Q: C3H7OH ), known as rubbing alcohol, is ________ amu (rounded to one decimal place). Group of answer…
A: Molar mass of the compound can be calculated by multiplying atomic mass of each atom in the molecule…
Q: Convert the glucose concentration of a drink containing 1.750mM of glucose from mM (millimolar) to…
A: The required volume of 1.750mM of glucose solution to prepare 100 mL with a 1/100 dilution factor is…
Q: How many grams of table sugar (C12H22O11) must be added to 200 g water to raise the boiling point of…
A:
Q: i. CH4 + H20 → CO2 + H,0 j. NH3+ Cu0 → Cu + N2 + H20
A: Balance the following chemical reactions---
Q: Me. EtO₂C CO₂Et Me NaOEt EtO₂C Me
A:
Q: Cs2SO4(aq) + H20(1)
A: Option second is the correct answer. Reaction: H2SO4 (aq.) + 2CsOH(aq.) = Cs2SO4(aq.) + H2O(l)
Q: C3H6 + 02 - CO2 CO2 + 6H20 1 0
A:
Q: Which response has the correct number of significant figures and units for the following…
A:
Q: a person consumed a total of 2,191.428 calories, 215.558g comes from carbs, 71.869g comes from…
A: To Calculate the percentage of calories that comes from carbs, protein, fat, saturated fats, and…
Q: 53.62 x 10-3 O 5.362 x 10-4 O 5.362 x 104 53.62 x 103
A: The scientific notation form of 53620 is
Q: (d). 1. MgBr 1. O3 (2 equivalents) 2. Me,s 2. H20 K2Cr207 H2SO4 (2 equivalents)
A:
Q: Insulin is a protein that is used by the body to regulate both carbohydrate and fat metabolism. A…
A: Given : Volume of insulin = 125ml Concentration = 50mg/ml
Q: H20 dil. H2SO4
A:
Q: What is the average composition of the triacylglycerol that makes up soybean oil?
A: Ans: as we know that triacylglycerol contains a glycerol esterified to 3 fatty acid chain of…
Q: 4. 5. NHCOCH3 COOH ОН H20 ОН SOCI₂
A:
Q: If fifty glycerin suppositories are made from the following formula, how many milliliters of…
A: Solution Glycerol conjointly referred to as glycerin or glycerin) may be a easy polyol compound.…
Q: 3295 kcal 13786.28 kJ 0.135 5.31 x 10 J kcal Incorrect
A: Given :- 3295 kcal 5.31 × 106 J To convert :- 3295 kcal to kJ 5.31 × 106 J to kcal
Q: 1. Given the table below: Molecular Weight Range 8,000-20,000 20,000-32,000 32,000-44,000…
A: Given: Molecular weight range (g/mol) Mean Mi (g/mol) xi wi 8,000-20,000 14,000 0.05 0.02…
Q: After burning a piece of food, a student collected 12.7 mL of water and the mass of the burned food…
A: The amount of the heat needed is described as the product between the enthalpy of the vaporization…
Q: An ore of silver contains 86.7 grams of silver, Ag per ton of ore. What is the concentration of…
A:
Q: 101- 100- 1651.07cm-1. 96.38%T 541.13cm-1. 96.45%T 718.32cm-1, 95.62%T 95 90 1709.41cm-1, 88.61%T…
A: IR spectrum indicates the presence of functional groups in the compound.
Q: 2CH₂CH₂CH₂CH₂-OH H₂504 140°C H₂O +
A: Organic reactions are those in which organic reactant react to form organic products.
Q: ming equal concentratic JO suope N2OCI / HOCI (K a= 3.2 x 10-8) NH 3/ NH 4CI (K a=5.6 x 10-10) NANO…
A: Buffer is an aqueous solution of a weak acid and its conjugate base or weak base and its conjugate…
Q: What is the mass concentration of a liter of milk? A liter of milk weighs 1060 g and includes 1.6…
A:
Q: NaOH / H20 / heat H. 1) CH3I (excess) 2) Ag20 / H20 / heat
A:
Q: #78-#8 A mixture of 2.0mol H₂, 1.0mol N2, and 2.5mol NH3 is placed in a 1.0L container at 472°C.…
A: For the given equilibrium reaction we are required to find the value of reaction quotient and then…
Q: [H30)=|
A:
Q: The solubility of PbCO3 in water at 25°C is measured to be 7.3x10−5g/L. Use this information to…
A: Complete solution in attached file
Q: A food label has the following values: Total kcal=360 Total fat= 20 g. Total carbohydrate 25 g.…
A: Given, Total kcal = 360 Total fat = 20 g. Total carbohydrate = 25 g
Q: 1. MeONa, MeOH 2. H3O workup
A: Abstraction of proton by a base, -OMe
Q: Name: 1. Convert 0.85 x 10-12 km to nm. 2. Convert 190.0 m to km. 3. Convert -858 kcal to mcal.
