does the top circle beat the bottom circle in priority since…
A:
Q: a) which bond is present in alcohols but not alkanes? b) is this band strong or weak (in terms of…
A: This question is related to IR spectroscopy. IR spectroscopy is a branch of spectroscopy that deals…
Q: cH3 Ĥ H3H CH3 -CH3 R or S ?
A: First check the correct priority order, as you have not set a correct priority order.
Q: 2.52 Rank the labeled protons in the following molecule in order of increasing pkg.
A:
Q: image of the pigments above, which is more polar? Why?
A: The biomolecules play an important role in the day to day life. The chlorophyll is used in…
Q: What is the muldiplicit, of highlighted proth.?
A: What is the multiplicity of the highlited proton?
Q: Br show homotopic protons by numbering the homotopic groups. show homotopic carbon nuclei by…
A:
Q: A B Which compound(s), if any, can be distinguished from the others by their UV-VIS spectra? None,…
A:
Step by step
Solved in 2 steps

- a. Which proton or set of protons in each of the following compounds is the least shielded?b. Which proton or set of protons in each compound is the most shielded?Using compounds m-p from image 1please fill in the columns in image 2pls give me answer of this question with explanation immiditely and i will rate you sir.
- Which of the labeled protons in each pair absorbs farther downfield: (a)CH3CH2CH3 or CH3OCH3; (b) CH3OCH3 or CH3SCH3?syringaldehyde H1 NMR structure with all H atoms and label itHello, For H-NMR, would the CH3 hydrogens adjacent to carbonyl group be more downfield or CH2 group protons in between carbonyl group and ether functionality?

