27. A seguir é mostrado o espectro de RMN de um éster com fórmula C,H,O,. O espectro no infravermelho apresenta bandas de intensidade média em 3270 e 2118 cm. Desenhe a es- trutura do composto. CşH,O2 2,10 1,01 3,15 50 40 3.5 30 23 20 15
27. A seguir é mostrado o espectro de RMN de um éster com fórmula C,H,O,. O espectro no infravermelho apresenta bandas de intensidade média em 3270 e 2118 cm. Desenhe a es- trutura do composto. CşH,O2 2,10 1,01 3,15 50 40 3.5 30 23 20 15
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter18: Electrochemistry
Section: Chapter Questions
Problem 29Q: When jump-starting a car with a dead battery, the ground jumper should be attached to a remote part...
Related questions
Question
27. The NMR spectrum of an ester with formula O spectrum in the Infrared has medium intensity bands at 3270 and 2118 cm*1. draw the structure of the compound. note: point out the structures on the chart for better understanding
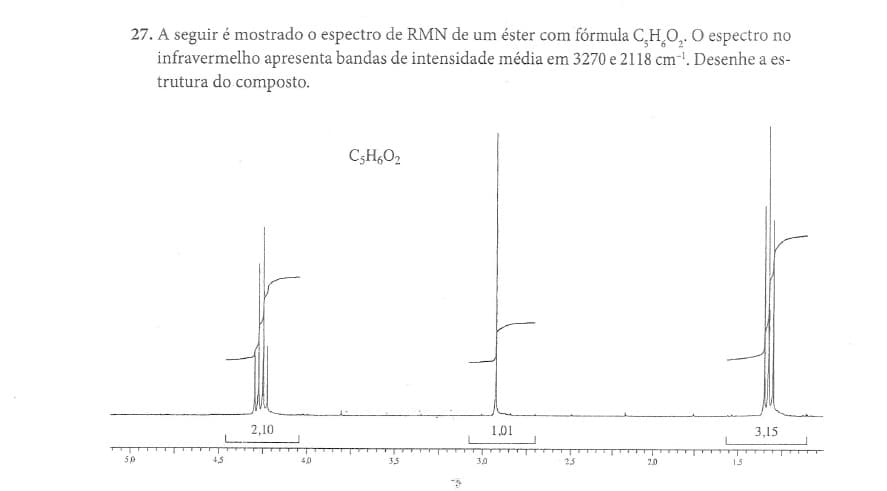
Transcribed Image Text:27. A seguir é mostrado o espectro de RMN de um éster com fórmula C,H,O, O espectro no
infravermelho apresenta bandas de intensidade média em 3270 e 2118 cm-. Desenhe a es-
trutura do composto.
C3H,O2
2,10
1,01
3,15
50
4,5
40
3,5
3,0
23
20
15
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax