20. Which of the following is a polysaccharide with B-1,4 glycosidic linkages? C. glycogen E. None of the above A. cellulose B. lactose D. sucrose
20. Which of the following is a polysaccharide with B-1,4 glycosidic linkages? C. glycogen E. None of the above A. cellulose B. lactose D. sucrose
Biology 2e
2nd Edition
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:Matthew Douglas, Jung Choi, Mary Ann Clark
Chapter3: Biological Macromolecules
Section: Chapter Questions
Problem 10RQ: Lactose is a disaccharide formed by the formation of a bond between glucose and glycosidic; lactose...
Related questions
Question
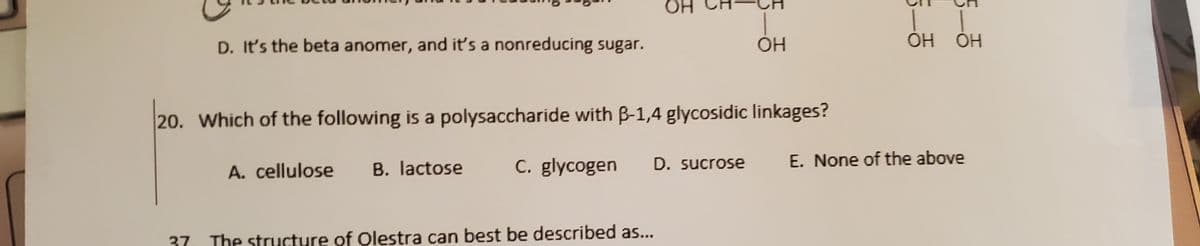
Transcribed Image Text:HO.
D. It's the beta anomer, and it's a nonreducing sugar.
20. Which of the following is a polysaccharide with B-1,4 glycosidic linkages?
C. glycogen
E. None of the above
A. cellulose
B. lactose
D. sucrose
37
The structure of Olestra can best be described as...
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax

Biology: The Unity and Diversity of Life (MindTap…
Biology
ISBN:
9781337408332
Author:
Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Publisher:
Cengage Learning

Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:
9781305389892
Author:
Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:
Cengage Learning

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax

Biology: The Unity and Diversity of Life (MindTap…
Biology
ISBN:
9781337408332
Author:
Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Publisher:
Cengage Learning

Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:
9781305389892
Author:
Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:
Cengage Learning