Q: Consider the reaction: C₄H₄(g) +2 H₂(g) ⇌ C₄H₈(g) Kc = 2.35 If at equilibrium the reaction vessel…
A: Given that, the reaction is:
Q: A reaction has a rate law of Rate = (1.25 M⁻²s⁻¹)[A][B]². What concentration of [B] would give the…
A:
Q: Which site will undergo most rapid substitution for an electrophile in an electrophilic aromatic…
A: ->Aromatic electrophilic substitution reaction is a type of reaction where an electrophile…
Q: 2. A solution of lithium hydroxide reacts with chromic acid in a neutralization reaction. a) Write…
A: a) The balanced chemical equation for the reaction between lithium hydroxide and chromic acid is:2…
Q: Consider the following reaction: CoCl₂(g) =CO(g) + Cl₂(g) If 1.56x10-² moles of COCI2, 0.246 moles…
A: In the given question->moles of COCl2 = 1.56 10-2 molesmoles of CO = 0.246 molesmoles of Cl2=…
Q: b. ОН
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: Draw the first step and resulting intermediate that lead to the major product. Include all lone…
A: The question is asking to give the intermediate product and the final product.The reaction will…
Q: A photon of light from a laser has an energy of 2.287 × 10−19 J. What is the total energy in two…
A: The objective of the question is to find the total energy in two moles of photons, given the energy…
Q: You find out this chemical splits apart into five ions when dissolved. You then buy a small amount…
A: mass of deicing agent = 20.0 gmass of water = 35.0 g = 0.035 kgDepression in freezing point (∆Tf) =…
Q: The equation that represents the equilibrium of phosphoric acid is H3PO4(aq) + H2O(l) ⇌…
A: The objective of the question is to identify the correct equation that represents the equilibrium of…
Q: Perform a conformational analysis of the following molecule.
A: Given is organic compound.Conformational analysis of 1-bromo-2-chloroethane to be performed…
Q: 3. Predict the major organic product for each of the following transformations. Cul Et₂O Li Br 1.2…
A: Gilman reagent is CuR2Li.Gilman reagent can be prepared by using lithium organometallic compound and…
Q: Which of the following equimolar solutions could act as a buffer system? K2SO4 (aq) and…
A: The objective of the question is to identify which of the given equimolar solutions can act as a…
Q: 50.0 ml of 0.250M hydrofluoric acid, HF, is titrated with 0.400M sodium hydroxide, NaOH. Calculate…
A: To calculate the pH of the solution after the addition of 20.0 ml of 0.400M NaOH solution to 50.0 ml…
Q: The radius of a calcium atom is approximately 0.23 nm What is the magnitude of the ionization…
A:
Q: Find the value of AG when R = 8.314JK-¹mol-¹, T= 350K 1 The equilibrium constant for a reaction is…
A:
Q: Lecture Problem 13 Write a mechanism to account for the following transformation. HO سلات H+ -OH
A: GivenDraw the mechanism.
Q: In class, we worked through mechanisms for acetal/hemiacetal and imine/enamine formation. Below,…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: OH CH3
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: Draw the product of the reaction shown below. Ign
A: Given is organic reaction.The given reaction is bromination reaction.This is a type of electrophilic…
Q: Choose all correct statements. 1. Gilman reagents react with alkyl halides, including vinyl and aryl…
A: Gilman reagents take part in coupling reactions with organochlorides. where alkyl halides,…
Q: Consider the following reaction: HC2H3O2(aq)+H2O(l)⇌H3O+(aq)+C2H3O2−(aq) where…
A: The objective of the question is to find the equilibrium concentration of H3O+ in a reaction…
Q: 7. Identify as enantiomers, diastereomers, or the same molecule OH Öx &r a.
A: Since you have posted a question with multiple sub parts, we will provide the solution only to the…
Q: The gas-phase decomposition of di-fert-butyl peroxide, (CH3)3COOC(CH3)a. is first order in the…
A:
Q: ?+ ? OH [H₂SO4] - H₂O Draw the required reactants for the reaction shown above. Edit Drawing CN
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: a NH trace H+ Draw product E. Select Draw Templates C H
A: Given,The reaction is:
Q: A) Assign R or S configuration to each chirality center in the following biological molecules HN. H…
A: Note: Since you have asked multiple questions, we will solve the first question for you. If you want…
Q: 1. threonine at acidic pH Goot H3N² -C-H1 H-C-OH 1 CH₂ Ansia srl ni einW) Covods aizenoriqonosle…
A: Note: Since you have posted a question with multiple sub-parts, we will solve the first three…
Q: Calculate the [H+] and pH of a 2.93x 10- 4 M hydrazoic acid solution. The Ka of hydrazoic acid is…
A: Answer:Ostlwald's dilution law is applicable for the dilute solutions of weak electrolytes and it…
Q: Arrange the following solutions in order of increasing hydroxide ion concentration (lowest to…
A: The objective of the question is to arrange the given solutions in order of increasing hydroxide ion…
Q: Br or DBU ? Modify the given copy of the starting material to draw the product of this reaction. Use…
A: The objective is to determine the product formed by the given reaction. Alkyl halides in the…
Q: Rank the compounds or ions in order of increasing acidity (or decreasing pK₂). CH₂ CH₂ CH₂ SH H…
A: Acidity is defined as the ability of a molecule to release protons.If a compound easily donates…
Q: Sodium ethoxide (NaOEt) is a suitable reagent to promote which mechanism(s) I. SN1 II. SN2 III. E1…
A: The objective of the question is to identify the reaction mechanisms that sodium ethoxide (NaOEt)…
Q: What is the shape and bond angle for tetrahedral shape, 120 bond angle tetrahedral shape, 109.5 bond…
A: We have been given a molecule and we have been asked to find the shape and bond angle of the…
Q: Give correct detailed Solution with explanation needed with structures..don't give Handwritten…
A: Increasing acidic order with explanation is attached with attachmentExplanation:Step 1: Step 2: Step…
Q: A solution of sulfuric acid (a strong acid) has a concentration of 0.42mol/L. Determine the pH of…
A:
Q: Which sequence correctly ranks the following dienes in order of increasing reactivity in the D-A…
A: In a Diels-Alder reaction, a diene reacts with a dienophile to form a cyclic compound known ad a…
Q: Suppose the lifetime of EACH(blue, green or black) component follows an exponential distribution…
A:
Q: A) Calculate the concentration in molality of a solution prepared by dissolving 2 mol of chloric…
A: A) B)
Q: 3. Which of the following is a diprotic acid? a) Hydrochloric acid b) Acetic acid c) Sulfuric acid…
A: Since you have posted multiple questions, we will provide the solution only to the first question as…
Q: 1. For the following intramolecular reaction, predict the product, then draw a mechanism explaining…
A: -> Mg/ether used for the formation of grignard reagent. Grignard reagent has nucleophilic…
Q: Calculate the pH of a solution that has (OH-) = 7.1 × 10–10 M. Write the answer in standard…
A: The objective of this question is to calculate the pH of a solution given the concentration of…
Q: Which of the following molecules would absorb light with a max at the lowest wavelength? A B A в C D
A: Correct answer is option D .Explanation:Step 1:We know that,lowest wavelength means it should have…
Q: Complete the balanced neutralization equation for the reaction below. Be sure to include the proper…
A: A neutralization reaction happens between an acid and a base. When acid and base react with each…
Q: At 367 K, this reaction has a Ke value of 0.0640. X(g) + 2 Y(g) 2Z(g) Calculate K₂ at 367 K. Note…
A:
Q: Consider the reaction 2NO(g)+Br2(g)⇌2NOBr(g),2NO(g)+Br2(g)⇌2NOBr(g), Kp=28.4�p=28.4 at 298 KK…
A: The objective of the question is to calculate the partial pressure of .
Q: C. HS
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: What is the mass in grams of one molecule propanol?
A: The objective of this question is to find the mass in grams of one molecule of propanol (C3H7OH).
Q: You are given two closed 1.0 L flasks (A and B) connected by a stopcock, both at room temperature…
A: Volume of both flask = 1.0 LPressure of flask A = 0.80 atmPressure of flask B = 1.5 atm
Q: Rank the following Newman projections in order of decreasing energy, from least stable to most…
A: To arrange the given Newman projections in the order of decreasing energy
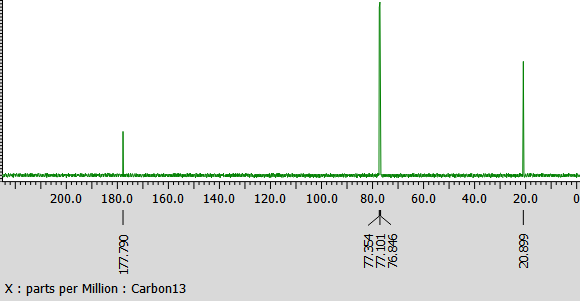
Interpret peaks on this c13 NMR of Ethanoic Acid with cooresponding structure
Unlock instant AI solutions
Tap the button
to generate a solution
Click the button to generate
a solution
- Directions Through the given situations below, answer the following questions 1 A mixture of monosodium glutamate, white sugar, salt and benzoic acid is to be melted by a group of Grade 7 students to determine the behavior of the mother during melting Predict how the mixture will look like as it is being heated. Explain your answer SUBSTANCE Sali White sugar Monosodium glutamate Benzoic acid MELTING POINT 801 C 186 C 232 C 122 C 2. Theo wants to observe what will happen to substance X (melting point is at 78 C) after heating it on an evaporating dish. After heating for a few minutes, substance X startsWhat if the second was parts per million instead of parts per billion? Will the 10 to the exponent 6 will be the one that you will always divided by in situations like this?Is glutinous rice a pure substance or a mixture?
- A student prepared a compound. the melting point of the crude the product was 42 to 48 degree C. after purifying the solid for the first time, the melting point was 47 to 51 degree C. after another purification, the melting point was 50 to 53 degree C. at this stage, can the student confidently say the product has been completely purified? justify your answerHow in the world do I solve this equation 1/k = (7.3x10^8)x(2.0x10^-5)+(1/1.00x10^-4) solve for kA solution is made by mixing 212.0 mL of ethanol initially at 10.0 C with 212.0 mL of water initially at 22.8 C what is the final temperature of the solution assuming that no heat is lost? The density of ethanol is 0.789 G/mL snd the density of water is 1.00 g/mL the specific heat of ethanol is 2.46 J/g and the specific heat of water is 4.184 J/g C.
- 2. Soap can be made using a variety of different fats, oils and bases. Suppose a student wanted to make a soft, liquid soap that lathers easily. What type(s) of fat/oil and base would you recommend? How would this differ from making a solid bar of soap? 3. Vegetable oils are typically liquid at room temperature, yet Crisco (which is made of vegetable oil) is solid at room temperature. Explain why/how this is the case. 4. Explain, with the use of a diagram how soap molecules work to clean “dirt” particles. 5. Amylase is a natural enzyme found in saliva. Describe this enzyme’s function. 6. Lactose is a sugar found in milk and other dairy products. Explain how it is broken down in most individuals. Explain lactose “intolerance”.Classify the following mixtures homogemeous or hetermgeneous? white wine?Food contains three main categories of compounds: carbohydrate, protein, and fat. Arctic explorers often eat a high-fat diet because fats have a high food value. The average food values for carbohydrate, protein, and fat are 4 Cal/g, 4 Cal/g, and 9 Cal/g, respectively. Consider a typical fat to have the chemical formula C18H36O2, glucose (C6H12O6) to be a representative carbohydrate, and the amino acid alanine (C3H6NO2) a building block of proteins. Express the food values given above in kJ/mol.
- Petroleum itself as a mixture is very useful but the components extracted from it are not beneficial. A. True B. False4. Gasoline can be reasonably approximated as C9H20 with a density of 0.718 g/mL. Diesel can be reasonable approximated as C14H30 with a density of 0.763 g/mL. (Both gasoline and diesel are complicated mixtures. These formulas are something like weighted averages of the formulas of the different components, where the weighting is based on various quantifiable behaviors. That is, using these artificial formulas for gasoline and diesel allows one to calculate average properties that match what petroleum engineers might measure.) The enthalpy change from burning one mole of gasoline is -6160 kJ. The enthalpy change from burning one mole of diesel is -7940 kJ. Determine which fuel generates the biggest enthalpy change per gram burned, and which generates the biggest enthalpy change per mL burned.The solubility of PbCO3 in water at 25°C is measured to be 7.3x10−5g/L. Use this information to calculate Ksp for PbCO3. Round your answer to 2 significant digits.











