Q: Given that a reaction absorbs energy and has an activation energy of 50 kJ/mol, which of the…
A: Given: Reaction absorbs energy and has an activation energy of 50 KJ/mol.
Q: The act of striking a match illustrates the role of activation energyin a chemical reaction.…
A: The act of striking a match illustrates the role of activation energyin a chemical reaction.
Q: The temperature dependence of rates of two chemical reactions was investigated experimentally. The…
A: The temperature increase increases the rate of reaction. When temperature increases, the number of…
Q: For the reversible single-step reaction given below, the activation energy of the forward reaction…
A:
Q: Describe activation energy in 20 words.
A: Since you are posted multiple questions. As per the rule, I answering the first question only.…
Q: fL was lowered by adding a catalyst, which of the following would be correct? 100 90- 80- 70- 60- 50…
A: The addition of catalyst change the activation energy by providing a new path. It changes the energy…
Q: How can the activation energy of a reaction be changed?
A: Activation energy of a reaction can be changed by using catalyst. A positive catalyst decrease the…
Q: 3. A midnight dumper discharged a tank truck full of industrial waste in a gravel pit. The truck was…
A:
Q: A catalyst increases the rate of a reaction by: increasing the temperature. В) A) decreasing the…
A: Catalyst :- A substance which when added in small amount to the reaction mixture , increases rate…
Q: Indicate whether each statement is true or false. (a) If you compare two reactions with similar…
A: The relationship between rate constant and activation energy is given by K = A e-Ea / RT where K =…
Q: Ea is the activation energy, a maximum collision energy required for a successful reaction to occur.…
A: Energitics of a reaction
Q: Is each of these statements true? If not, explain why.(a) At a given T, all molecules have the same…
A: Note - Since you have posted a question with multiple sub-parts, we will solve first three…
Q: For a reaction with reactant energy of 44 kJ and heat of reaction of 46 kJ and product energy of 90.…
A:
Q: 11. The role of catalyst is to Mark only one oval. Increase the activation energy Raise the…
A: A catalyst is a substance which does not take participate into reaction but enhances the rate of…
Q: rant to draw the energy diagram) an activation energy greater than 50 kJ/mol. absorb energy. = an…
A: As we know, For endothermic reaction, □H > 0
Q: What is the activation energy for the reverse reaction?
A: Activation energy is the energy required to reach at transition state ( activated complex) and…
Q: 15) The activation energy of a chemical reaction is the energy A) must be removed from the mixture.…
A: The activation energy of a chemical reaction is the minimum energy that is required to break the…
Q: %24 80 15- The activation energy for the forward reaction is O-25 kJ 25 kJ O-65 kJ Question 11 (1…
A: The given graph is : We have to calculate the activation energy for the forward reaction.
Q: Which of the following would change the value of the activation energy for a heterogeneous reaction?…
A: The catalyst is a chemical species that increases the rate of a chemical reaction and it does not…
Q: I Review | Constants | Periodio The rate constant of a chemical reaction increased from 0.100 s 1 to…
A:
Q: 29.What does adding a catalyst to a reaction do? Select two that apply. increase the rate of…
A: A catalyst is a substance which alters the rate of reaction without being consumed in the chemical…
Q: The change in temperature from 10 °C to 20 °C is found to double the rate of a particular chemical…
A: Rate of reaction depends on temperature . temperature increase rate of reaction also increase.
Q: 16. The activation energy of a reaction can be lowered by (a) raising temperature. (b) removing a…
A:
Q: A (aq) C (g) + (aq) (aq) Initial concentration of Initial concentration of B Initial rate of C (g) A…
A:
Q: What can be done to lower the activation energy? Question 6 options: increasing the…
A: "Catalysts " can lower the activation energy and increase the reaction rate without being consumed…
Q: I. Choose the letter of the best answer and write your answer on the space provided before the…
A: Given, Rate of a reaction increases with increase in temperature Activation energy is lowered in a…
Q: Which of the following statements is correct? a) For an endothermic reaction, the activation energy…
A: Which of the following statement is true:
Q: If L was lowered by adding a catalyst, which of the following would be correct? 70- 60- 50 40- 30-…
A: On adding catalyst, Reaction goes through alternate pathway.
Q: Which of the following changes will affect the activation energy of a reaction? 1. Increasing or…
A: Activation energy: When the reactant molecules convert into the products, they required the minimum…
Q: 3. Which of the following is NOT TRUE about a catalyst? A) It lowers the activation energy of the…
A: The catalyst is a chemical species that increases the rate of a chemical reaction and it does not…
Q: 47. Which of the following affects the activation energy of a reaction? temperature of the reactants…
A: As per bartleyby guidelines i answered only first question so please don't mind. Kindly post other…
Q: 16. Which is not a correct statement about a catalyst? A) It changes the reaction mechanism. B) It…
A:
Q: How to find activation energy using the Arrhenius Equation
A: Activation energy is the minimum energy needed by the reactants to go to product state.
Q: 1. Define the term activation energy 2. Study the graph below of a reaction 121 2.1. Calculate the…
A: Activation energy is defined as the minimum amount of energy that is required by the substituents to…
Q: uestion 3 A reaction has a rate constant of 1.24*10is at 28 "Cand 0.226/s at 76 "c What is the…
A: Arrhenius equation:If we know the value of the rate constant at two different temperatures, we can…
Q: Which of the following changes would most likely increase the rate of a reaction? a. increasing…
A: The rate of a chemical reaction helps in determining the speed of the reaction. It also tells about…
Q: a.) A substate that changes the reaction path of a reaction and is itself not consumed is called a:…
A: Hey, since there are multiple questions posted, we will answer first question. If you want any…
Q: Which one of the following statements is False? The rate constant depends on the activation…
A: Rate constant, in chemical kinetics define the rate of reaction of a chemical reaction. The factors…
Q: All the following are true statements concerning catalysts except a catalyst will be used up in a…
A: A substance that modifies the transition state to lower the activation energy is term as a catalyst.…
Q: 2. (a)How does temperature influence the rate of chemical reactions? (b) Consider the following…
A: (a) Temperature affects the rate of reactions in a directly proportional way. That is, on increase…
Q: 17. The diagram below shows the energy profile of a reaction, A+B_making C+D Potential Energy A +B C…
A: Rate constant increases exponentially when activation energy decreases.
Q: Which of the following statements are true? I. Activation energy of a reaction is smaller at higher…
A: Activation energy It is defined as the minimum energy required by the reactants so that they can…
Q: For a reaction with activation energy of 44 kJ and heat of reaction of -31 kJ and product energy of…
A: Given Activation energy = 44 kJ Energy of products = 90 kJ Heat of reaction = -31kJ Energy of…
Q: Which energy difference in the energy profile below corresponds to the activation energy for the…
A: Activation energy is defined as the minimum amount of energy required for the activation of reactant…
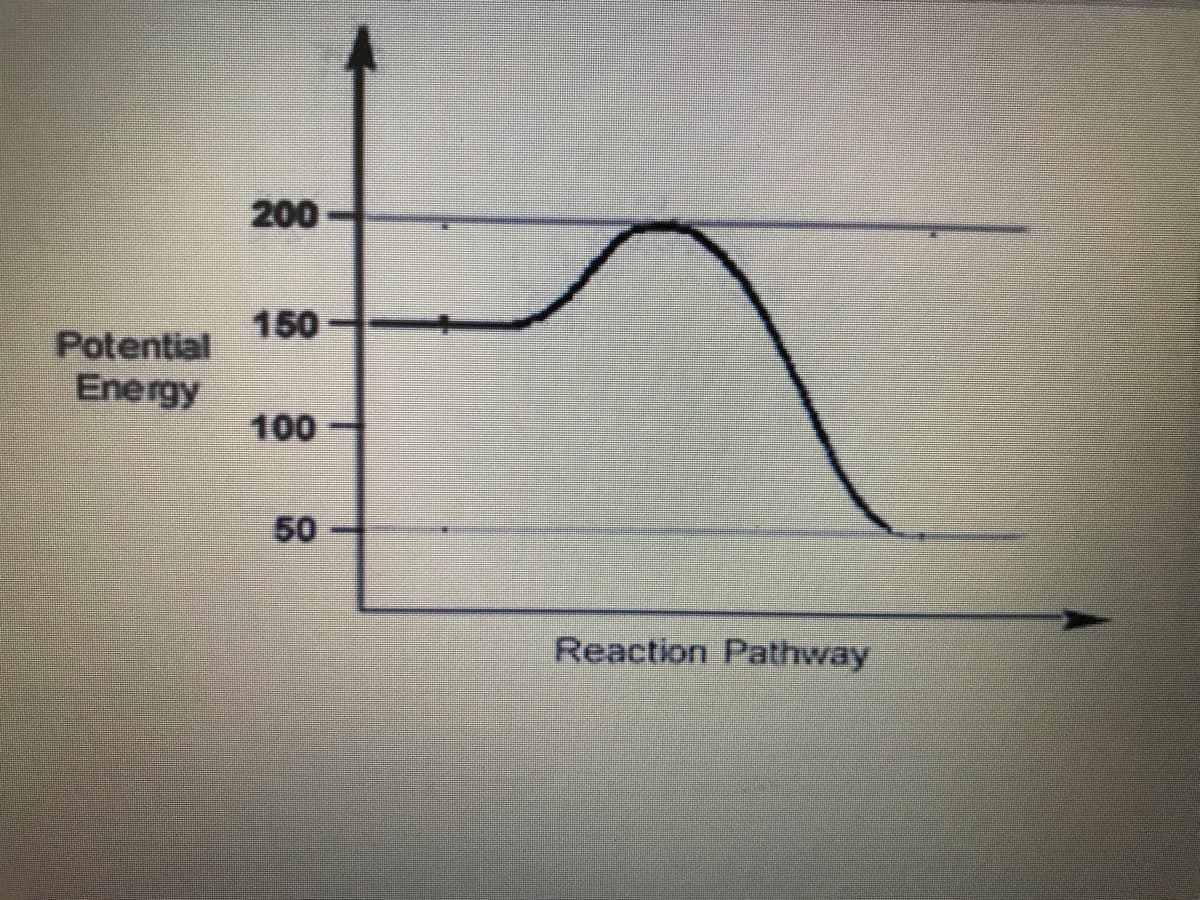
Q: This is a potential energy diagram for a particular reaction. What is the activation energy for the…
A:
Q: The rate of a particular reaction quadruples when the temperature is increased from 25°C to 35°C.…
A: Rate of a reaction is quadruples. Initial temperature = 25°C .....Or because °C + 273 = k…
Q: 51. The major reason an increase in temperature causes an increase in reaction rate is that (A) the…
A: An increase in temperature typically increase the rate of reaction .The increase in temperature will…
Q: Which of the following statements is false? Lowering the activation energy increases the rate of a…
A: We know that: Activation energy is the minimum amount of energy that is required to activate atoms…
Q: Calculate the activation energy (EA) for the forward reaction.
A:
Step by step
Solved in 2 steps with 1 images