A: 1 km = 1012 nm 1 m = 10-3 km 1 kcal = 106 mcal Note: According to Q&A guidelines of Bartleby,…
Q: NH2 Br So-H Quston it mpp
A: Applying concept of reagents and reaction.
Q: A mixture of water (D = 1,000 kg/m3) and alcohol (D = 791 kg/m3) has a density of 920kg/m3. Is this…
A: The quantities which come across during the scientific studies are named as physical quantities. The…
Q: The recommended daily allowance (RDA) of a certain amino acid is 15 mg per kg of weight. How many…
A:
Q: [H+] [OH-] pH pOH
A: Since you have asked multiparts, we will solve the first three subparts for you. If you want any…
Q: 2. Indicate the reactants and products as appropriate: LOH Hz SOy 1402- 17°c Pociy :2
A:
Q: 150 lb person uses 5.7 calories per minute when walking at a speed of 4 miles per hour how long must…
A: Given that: man use 5.7 calories per minute to run with an Speed of 4 miles per hour. Total calories…
Q: (h) AH° = -1137 kl; AS° = 0.496 kJ/K
A: The delta G is the energy required by a chemical reaction to carry out the process. It is defined as…
Q: one day a week. Their round trip to work is 6 miles. They save 12.5 gallons annually by biking one…
A:
Q: The average American woman self-reports consuming 1.785x10^3 kcal/day. For a 139.0lb woman, express…
A: Given, The average American women consumes 1.785*103 kcal/day. To convert the units, we must know…
Q: CI K2CO3 HCI H20
A: Nitrogen is less electronegative than oxygen so it can donate electrons more easily than oxygen.…
Q: How many molecules are in 25.5 g CH4? a.1.54 x 1025 molecules b. 1.56 molecules c. 6.78 x 10-22…
A:
Q: The nutrition facts label for one slice of wheat bread gives the percent daily value of various…
A: Given, A slice of bread , has calories = 76
Q: If the RDA for vitamin C is 60 MG per day and there are 70 MG of vitamin C per 100 G of orange how…
A: Given : mass of orange = 3 ounce = 3 X 28.35 = 85.05 g approx…
Q: H2O + H2O ⇔ H3O+1 + OH-1 [H3O+1][OH-1] Kw = ––––––––––––– =…
A: As per our guidelines we can only solve first three sub-parts of a question. Please resubmit the…
Q: Without using a calculator, decide which would give a significantly smaller value than 6.12 × 10-2,…
A: The number given is 6.12 X 10-2
Q: 1.345 kcal = ____ J
A: Given :- energy = 1.345 kcal To calculate :- energy in J
Q: 1. Given the table below: Molecular Weight Range 8,000-20,000 20,000-32,000 32,000-44,000…
A: Hii there, As there are multiple question posted. we are answering first question. If you need…
Q: Select from the list below, the Kavalue of acetic acid, HC2H3O2. (You will have to do a bit of…
A: Here, we have to select the Ka value for acetic acid, HC2H3O2 from the given values.
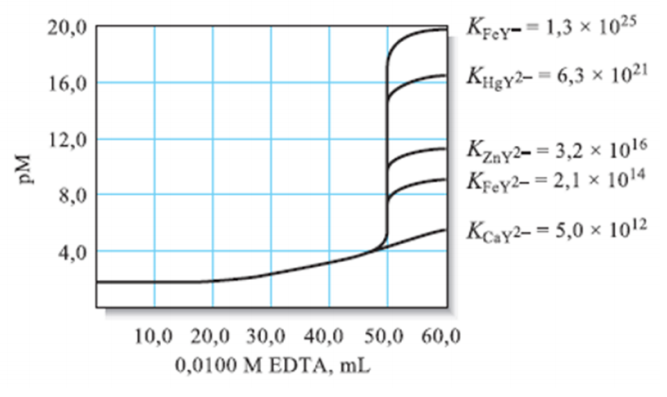
Using the graph given, which metal can be titrated better with EDTA?
Step by step
Solved in 2 steps

- Ksp for Co(OH)2 is 5.92x10^-15It is estimated that the net amount of carbon dioxide fixed byphotosynthesis on the landmass of Earth is 5.5 * 1016 g>yrof CO2. Assume that all this carbon is converted into glucose.(a) Calculate the energy stored by photosynthesis onland per year, in kJ. (b) Calculate the average rate of conversionof solar energy into plant energy in megawatts, MW11W = 1 J>s2. A large nuclear power plant produces about103 MW. The energy of how many such nuclear power plantsis equivalent to the solar energy conversion?Integrative ExercisesA wet pipette was used to transfer 10.00 mL of 1M HNO3 in the determination of ΔHrxn. [magnitude of ΔHrxn] In a maximum of 3 sentences explain why ΔHrxn decreases.
- The Ksp value of magnesium fluoride is 5.16 X 10^-11 at 25 Celcius. MgF2 (s ) -> Mg^2+ (aq) +2F^- (aq)Calculate delt g for Pbl2(s) + Pb2+(aq) +2I-(aq) T= 25C Kp= 8.7x10-9Calculate the Ksp for Ni(OH)2 Ni(OH)2(s) + 2 e- ⟶ Ni(s) + 2OH-(aq); Eo = -0.720 VNi2+(aq) + 2 e- ⟶ Ni(s); Eo = -0.250 V 7.56x1015 1.15x10-8 1.70x10-33 1.32x10-16
- Extraction of DNA involves the use of a 1 M phosphate buffer (1-liter volume) having the hydrogen ion concentration of 6.165950019 x 10-9 M. Three pKa of phosphoric acid are available Concentrated phosphoric acid (85% w/w; Sp. Gr. = 1.70; MW = 98 g/mol) Sodium dihydrogen phosphate monohydrate (MW = 138g/mol) Sodium biphosphate heptahydrate (MW = 268g/mol) Tribasic sodium phosphate monohydrate (MW = 182 g/mol) Sodium hydroxide pellets (40 grams/mol) Question: What is the pH of the solution? (answer in two decimal places) How much of the weak acid component should be used? (Note for answer: Round to the nearest hundredth and include the unit such as "grams or milliliters"; Ex. 100.00 grams or 100.00g for weight & 100.00 milliliters or 100.00 mL for volume) Question: What is the suitable buffer component to be used? (Note for the answer: Separate the two components with “and”; Ex: Ammonia and Ammonium chloride) Question: How much of the salt component should be used?…You are trying to come up with a drug to inhibit the activity of an enzyme thought to have a role in liver disease. In the laboratory the enzyme was shown to have a Km of 1.0 x 10-6 M and Vmax of 0.1 micromoles/min.mg measured at room temperature. You developed a competitive inhibitor. In the presence of 5.0 x 10-5 M inhibitor, the apparent Km of the enzyme was found to be 1.5 x 10-5 M. What is the Ki of the inhibitor?You are trying to come up with a drug to inhibit the activity of an enzyme thought to have a role in liver disease. In the laboratory the enzyme was shown to have a Km of 1.0 x 10-6 M and Vmax of 0.1 micromoles/min.mg measured at room temperature. You developed an uncompetitive inhibitor. In the presence of 5.0 x 10-5 M inhibitor, the apparent Vmax was determined to be 0.02 micromoles/min.mg. What is the Ki of the inhibitor?
- Hiii!! does anyone here can help me with this said activity? How can i know what the vid is telling ( https://youtu.be/dinNjEiqxcg , https://youtu.be/Xj0X3CS9J3k , and https://youtu.be/1IaaMeGQwdg ). thank u9. Calculate the Go in kJ/mol. 3Fe2+ (s) + 2Cr(aq) ⟶2Cr3+(aq) + 3Fe (s); Eocell=0.30 V Group of answer choices -170 +170 +87 +58 -195 10. Calculate Ecell in volts. Zn(s)|Zn2+(2.50x10-4M)||Sn2+(1.50M)|Sn(s) Eocell=+0.624 V Group of answer choices 0.736 0.635 0.512 0.848 no correct answer2 what is the purpose of BSC Level 1 & 2, proper layout, performance, and its application in biosafety assessment

