2CIO2 (aq) + 2 OH" (aq) – CIO3 (aq) + CIO2" (aq) + H20 (1) Initial [CIo,1 (M) Initial [OH] (M) Initial Rate (M/s) Experiment 1 Experiment 2 Experiment 3 0.020 0.030 0.00276 0.060 0.030 0.0248 0.020 0.090 0.00828 Determine the following: Reaction order in terms of [CIO21 Reaction order in terms of [OH]_ Overall reaction order Rate law expression. Rate constant (k) value
2CIO2 (aq) + 2 OH" (aq) – CIO3 (aq) + CIO2" (aq) + H20 (1) Initial [CIo,1 (M) Initial [OH] (M) Initial Rate (M/s) Experiment 1 Experiment 2 Experiment 3 0.020 0.030 0.00276 0.060 0.030 0.0248 0.020 0.090 0.00828 Determine the following: Reaction order in terms of [CIO21 Reaction order in terms of [OH]_ Overall reaction order Rate law expression. Rate constant (k) value
Chemical Principles in the Laboratory
11th Edition
ISBN:9781305264434
Author:Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Chapter21: Rates Of Chemical Reactions, Ii. A Clock Reaction
Section: Chapter Questions
Problem 2ASA
Related questions
Question
![Using the experimental data, fill in the blanks below which describe the simple rate
law. Again, this may be easier to submit through your handwritten work only. You can
type "see handwritten work" below if needed.
2CIO2 (aq) + 2 OH" (aq) – CIO3 (aq) + CIO2" (aq) + H20 (I)
Initial [C0,1 (M)
Initial (OH] (M)
Initial Rate (M/s)
Experiment 1
Experiment 2
Experiment 3
0.020
0.030
0.00276
0.060
0.030
0.0248
0.020
0.090
0.00828
Determine the following:
Reaction order in terms of [CIO2 _-
Reaction order in terms of [OH]_.
Overall reaction order
Rate law expression.
Rate constant (k) value](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2F8b2b1966-cbdc-46cc-aafa-333a90f3e9b8%2F094a3290-5562-477b-92ed-ec59ce4de8d0%2F72dii2q_processed.jpeg&w=3840&q=75)
Transcribed Image Text:Using the experimental data, fill in the blanks below which describe the simple rate
law. Again, this may be easier to submit through your handwritten work only. You can
type "see handwritten work" below if needed.
2CIO2 (aq) + 2 OH" (aq) – CIO3 (aq) + CIO2" (aq) + H20 (I)
Initial [C0,1 (M)
Initial (OH] (M)
Initial Rate (M/s)
Experiment 1
Experiment 2
Experiment 3
0.020
0.030
0.00276
0.060
0.030
0.0248
0.020
0.090
0.00828
Determine the following:
Reaction order in terms of [CIO2 _-
Reaction order in terms of [OH]_.
Overall reaction order
Rate law expression.
Rate constant (k) value
Expert Solution
Step 1
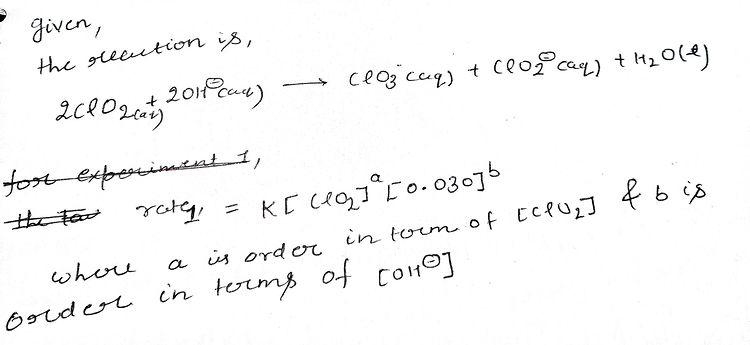
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole

Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole