20.0 6mm Ha 5) What pressure (mm Hg) is required to compress 196.0 liters of air at 1.00 atmosphere into a cylinder whose volume is 26.0 liters? P, V₁ = P₂ V₂ V₁196.04
20.0 6mm Ha 5) What pressure (mm Hg) is required to compress 196.0 liters of air at 1.00 atmosphere into a cylinder whose volume is 26.0 liters? P, V₁ = P₂ V₂ V₁196.04
Introduction to General, Organic and Biochemistry
11th Edition
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Chapter5: Gases, Liquids, And Solids
Section: Chapter Questions
Problem 5.118P: 5-118 Isooctane, which has a chemical formula C8H18 is the component of gasoline from which the term...
Related questions
Question
100%
Can you help me with number 5? Was this the correct answer that shows my work includes the significant figures? Also, can you help me with Boyle's Law formula to plug in?

Transcribed Image Text:Charles's Law (temperature, volume)
1) A 550.0 mL sample of nitrogen gas is warmed from 77 °C to 86 °C.
remains constant.
C to 86 °C.
V₂ = V₁ T₂ = (550.0 ML) X
350k
T₁
564 ml
--та
2.22 L
K=c't273.15
V₁ = 550.0mL
T₁ = 77°C = 17+273) k
T₁ = 350 k
Gas Laws Worksheet T₂-86°C = 86 +273) K= 359k
(564 m²
2) A gas occupies 1.00 L at 0.00°C. What is the volume at 333.0 °C? 2 = 333.0°C (333.0+273.15k)-
V₂
Vi
7₁ V₂ =V₁ Ta
TI
Ta
t₁ = 0,00° C = (0.00+ 273.15 k) = 273.15k
2.219110379
V2= V₁T₂
1
=(1.00L) x(606.15k)
(273.15K)
= 2.222
"Patta Pa
Va
V₂=564mL
26.6 L
760.0 mm Hgx 14.0L = 26.6L)
400.0 mm Hg
Boyle's Law (pressure, volume)
3) Convert 338 L at 63.0 atm to its new volume at 1.00 atm.
Pr=nrt constant P₁V₁ = P₂ V₂, P₁.V₁
21300 L
or 2.13 x 104 L
Və
2.12:24x10
V₂ = P₁V₁ -(63.0 atm) x (3384) = 2.13 X 10 L
1.00 atm
x (359x)
7.549+m
こ
T
P₁ = 63.0 atm Pa = 1.00 atm,
V2 = 2.13x10 "L
V₁ = 338 L
4) A tank of nitrogen has a volume of 14.0 L and a pressure of 760.0 mm Hg. Find the volume of nitrogen
when its pressure is changed to 400.0 torr while the temperature is held constant.
P₁x V₁ = P₂xV₂ P₁V₁ = P₂ V₂ V₂₂ = P₁ V₁ Initial volume nitrogen (V₁) = 14.0L
Pa
Find its new volume if the pressure
= constant V₁ = V₂
Ti Ta
1.00 atm x 196.0L
26.04
= P, V,
Va
Pressure (P2) = 400.0 torr.
1 torr=1mm Hg 400.0 Jorr = 400.0mm Hg
5) What pressure (mm Hg) is required to compress 196.0 liters of air at 1.00 atmosphere into a cylinder
whose volume is 26.0 liters?
P₁V₁= P₂ V₂
V₁196.02
latm=760mm Hg P₂ = P₁V₁
5730 mmHg
Va
1₁-19 atm
V2=26.04
12=5730 mm Hg
Initial pressure nitrogen (P₁)=760.0mm Hg
Final volume (V₂)
760mm Hg
Tatm
=(5730 mm Hg
Gay-Lussac's Law (temperature, pressure)
6) A gas has a pressure if 0.0370 atm at 50.0 °C. What is the pressure at 0.00 °C?
P₁ - Pa
Pa = P₁ T₂
0.031275104
T₁ T2
T₁
0.0370 atm x 273.15K
323.15 K
.0313 atm
Pa T₂ = P₂-T₂ P₁ Pressure (Pi) = 0.0370 atm
Pa
= 7.54 atm
273,15k
L
= 0.0313 atm
Temp (Ti) = 50.0°C
(S0 + 273.15) K = 323.15K P2= 0.0313 atm
Tx
7) If a gas in a closed container, with an original temperature of 25.0 °C, is pressurized from 15.0
atmospheres to 16.0 atmospheres, what would the final temperature of the gas be?
T₁=372 25+273,15K=298,15K
Initial temp = 25c
Ta=0.00 L=0.00+ 273.15k = 273.15
318 K
Initial pressure (₁) = 15.0 atm
atm
K = C²7273,15 K 16.00 gtm x 2.98.15K - 318 K). Final pressure (P2) = 16.0
Final temperature (1₂)
T₂= Parti
15.0 atm
Ta= T₁
ра Pi
318.0266667
Expert Solution
Step 1
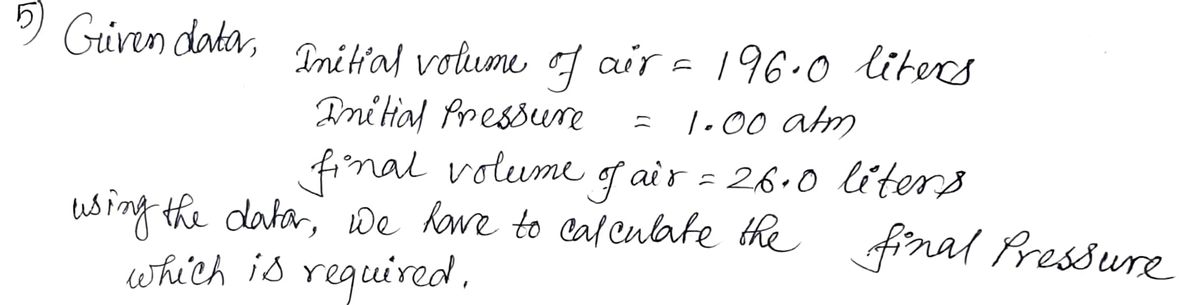
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning


Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning