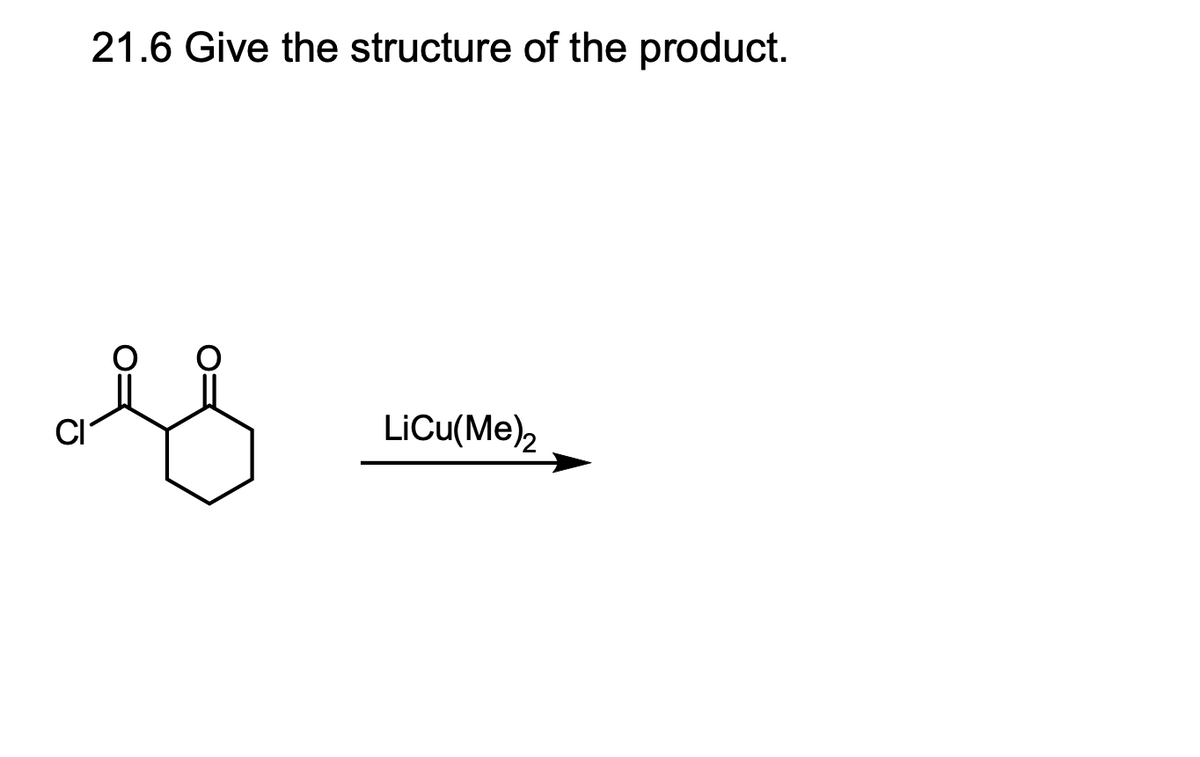
Q: 21.9 Suggest reagents for the transformation shown below. OMe ? OH
A: LiAlH4 is a strong reducing agent, it can reduce esters into alcohols.
Q: A 43.8 mL sample of a 0.200 M solution of NaCN is titrated by 0.130 M HCl. K for CN is 2.0 x 10-5.…
A:
Q: A sample containing 0.450 g of nicotinic acid, a monoprotic acid, required 34.74 mL of 0.1053 M…
A: we have to calculate the molar mass of nicotinic acid
Q: Macmillan Learning Arrange the compounds by boiling point. hexane: pentane: H₂C-CH₂-CH₂-CH₂-CH₂…
A: Since, On increasing carbon chain, boiling point will be increases.This is because on increasing…
Q: 6) What is the product of the following elimination? CI ome to do to to do ||| IV
A: In anti elimination the group opposite to each other leaves to form an alkene. The stereo of the…
Q: How many moles of HCI are there in 75.0 mL of 0.315 M HCI?
A:
Q: 3 On the plot, select the location for pka2. % of total 100 90 80 70 60 50 40 30 20 10 0 4 H₂CO3(aq)…
A:
Q: THN Br
A: The question is based on the concept of organic synthesis. we need to synthesize product from the…
Q: Consider the titration of a 29.0 mL sample of 0.170 mol L-1 CH₂COOH (K=1.3 x 10-5) with 0.120 mol…
A: Since you have posted a question with multiple sub-parts, we will provide the solution only to the…
Q: If a liquid has a low boiling point how many of these properties are likely? high intermolecular…
A: The molecules of a molecular compound are held together by a force known as intermolecular force of…
Q: Problem 23: Calculate the standard enthalpy of formation of CS₂ (1) given that: C(graphite) + O₂…
A:
Q: нахон CI (1 eq) pyridine
A:
Q: What is the molar solubility of Mg(OH)2 in wter? What would the molar solubility be in a solution…
A: We have to calculate the molar solubility
Q: 1. A student finds that 34.6 mL of 0.104 M HCI is required to titrate 20.2 mL of a NaH solution to a…
A:
Q: A 5.00 L tank at 4.58 °C is filled with 15.2 g of chlorine pentafluoride gas and 7.62 g of sulfur…
A: Ideal gas equation: PV = nRT P = Pressure V = Volume n = Number of moles R = Gas constant T =…
Q: How many moles are in 5.69×1024 atoms of iodine?
A: Mole is equal to number of atom divide by avagadro number
Q: 6 What volume of 0.220 M HBr solution (in mL) is required to obtain 0.0600 moles of HBr? A) 3.67 L…
A:
Q: How would you synthesize the following molecule (1,4-dibromobutane) starting with…
A: This problem is based on general organic chemistry reaction and its mechanism. The solution is given…
Q: O CH₂C-O_CCH₂ -CH₂CH₂OH a. Draw the structure of the tetrahedral Intermediate INITIALLY FORMED In…
A: The reaction between acetic anhydride and 2-phenyl ethanol produces 2-phenylethylacetate.
Q: Calculate [OH−] for the strong base solution formed by mixing 13.0 mL of 1.00×10−2 M Ba(OH)2with…
A:
Q: 21.1 Draw the structure. N-methylpropanamide
A:
Q: What is the pH of 0.0800 M Fe(NO3)3 (Ka of Fe3+ = 3.00x10-3)? Express your answer to two decimal…
A:
Q: H₂CO What are the functional groups of the following compounds? Br (HO)₂B.
A: In organic chemistry, a functional group is a specific group of atoms or bonds that are responsible…
Q: Part A A balloon is floating around outside your window. The temperature outside is -3 °C, and the…
A: Given Part A T = -3°C P = 0.700 atm n = 4.00 moles Part B T = 3°C V = 18.0 L n = 6.00 moles
Q: Draw a titration curve for a strong acid titrated with a strong base. On the same axes draw a…
A: Answer: When acid is titrated against base then pH of the solution rises as acid gets neutralized…
Q: Draw out the Lewis structures of these reactants and products and apply the analysis indicated in…
A: Redox reaction : The reactions in which oxidation and reduction takes place simultaneously are…
Q: Submit Answer Use the References to access Important values If needed for this question. A buffer…
A: Given => A buffer solution contains 0.449 M hydrocyanic acid(HCN) and 0.350 M Sodium cynide…
Q: What is the expected major product of the following reaction sequence? 1. -BuOK, t-BuOH 2. HBr
A: The reagent t-BuOK/t-BuOH is used for E2 elimination reaction, which results in the formation of an…
Q: An acid HX is 34% dissociated in water. If the equilibrium concentration of HX is 1.23 M, calculate…
A:
Q: 3. Complete and balance the following redox reaction a. Zn(s) → Zn(OH)42(aq) (acidic media) 210 b.…
A: Answer - Rodox reactions are those chemical reactions in which both reactions, oxidation and…
Q: To standardize a solution of NaOH, a student measured 0.326 g of potassium hydrogen phthalate…
A: The balanced chemical equation for the reaction between NaOH and KHC8H4O4 is: NaOH + KHC8H4O4 →…
Q: O Macmillan Learnin For a particular isomer of Cg H₁g, the combustion reaction produces 5099.5 kJ of…
A:
Q: 7. Propose a most efficient synthesis of this alcohol ether. For full credits, each reaction must…
A:
Q: Determine the AG in KJ for a voltaic cell made of a Ag strip and an Al strip immersed in 1.0 M…
A: Greater the value of standard reduction potential of a redox couple , stronger be the oxidizing…
Q: Use the References to access important values if needed for this question. A solution contains…
A: The question is based on the concept of solubility product principle. It that states that when a…
Q: Using this information and the image provided, describe the reaction catalyzed by an esterase and…
A: Given
Q: 3. What would be the approximate concentration of the oxalate ion (C₂04²) of a 0.020 M solution of…
A:
Q: 5. Draw product for following reaction. Br CI CI CI NaCN NaOEt DMSO EtOH NaOEt
A:
Q: If there are two weak acids, one with a pK, of 4 and another with a pK, of 9, an equimolar solution…
A: When an acid is dissolved in water, it can dissociate into its conjugate base and a hydrogen ion.…
Q: Draw a potential energy diagram for the following reaction: H2O(l) + 285.8kJ --> H2(g) + ½ O2(g)
A:
Q: Calculate the molar ratio of NaF to HF required to create a buffer with pH=4.20. (Ka(HF)=6.8×10-4 .)…
A: Answer: Buffer solution is a type of solution that resists the change in its pH on adding small…
Q: Write balanced net ionic equations for all reactions in which a precipitate formed. You must show…
A: To write the net ionic reaction in which a precipitation will take place. The cation and anions…
Q: a) Show the reaction (naming organic molecules) that would produce 4 bromo 4 phenyl n propyl…
A: The reaction that produces 4-bromo-4-phenyl-n-propylpentanote can be achieved by the esterification…
Q: Draw the major product of this reaction. Ignore inorganic byproducts. HO PCC, CH2Cl2
A: PCC(pyridinium chloro chromate) is a oxidising reagent,it oxidise primary alcohol to aldehyde…
Q: Which is not used to determine an electron configuration for an atom? Pauli exclusion principle…
A: We have to select the option which is not used to determine the electron configuration of atom
Q: You may want to reference (Pages 338-340) Section 10.5 while completing this problem Calculate the…
A: Since, At 25 °C, [H3O+] [OH-] = 10-14 Then,
Q: Give detailed Solution of all sub parts if you not then don't give answer
A:
Q: 15. Calculate the pH of a 0.10 M NH4CN solution 16. Calculate the pH of a solution prepared by…
A: The pH of a solution is a measure of its acidity or basicity. It is defined as the negative…
Q: Write the correct IUPAC name for each of the following structures
A: For the systematic name or IUPAC name of organic compounds there are some rules as follows; Choose…
Q: Calculate the change in entropy when 25 kJ of energy is transferred reversibly and isothermally as…
A: For the reversible isothermal process, the change in the entropy is given by: where qrev is the…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- show how a mass fragment of 121 could be obtained from this structure using homolytic or heteorolytic cleavage or rearranagmenet, draw mechanism using arrowsA coupling reaction between lithium dimethylcuprate and an organobromide produces the compounds shown. Draw the structure of the most efficient organobromide reactant, assuming monoalkylation occurs.Can ferrocene be acetylated twice at both iron atoms
- please explain the reaction and mechanism of sulfide to sulfimide and from sulfimide to allylic amineMost imine formation reactions are performed in the presence of molecular sieves ormagnesium sulfate. What is the purpose of these added reagents?Compound A has the molecular formula C14H25Br and was obtained by thereaction of sodium acetylide with 1,12-dibromododecane. On treatment ofcompound A with sodium amide, compound B (C14H24) was obtained. Ozonolysisof compound B gave the diacid HO2C(CH2)12CO2H. Catalytic hydrogenation ofcompound B over Lindlar palladium gave compound C (C14H26), andhydrogenation over platinum gave compound D (C14H28). Further, C yieldedO=CH(CH2)12CH=O on ozonolysis. Assign structures to compounds A through Dso as to be consistent with the observed transformations.
- tert-Butoxycarbonyl azide was developed as a reagent for peptide synthesis at OWL's home institution, the University of Massachusetts, by Prof. L.A. Carpino. It is prepared by treating tert-butoxycarbonyl chloride with sodium azide. Propose a structure for the initially-formed intermediate in this reaction.Using palmitoleic acid and neglecting stereochemistry,illustrate the reaction of palmitoletic acid with theaddition of bromineExplain in details what purpose of making a "derivative " like a semicarbizides, phenyhydrazin or DNP Solid. How is this helpful in identity your unknowns.
- Describe what the purpose of making a "derivative" like a semicarbizides, phenyhydrazin or DNP Solid. How is this helpful in identify your unknowns(s)?During purification of 2,2’-bipyridine, the compound was accidentally exposed to a mineral acid. Elemental analysis gave the followingresults: C 62.35%, H 4.71%, N 14.54%. Suggest theidentity of the isolated compoundAccount for the rapid rate of methanolysis of ClCH2OCH2CH3 even though the substrate is a primary halide. Hint: The reaction of the substrate with ethanol proceeds by SN1 mechanism. Draw the Lewisstructures.



