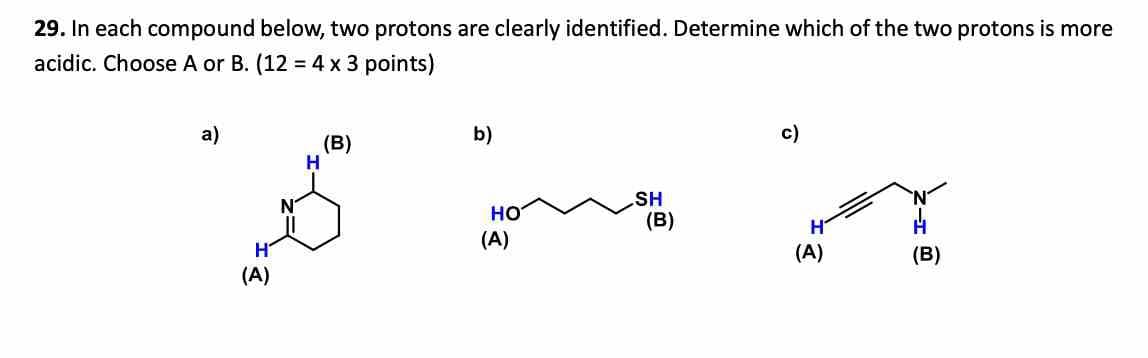
29. In each compound below, two protons are clearly identified. Determine which of the two protons is more acidic. Choose A or B. (12 = 4 x 3 points) a) (B) b) H (A) HO (A) c) SH (B) (A) (B)
Q: None
A: a. 1,2-dihydroxybenzene (common name: catechol) - This compound is a benzene ring with two hydroxyl…
Q: 5 Worksheet: Chirality - Handedness of Molecules 1 Label the chirality center/stereocenter in the…
A: Chiral Centre is that centre which have all the different groups around it.In case of carbon, if all…
Q: Show the products of the following reactions
A:
Q: 2. Predict the products. (Hint: for the 2rd one, remember the driving force for a Claisen reaction,…
A: The first reaction involves treating a compound with LDA (lithium diisopropylamide) followed by MeI…
Q: Reaction of Aldehydes and Ketones with Organometallics: Grignard and Lithium For the following…
A: Step 1: Step 2: Step 3: Step 4:
Q: Chemistry
A: To convert the given alcohol into the required product, two reactions are involved. Step 1: The…
Q: *Reaction is in image* Questions based on reaction in image: The oxidizing agent is: The reducing…
A:
Q: None
A: Certainly, let's draw the structure of (2R, 5R) - 2 - bromo-5-methylheptanal:1. **Base Chain:** The…
Q: None
A: In urea (H₂NCONH₂), the carbon atom is situated within the central molecular structure, forming…
Q: Show the products of the following reactions
A:
Q: Supply the missing isotope in the nuclear equation below. When entering isotopes, use the text…
A:
Q: Predict the major products of this organic reaction. NBS ? hv
A:
Q: 1. σ=[(Σd2)/(n-1)]½ n= number of trials d= measured value - mean…
A: There are two formulas in finding Standard Deviation, population standard deviation denoted by σ…
Q: Help calculate.
A: Step 1: Step 2: Step 3: Step 4:
Q: Which of the following is a neutral ion? O Ca2+ ○ W6+ O NO₂ OHSO4 CIO
A: Neutral anion are those which come from strong acid whereas neutral cation are those which come from…
Q: Can you write down retrosynthesis ( RSA)and show pilar dont provide hnadwriting solution...
A: Step 1: Bromobenzene's Retrosynthesis : * Target Molecule : C6H5Br (bromobenzene). * Functional…
Q: ABCD Question 2 Please predict the product for each of the following reactions. Make sure to clearly…
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: Using these metal ion/metal standard reduction potentials Cd2+(aq)|Cd(s)…
A: Standard reduction potentialsCd2+ + 2e- → Cd E˚ = -0.40 VCu2+ + 2e- → Cu…
Q: Here is the liquid water is 4.18 J/g °C. What is the amount of heat energy that is associated with…
A:
Q: None
A: The reactant is an organic molecule with an amine (NH2) group and a ketone (C=O) group on adjacent…
Q: 5. (8 pts) Draw both chair conformers for the following compounds. Circle the one that is the most…
A: Solution: I can't physically draw, but I will describe the chair conformers for each compound and…
Q: None
A: Step 1: Introduction:Dipole-dipole interactions: These forces arise between molecules that have…
Q: To preview image click here Give a curved arrow pushing mechanism for the following reaction,…
A: Step 1:Carboxylic Acids.1) Draw the general structure of a carboxylic acid Step 2:2) T/F -…
Q: C. Br HO cat. H3O HO Racemic Mixture Compound 1 H Mg Compound 2 2) Referring to the above scheme in…
A:
Q: None
A: Step 1: Step 2: Step 3: Step 4:
Q: How would you answer part III of this FRQ? this is a non graded practice.
A: (iii) Polar and Non-Polar Molecules: Iodine Monofluoride (IF₅):Iodine monofluoride has a trigonal…
Q: Please show the mechanism for the reagents p-nitrobenzyl ester AND p-Bromophenacyl ester. The…
A: Step 1: Step 2: Step 3: Step 4:
Q: Identify the Haworth structure showing the ẞ-isomer of the following Fischer projection. H (Hint:…
A: Beta form -OH group is above the plane, here there are two form in which OH is above the palne . A…
Q: Name: Formula of a Hydrate Lab Chemistry Date: Per._ 6. The following data were obtained when a…
A: 6)Given mass of empty test tube is 18.42g.Mass of test tube and hydrate(before heating) is…
Q: A hot 120.8 g lump of an unknown substance initially at 176.2 °C is placed in 35.0 mL of water…
A: For No. 1: The amount of energy in the form of heat that is released by the hot lump of unknown…
Q: Complete solutions need . handwriting
A: The solutions outlined here are determined by standard procedures for preparing solutions using the…
Q: please answer in text form and in proper format answer with must explanation , calculation for each…
A: Approach to solving the question:NoneDetailed explanation:NoneExamples:NoneKey references:None
Q: None
A: An electron's state is described by four quantum numbers:Principal quantum number (n): Defines the…
Q: The main hormonal organs of the gonadal system are: Question 9 options:…
A: The gonadal system refers to the reproductive system's organs that are responsible for producing…
Q: Please explain this question
A: Entropy, denoted by S, is a measure of the number of specific ways in which a system may be…
Q: 16.36 For the reaction 2A+B+C+D+ 2E, data for a run with [A], 800 mmol/L and [B] = 2.00 mmol/L are…
A: Step 1:Step 2: Step 3: Step 4:
Q: How many protons are in the Pb-208 isotope? 126 208 82 290
A: A. How many protons are in Pb-208 isotope?-Pb corresponds to element Lead(Pb).-Pb on the periodic…
Q: Solve all complete solutions
A: Here are the IUPAC names and structures of the esters provided:Propyl propanoate:…
Q: CIO (aq) + Cr(OH)3(s) CrO42(aq) + Cl¯(aq) basic What is the standard cell potential, E° cell, for…
A: Step 1:The standard electrode potential, often referred to as standard potential, indicates the…
Q: Calculating the pH of a 0.1 L aqueous solution of HA 2.50 10 - 3M (Ka = 1.74 10 -6) requires…
A: To calculate the pH of the given aqueous solution of HA, we'll use the acid dissociation constant…
Q: The equation of tris(1, 10-phenanthroline)iron(II) in acid solution takes place according to the…
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: Question 2 Which set of Newman projections represents correct the most stable and the least stable…
A: Because it's most stable conformer is arranged in anti form in which both CH3 and C2H5 are opposite…
Q: In the following FTIR Spectrum, what functional group can you recognize?
A: Step 1:Given Infrared spectrum of an unknown molecule. Determine the functional group present in the…
Q: Predict the products of the following reactions. Show stereochemistry when applicable.
A: #1 is a hydroboration-oxidation reaction. it is a reaction that uses anti-Markovnikov addition of…
Q: 9. Draw the following chemical reactions. a) cyclopentylamine + water b) cyclopentylamine + butanoic…
A: Step 1: Step 2: Step 3: Step 4:
Q: Draw the structure(s) of the major organic product(s), including counterions, of the following…
A: I can help you with this chemistry question which asks you to draw the structure of the major…
Q: A chemist wishes to convert iron into iron(II) bromide using this reaction: Fe(s) + 2 HBr(aq). →…
A: Heat the reaction mixture and increase the HBr concentration can speed up the reaction. Heat the…
Q: I really need help on how to solve this problem ! I would appreciate any help :)
A: Step 1:Enthalpy change value for any reaction…
Q: Draw a Haworth projection of a common cyclic form of this monosaccharide:
A: The sugar in open chain form is a 5-carbon aldose (aldopentose) known as D-Arabinose.It is…
Q: Please don't provide handwritten solution ....
A: Step 1: **a) CH3CH=CH=OH*** Determine the functional group: An enolate ion, which has a…
Step by step
Solved in 2 steps with 1 images

- Camphor, a saturated monoketone from the Asian camphor tree, is used among other things as a moth repellent and as a constituent of embalming fluid. If camphor has M+=152.1201 by high-resolution mass spectrometry, what is its molecular formula? How many rings does camphor have?Which of the following protons will be the most deshielded - Ha, Hb, or Hc? Explain your answerDraw the structure of the important peaks in the mass spectrum of C9H8O (unsaturation = 6) Major Peaks: m/z = 132, 103, 77, 53
- For each compound, assign likely structures to the fragments at each m/z value, and explain how each fragment is formed.a. C6H5CH2CH2OH: peaks at m/z = 104, 91b. CH2 = C(CH3)CH2CH2OH: peaks at m/z = 71, 68, 41, 31How many types of non-equivalent protons are on fluorobenzene? 2 5 4 3 6Draw structures for a saturated hydrocarbon that has a molecular ion with an m/z value of 128.
- A compound of formala ( C20H29NO) absorbs 4 molar equivalents of hydrogen. How many rings does it contain ? select one : A) 3 B) 2 C) 1 D) 4Begin by explaining the criteria for interpreting CNMR Complete the table below, and provide a structure of a compound that is consistent with molecular formulaC 9 H 18 O 2 and the following 13 C DEPT data. Please show your work and write your final answer inthe box below.The mass spectra of two very stable cycloalkanes both show a molecular ion peak at m/z = 98 . One spectrum shows a base peak at m/z = 69, the other shows a base peak at m/z = 83. Identify the cycloalkane.
- How could you use 1H and 13C NMR to help distinguish the following isomeric compounds of the formula C4H8?Assign a priority order (from least to highest priority) to each of the following sets of groups: I. -OCH II. -NH(CH3)2 III. CH2NH2 IV. -OH A. IV, I, III, II B. III, II, IV, I C. II, III, IV, I D. III, IV, I, IIWhat is the structure of the compound if the mass spectrum shows M+ = 72, IR shows peak near 1720 cm-1, 13C-NMR shows 4 lines. The proton NMR is: 2.4 ppm (2H quartet) 2.1 ppm (3H singlet) 1.1 ppm (3H triplet)

