Part A A balloon is floating around outside your window. The temperature outside is -3 °C, and the air pressure is 0.700 atm. Your neighbor, who released the balloon, tells you that he filled it with 4.00 moles of gas. What is the volume of gas inside this balloon? Express your answer to three significant figures and include the appropriate units. ► View Available Hint(s) Vballoon = 104.5 Submit Part B Ptank = HA Previous Answers 17.62 V A 18.0 L gas cylinder is filled with 6.00 moles of gas. The tank is stored at 3 °C. What is the pressure in the tank? Express your answer to three significant figures and include the appropriate units. ► View Available Hint(s) μA P Review | Constants | Periodic Table ? ?
Part A A balloon is floating around outside your window. The temperature outside is -3 °C, and the air pressure is 0.700 atm. Your neighbor, who released the balloon, tells you that he filled it with 4.00 moles of gas. What is the volume of gas inside this balloon? Express your answer to three significant figures and include the appropriate units. ► View Available Hint(s) Vballoon = 104.5 Submit Part B Ptank = HA Previous Answers 17.62 V A 18.0 L gas cylinder is filled with 6.00 moles of gas. The tank is stored at 3 °C. What is the pressure in the tank? Express your answer to three significant figures and include the appropriate units. ► View Available Hint(s) μA P Review | Constants | Periodic Table ? ?
Introductory Chemistry: An Active Learning Approach
6th Edition
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Mark S. Cracolice, Ed Peters
Chapter4: Introduction To Gases
Section: Chapter Questions
Problem 4.1TC: Target check For each of the macroscopic charcateristics unique to the gas phase of matter described...
Related questions
Question
100%
Can someone help, and explain please,and thank you!
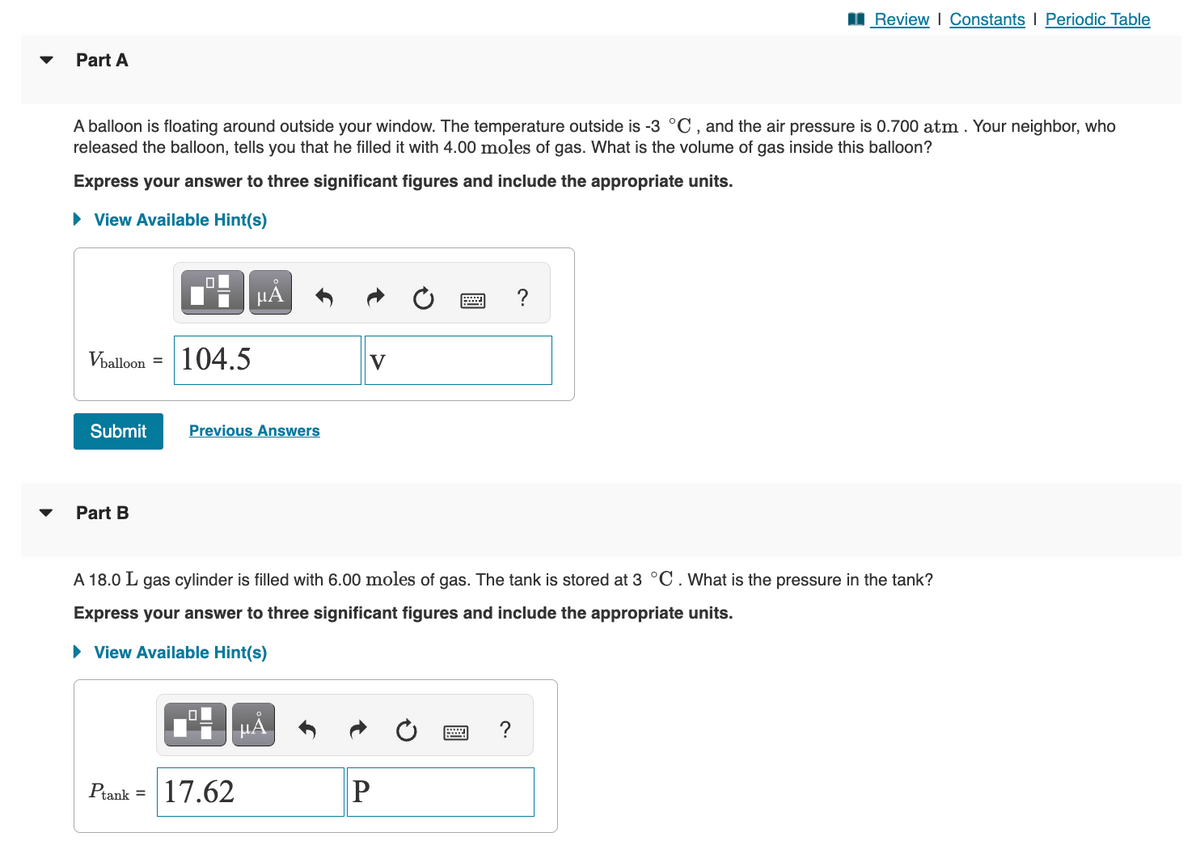
Transcribed Image Text:Part A
A balloon is floating around outside your window. The temperature outside is -3 °C, and the air pressure is 0.700 atm. Your neighbor, who
released the balloon, tells you that he filled it with 4.00 moles of gas. What is the volume of gas inside this balloon?
Express your answer to three significant figures and include the appropriate units.
► View Available Hint(s)
Vballoon =
Submit
Part B
0 μÅ
Ptank =
104.5
Previous Answers
17.62
V
μĂ
A 18.0 L gas cylinder is filled with 6.00 moles of gas. The tank is stored at 3 °C. What is the pressure in the tank?
Express your answer to three significant figures and include the appropriate units.
► View Available Hint(s)
2
P
www.
Review | Constants | Periodic Table
?
?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning